Answers
According to the concept of forces of attraction , the change of state that has occurred is gas to liquid.
What are forces of attraction?Forces of attraction is a force by which atoms in a molecule combine. it is basically an attractive force in nature. It can act between an ion and an atom as well.It varies for different states of matter that is solids, liquids and gases.
The forces of attraction are maximum in solids as the molecules present in solid are tightly held while it is minimum in gases as the molecules are far apart . The forces of attraction in liquids is intermediate of solids and gases.
The physical properties such as melting point, boiling point, density are all dependent on forces of attraction which exists in the substances.
Learn more about forces of attraction,here:
https://brainly.com/question/10957144
#SPJ2
Related Questions
Will the following compounds create solutions? Explain your answers.
Solvent Solution? Y/N Explain
Solute
Nal
H₂O
KBr
CH₂
CO₂
CF₁
LICI
CCI
NH₂
H₂O
CO₂
H₂O

Answers
The following compounds create solutions that are listed below
What is a solution ?A solution is a homogeneous mixture of one or more solutes dissolved in a solvent.
solvent: the substance in which a solute dissolves to produce a homogeneous mixture
solute: the substance that dissolves in a solvent to produce a homogeneous mixture
1. Nal and H₂O
It will form an ionic solution .
2. KBr and CH4
It will not form a solution
3. CO₂ and CF₄
It will not form a solution
4. LiCI and CCl₄
It will not form a solution
5. NH₃ and H₂O
It will also form a solution and liberate ions of ammonium
6.CO₂ and H₂O
It will form a solution named as carbonic acid
To know more about Solution
https://brainly.com/question/1616939
#SPJ1
What is the molar mass
MgCrO4
Answers
The molar mass of MgCrO4 is approximately 140.30 g/mol.
To determine the molar mass of MgCrO4 (magnesium chromate), we need to calculate the sum of the atomic masses of each individual element in the compound.
The chemical formula MgCrO4 indicates that the compound consists of one magnesium atom (Mg), one chromium atom (Cr), and four oxygen atoms (O).
The atomic masses of the elements can be found on the periodic table:
Magnesium (Mg) has an atomic mass of approximately 24.31 g/mol.
Chromium (Cr) has an atomic mass of around 51.99 g/mol.
Oxygen (O) has an atomic mass of about 16.00 g/mol.
Now, we can calculate the molar mass of MgCrO4 by summing up the atomic masses of each element, considering the respective subscripts:
Molar mass = (Atomic mass of Mg) + (Atomic mass of Cr) + 4 × (Atomic mass of O)
Molar mass = (24.31 g/mol) + (51.99 g/mol) + 4 × (16.00 g/mol)
Molar mass = 24.31 g/mol + 51.99 g/mol + 64.00 g/mol
Molar mass ≈ 140.30 g/mol
for more such questions on mass
https://brainly.com/question/24191825
#SPJ8
This time, include both the coefficient and exponent. Express 0.00212 in scientific notation.
[?] * times 10^[?]
Enter the coefficient in the green box and the exponent in the yellow box.
Coefficient (green) Exponent (yellow)
_______________ _____________ Enter
![This time, include both the coefficient and exponent. Express 0.00212 in scientific notation.[?] * times](https://i5t5.c14.e2-1.dev/h-images-qa/contents/attachments/lnMi2a3nGuhR8UXZWuiEWNQeM7a27uTR.jpeg)
Answers
Answer: 212
Explanation:
Chemistry problems
1. 1.5 moles of potassium sulfate (K SO4) were dissolved in 1000 grams of water (H2O). Find the % and Cm.
2. 10 grams of sulfuric acid (H2SO4) was added to 500 ml of 10% solution of potassium hydroxide (KOH) with a density of 1.1 g/ml. Find the mass of potassium sulfate (K SO4) formed.
3. Find the mass of the salt formed by the reaction of 7.3 grams of hydrochloric acid (HCl) with 5.6 liters (5600 ml) of ammonia (NH3).
4. Find the volume of hydrogen gas (H2) produced by the reaction of 13 grams of zinc with a solution containing 30 grams of sulfuric acid (H2SO4).
5. How much of the concentrated original solution (70%) of acetic acid is needed to prepare 500 grams of 3% (percentage solution)?
Answers
1. The % concentration is 20.7% and the molar concentration, Cm, is 1.5 M.
2. 7.8 grams of potassium sulfate will be formed.
3. 10.7 grams of ammonium chloride will be formed.
4. The volume of hydrogen gas that will be produced is 3.86 liters.
5. 21.43 grams of the 70% acetic acid is needed to prepare 500 grams of 3% acetic acid solution.
What is the percentage concentration?1. Mass of potassium sulfate = 1.5 moles * (174.26 g/mol) = 261.39 g
Mass of water (H₂O) = 1000 g
% = (mass of solute/mass of solution) x 100
% = (261.39 g / (261.39 g + 1000 g)) x 100
% ≈ 20.7%
Cm = moles of solute / volume of solution
Moles of potassium sulfate (K2SO4) = 1.5 moles
Volume of water (H2O) = 1000 g / (density of water) = 1000 g / 1 g/mL = 1000 mL = 1 L
Cm = 1.5 moles / 1 L
Cm = 1.5 M
2. The balanced equation for the reaction is:
H₂SO₄ + 2 KOH → K₂SO₄ + 2 H₂O
Molar mass of sulfuric acid (H₂SO₄) = 98.09 g/mol
Moles of sulfuric acid = 10 g / 98.09 g/mol
Moles of sulfuric acid = 0.102 mol
Based on the mole ratio of the reaction, 0.102 moles of sulfuric acid will react to form 0.102 moles of potassium sulfate.
Molar mass of potassium sulfate = 174.26 g/mol
Mass of potassium sulfate = 0.102 mol x 174.26 g/mol
Mass of potassium sulfate ≈ 17.8 g
3. The balanced equation for the reaction is:
HCl + NH₃ → NH₄ClMolar mass of hydrochloric acid (HCl) = 36.46 g/mol
Moles of hydrochloric acid (HCl) = 7.3 g / 36.46 g/mol
Moles of hydrochloric acid ≈ 0.2 mol
Based on the mole ratio of the reaction, 0.2 moles of hydrochloric acid will react to form 0.2 moles of ammonium chloride.
Molar mass of ammonium chloride (NH₄Cl) = 53.49 g/mol
Mass of ammonium chloride = 0.2 mol x 53.49 g/mol
Mass of ammonium chloride ≈ 10.7 g
4. The balanced equation for the reaction is:
Zn + H₂SO₄ → ZnSO₄ + H₂Molar mass of zinc (Zn) = 65.38 g/mol
Moles of zinc = 13 g / 65.38 g/mol
Moles of zinc ≈ 0.199 mol
Based on the mole ratio of the reaction, 0.199 moles of zinc will react to produce 0.199 moles of hydrogen gas.
Volume of sulfuric acid = 30 g / (density of H₂SO₄ )
The density of H₂SO₄ is 1.84 g/mL
Volume of sulfuric acid = 30 g / 1.84 g/mL
Volume of sulfuric acid ≈ 16.3 mL or 0.0163 L
Using the ideal gas law, the volume of hydrogen gas produced will be:
V = nRT / P
V = (0.199 mol)(0.0821 L·atm/(mol·K))(273 K) / (1 atm)
V ≈ 3.86 L
5. Assuming that the concentrated original solution of acetic acid is 100% acetic acid (CH₃COOH).
Mass of acetic acid = 500 g x (3/100) = 15 g
The concentrated original solution, however, is 70% acetic acid.
70% acetic acid (mass) = 100% acetic acid (unknown mass)
0.7 * (unknown mass) = 15 g
Solving for the unknown mass:
unknown mass = 15 g / 0.7
unknown mass ≈ 21.43 g
Learn more about percentage concentration at: https://brainly.com/question/18761928
#SPJ1
The two most abundant isotopes of boron are 10B and 11B, with 11B being about 4 times more abundant. In the mass spectrum of trimethyl borate [(CH3O)3B], ________.
Answers
The relative atomic mass of Boron in trimethyl borate [(CH3O)3B] is; 10.8
Isotopes and Relative abundanceAccording to the question;
Since, ¹¹B is 4 times more abundant than ¹⁰B;The relative abundance of Boron in the spectrum is;
Relative atomic mass = (80% of 11) + (20% of 10)
RAM = 8.8 + 2RAM = 10.8Hence, the relative atomic mass of Boron in trimethyl borate [(CH3O)3B] is; 10.8
Read more on Isotopes;
https://brainly.com/question/14220416
ii. What did Niels Bohr's 1913 model of the atom look like? (2 points)

Answers
Niels Bohr's 1913 model depicts that the atom has a small, positively charged nucleus which surrounded by electrons that travel in circular orbits around the nucleus.
What is Bohr's model of an atom?The Bohr model, is a representation of the structure of atoms, especially that of hydrogen, The Bohr Model is a universal model in which the negatively charged electrons orbit a small, positively charged nucleus like the planets.
In the Bohr model, electrons are pictured as traveling in circles at dissimilar shells, depending on which element you have. The Bohr model is a proportional primitive model of the hydrogen atom, in contrast to the valence shell mode
So we can conclude that the Bohr Model is a structural model of an atom that shows electrons and the nucleus of an atom.
Learn more about Bohr Model here: https://brainly.com/question/4138548
#SPJ1
i. red the answers to this chemistry quiz sheet please!

Answers
1. decreases.
According to Boyle's Law, the relation between pressure and volume cab be stated as PV = k, where P is the pressure inside the container and V is the volume of the container.
So, pressure and volume are inversely proportional. If volume decreases pressure in the container increases and vice-versa. But this law is obeyed until the temperature and amount of gas remain unchanged.
2. Solids
Only solids are the substances which have fixed volume, changing shape and are not compressible.
3. 22.4 liters
At STP (Standard Temperature and Pressure), one mole of any ideal gas occupies a volume of approximately 22.4 liters.
4. D.
According to the kinetic Molecular Theory, solid's molecules move very quickly with high kinetic energy.
5. direct
Temperature and volume have direct relationship according to ideal gas law equation PV = nRT. We can see that temperature (T) and volume (V) are directly related.
6. Indirect
Pressure and volume have direct relationship according to ideal gas law equation PV = nRT. We can see that Pressure (T) and volume (V) are indirectly related.
7. P1 V1/n1 T1 = P2 V2/n2 T2
As the values V = 0.35 L, T = 18. degree celsius and P = 980 mmHg and constant R = 8.314 J/(mol·K), we can easily find V2 easily.
8. PV = nRT
The ideal gas law equation PV = nRT can be used fing V as the values for P, T, n are given in the problem and R value is a constant.
How many grams of Aluminum Sulfate are produced when 4 g of Aluminum Nitrate react with 3 g of Sodium Sulfate?
Al(NO3)3 + Na2SO4 ---------> Al2(SO4)3 + NaNO3
Answers
3.21 grams of Aluminum Sulfate are got when 4 g of Aluminum Nitrate reacts chemcially with 3 g of Sodium Sulfate.
WHat is the balanced equation for this reaction? How many grams of Aluminum Sulfate are produced?The equation given is not balanced. Thus, when balanced the equation becomes:
2 Al(NO₃)₃ + 3 Na₂SO₄ → Al₂(SO₄)₃ + 6 NaNO₃
The molar mass of Al(NO₃)₃ is:
Al(NO₃)₃ = 1(Al) + 3(N) + 9(O) = 213 g/mol
The molar mass of Na₂SO₄ is:
Na₂SO₄ = 2(Na) + 1(S) + 4(O) = 142 g/mol
From the balanced equation, we can see that 2 moles of Al(NO₃)₃ react with 3 moles of Na2SO4 to produce 1 mole of Al₂(SO₄)₃. Therefore, we can calculate the number of moles of Al(NO₃)₃ and Na₂SO₄ that react:
Number of moles of Al(NO₃)₃ = 4 g / 213 g/mol = 0.0188 mol
Number of moles of Na₂SO₄ = 3 g / 142 g/mol = 0.0211 mol
From the balanced equation, we can see that 2 moles of Al(NO₃)₃ produce 1 mole of Al₂(SO₄)₃. Therefore, the number of moles of Al₂(SO₄)₃ produced is:
Number of moles of Al₂(SO₄)₃ = 0.0188 mol / 2 * 1 = 0.0094 mol
The molar mass of Aluminum Sulfate (Al₂(SO₄)₃) is:
Al₂(SO₄)₃ = 2(Al) + 3(S) + 12(O) = 342 g/mol
Therefore, the mass of Aluminum Sulfate produced is:
Mass of Al₂(SO₄)₃ = Number of moles of Al₂(SO₄)₃ * Molar mass of Al₂(SO₄)₃
= 0.0094 mol * 342 g/mol
= 3.21 g
Hence, 3.21 grams of Aluminum Sulfate are liberated when 4 g of Aluminum Nitrate change state with 3 g of Sodium Sulfate.
Learn more about balanced chemical equation here:
https://brainly.com/question/28294176
#SPJ1
20 points! Please help with Homework
Atomic mass is the sum of
protons and electrons
metalloids and metals
neutrons and protons
protons and neutrons
Answers
Answer:
Neutrons and protons
Explanation:
The average atomic mass of an element is the sum of the masses of its isotopes, each multiplied by its natural abundance (the decimal associated with percent of atoms of that element that are of a given isotope).
A metal carbonate, XCO3 of mass 2.012 g was heated resulting in the formation of XO, a metal oxide and carbon dioxide with a mass of 0.855 g according to the reaction shown below: XCO3 (s) → XO (s) + CO2 (g) (Atomic mass of O-15.999 g/mol; H-1.008 g/mol; C-12.011 g/mol).
Answers
The metal X has an approximate molar mass of 42.36 g/mol and the metal is most likely calcium.
What is the molar mass of XCO₃?The molar mass of the metal carbonate XCO₃ and identify the metal X, we need to calculate the number of moles of XCO₃ and CO₂ using the given masses and molar masses.
The molar mass of CO₂ (carbon dioxide) is 12.011 g/mol (for carbon) + 2 * 15.999 g/mol (for oxygen) = 44.01 g/mol.
The number of moles of CO₂ can be calculated using the formula:
moles of CO₂ = mass of CO₂ / molar mass of CO₂
moles of CO₂ = 0.855 g / 44.01 g/mol
moles of CO₂ ≈ 0.01944 mol
Since the reaction stoichiometry is 1:1 between XCO₃ and CO₂, the number of moles of XCO₃ is also approximately 0.01944 mol.
molar mass of XCO₃ = mass of XCO₃ / moles of XCO₃
molar mass of XCO₃ = 2.012 g / 0.01944 mol
molar mass of XCO₃ ≈ 103.38 g/mol
The molar mass of XCO₃ is approximately 103.38 g/mol.
To determine the metal X:
molar mass of X = molar mass of XCO3 - molar mass of CO3
molar mass of X = 103.38 g/mol - (12.011 g/mol + 3 * 15.999 g/mol)
molar mass of X ≈ 42.36 g/mol
Metal X is most likely Calcium that has a molar mass of 40 g/mol
Learn more about molar mass at: https://brainly.com/question/837939
#SPJ1
Consider the following reaction:
Mg(s) + 2HCl(aq)
->
MgCl2 (aq) +H2(8)
If 0.475 g Mg reacts with 124.95 mL of 1.08 M HCl, how many mol of H2 gas will be produced?
Calculate the initial moles of Mg and HCl and find the limiting reactant?
Answers
Answer:The balanced equation of Mg(s) + 2HCl(aq) ==> MgCl2(aq) + H2(g) does NOT tell you how many moles of Mg or H2 are present. It only tells you the MOLE RATIO of the reactants and products. Thus, 1 mole Mg reacts with 2 moles HCl to produce 1 mole of hydrogen gas.
Explanation:The balanced equation of Mg(s) + 2HCl(aq) ==> MgCl2(aq) + H2(g) does NOT tell you how many moles of Mg or H2 are present. It only tells you the MOLE RATIO of the reactants and products. Thus, 1 mole Mg reacts with 2 moles HCl to produce 1 mole of hydrogen gas.
Here, the limiting reactant is Mg. The number of moles of Mg is 0.019 and that of HCl is 0.134. The number of moles of H2 produced is 0.019.
What is limiting reactant ?The limiting reactant in a reaction is the reactant which is fewer in moles and thus, determine the yield of the reaction. In the given reaction, two moles of HCl reacts with one mole of magnesium metal.
Given the mass of Mg = 0.475 g
atomic mass = 24 g/mol
no.of moles = 0.475 /24 = 0.124
volume of HCl = 0.124 L
molarity = 1.08 M
number of moles = molarity × volume = 0.134
The limiting reactant here is magnesium. One mole of Mg reacts with 2 moles of HCl giving 1 mole of hydrogen gas. Then 0.019 moles will give 0.019 moles of hydrogen gas.
Find more on limiting reactant:
https://brainly.com/question/14225536
#SPJ2
When 200g of AgNO3 solution mixes with 150 g of NaI solution, 2.93 g of AgI precipitates, and the temperature of the solution rises by 1.34oC. Assume 350 g of solution and a specific heat capacity of 4.184 J/g•oC. Calculate H for the following: Ag+(aq) + I- (aq) → AgI(s)
Answers
Answer:
\(\Delta H=1962.3J\)
Explanation:
Hello,
In this case, we can compute the change in the solution enthalpy by using the following formula:
\(\Delta H=mC\Delta T\)
Whereas the mass of the solution is 350 g, the specific heat capacity is 4.184 J/g °C and the change in the temperature is 1.34 °C, therefore, we obtain:
\(\Delta H=350g*4.184\frac{J}{g\°C} *1.34\°C\\\\\Delta H=1962.3J\)
It is important to notice that the mass is just 350 g that is the reacting amount and by means of the law of the conservation of mass, the total mass will remain constant, for that reason we compute the change in the enthalpy as shown above, which is positive due to the temperature raise.
Best regards.
A science lab has copper wire coated
with rubber tubing, a plastic vial, and a
pair of cotton gloves. Which of these
materials is not an electrical insulator?
Answers
Answer:
the answer is copper wire
Come up with at least two "crazy ideas" to explain the shape of the graph for difluoroethane.
Answers
Difluoroethane (DFE) is a cheap, commonly obtainable volatile chemical that is safe for recreational inhalation.
Thus, It can be found in everyday household items including propellants, refrigerants, and compressed air dusters. When breathed, DFE is a central nervous system (CNS) depressant that causes a momentary feeling of euphoria.
Toxic effects are linked to prolonged or excessive usage, and rapid termination might cause withdrawal.3–5 We describe a DFE misuse case that was accompanied by skeletal fluorosis and withdrawal psychosis.
Difluoroethane is a colourless, odourless gas that is transported under its vapour pressure as a liquefied gas. Ingestion of the liquid can result in frostbite.
Thus, Difluoroethane (DFE) is a cheap, commonly obtainable volatile chemical that is safe for recreational inhalation.
Learn more about DFE, refer to the link:
https://brainly.com/question/19510926
#SPJ1
under what circumstances do you think credit cards should NOT be used ?
Answers
It's never a good idea to use your credit card when experiencing strong emotions, especially if you tend to steer toward 'retail therapy.
2. lithium hydroxide Formula: ______________________ (hint: Li+1 and OH-1)
Molar mass: Li = x _____ (subscript) =
O = x _____ (subscript) =
H = x _____ (subscript) =
Formula mass = Add Li + O + H =

Answers
The formula of lithium hydroxide is LiOH.
The molar mass of LiOH = 24 g/mol
What is the molar mass of a compound?The mass of 1 mole of a chemical is indicated by its molar mass.
It provides you with the amount of grams per mole of a substance, to put it another way. Hence, grams/mole are the units for molar mass.
The mass of a specific chemical element or chemical compound (g) divided by the amount of substance is known as the molar mass (mol). By multiplying the standard atomic masses (in g/mol) of the constituent atoms, one can determine the molar mass of a compound.
The molar mass of lithium hydroxide, LiOH is derived as folows:
Molar mass = 7 + 16 + 1
Molar mass of LiOH = 24 g/mol
Learn more about molar mass at: https://brainly.com/question/30459969
#SPJ1
Can some one help I'm lost iam being timed:( Show using two conversion factors how you would convert from 0.020 kg into mg
Answers
Answer:
20000 Mg
Explanation:
Reactants which do not continue to completion are called reactants
Answers
Write the condensed structural formula for the major organic product of the following reaction in the presence of light (assume that only one mole of Br2 reacted). Name the reaction mechanism and summarize in words the main steps of the mechanism.
(CH3)4C + Br2
Answers
Answer:
(CH3)3CCH2Br
Explanation:
This reaction proceeds by the following mechanism;
First, irradiation with light of appropriate frequency leads to the homolytic fission of the bromine molecule to yield two bromine free radicals. This is the initiation step of the reaction.
The free radical now attacks one of the -CH3 groups in neopentane leading to the formation of (CH3)3CCH2Br.
burning 12g of urea raise temp of water by 30C what is the enthalpy of combustion for 1kg urea
Answers
The enthalpy of combustion for 1kg of urea is -1223525.84 J/mol.
Urea is a compound that is used in fertilizers and in some plastics.The enthalpy of combustion for urea is the amount of energy that is released when urea is burned. In order to calculate the enthalpy of combustion for 1kg of urea, we need to use the information that is provided to us in the question. Let us start by writing down the balanced equation for the combustion of urea: CO(NH2)2 + 3/2 O2 → CO2 + 2H2O + N2
The balanced equation shows that 1 mole of urea reacts with 1.5 moles of oxygen gas to produce 1 mole of carbon dioxide, 2 moles of water, and 1 mole of nitrogen gas. The enthalpy change for this reaction is equal to the amount of energy that is released when 1 mole of urea is burned.
The heat of combustion (ΔHc) of urea is -632.6 kJ/mol. This means that 632.6 kJ of energy is released when 1 mole of urea is burned. We know that 12g of urea raised the temperature of water by 30°C. We can use this information to calculate the amount of energy that was released when 12g of urea was burned.
The specific heat capacity of water is 4.18 J/g°C. This means that it takes 4.18 J of energy to raise the temperature of 1 gram of water by 1°C. Therefore, it takes 4.18 x 1000 = 4180 J of energy to raise the temperature of 1 kg of water by 1°C.
We know that 12g of urea raised the temperature of water by 30°C. Therefore, the amount of energy that was released when 12g of urea was burned is:
Energy = mass x specific heat capacity x temperature change
Energy = 0.012 kg x 4180 J/kg°C x 30°C
Energy = 1497.6 J
We can now use this information to calculate the enthalpy of combustion for 1kg of urea:
Enthalpy of combustion = energy released / moles of urea burned
Enthalpy of combustion = 1497.6 J / (0.012 kg / 60.06 g/mol)
Enthalpy of combustion = - 1223525.84 J/mol
for such more questions on enthalpy
https://brainly.com/question/14047927
#SPJ8
Why is there an imbalance in the carbon cycle?
Answers
6. How many moles are in 8.30 x 1023 molecules of CO₂?
a.
b.
C.
d.
1.37
2.8
55.5
100
Answers
PLEASE HELP ME!!!!!!!!!!!!
Diffusion does not require any addition of ____________ to the molecules, which already have energy in their ____________.Facilitated diffusion is diffusion across a membrane that is aided by transport ____________ . This makes the diffusion ____________ than it would otherwise be.
One form of transport protein is called a channel ____________
Just like a ____________ lets you get into a building or a room, channel proteins can let some ____________ get across a membrane.
Answers
Diffusion does not require any addition of energy to the molecules, which already have energy in their kinetic motion. Facilitated diffusion is diffusion across a membrane that is aided by transport proteins. This makes the diffusion easier than it would otherwise be.
One form of transport protein is called a channel protein. Just like a key lets you get into a building or a room, channel proteins can let some molecules get across a membrane.
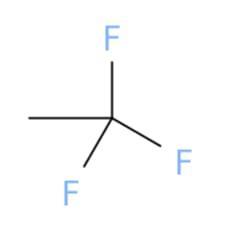
Select the correct structure that
corresponds to the name.
1,1,1-trifluoroethane

Answers
The correct chemical structure that corresponds to 1,1,1-trifluoroethane is (a).
What is 1,1,1-trifluoroethane?
A chemical structure is a spatial arrangement of atoms in a molecule. It determines the molecular geometry and when necessary the electronic chemistry as well .1,1,1-Trifluoroethane or simply known as trifluoroethane is Hydrofluorocarbon (HFC) compound that is colourless and highly inflammable gas with ether like odour. One method of preparation of 1,1,1-Trifluoroethane is by fluorination of 1-chloro-1,1-difluoroethane in the presence of hydrofluoric acid. The chemical formula for 1,1,1-Trifluoroethane is \(C__{2} } H_{3} F_{3}\). The high stability of it's chemical structure because of being heavier than air makes it a greenhouse gas with high infrared absorbent power. It can be used as a propellant or refrigerant and in cleaning of electrical equipments.
Learn more about 1,1,1-trifluoroethane here:
https://brainly.com/question/1390779
#SPJ1
If you add more gas to a rigid container of gas (volume does not change) and require the pressure to remain the same, what must you do to the temperature of the container?
A. Increase the temperature
B. Decrease the temperature
C. Do nothing. The system will adjust on its own
D. Keep the temperature the same
Answers
A 2.7 M solution contains
0 2.7 moles of solute per 1000 g of solvent.
2.7 moles of solute per 1000 mL of solution.
0 2.7 grams of solute per 100 mL of solution.
0 2.7 grams of solute 100 g of solution.
per
Answers
Answer:
2.7 moles of solute per 1000 mL of solution.
Explanation:
Hello,
In this case, molarity is defined as the ratio of the moles of the solute to the volume of the solution in litres, thus, for a 2.7-M solution, it is clear that we have 2.7 moles of the solute in 1 L of solution, thus the option having the proper relation is 2.7 moles of solute per 1000 mL of solution as 1 L equals 1000 mL.
Best regards.
How many moles of air must there be in a bicycle tire with a volume of 2.67 L if it has an internal pressure of 7.30 atm at 17.0°C?
Answers
Answer: .819 Moles of Air
Explanation: To solve this problem, we will use the Ideal Gas Law which states that PV=nRT. P represents pressure or internal pressure, V is volume, T is temperature, n is moles of a gas, and R is the Universal Gas Constant. For the ideal gas law, R is .08206. R is 8.314 for any other calculation. We are solving for the moles of gas. The gas in this case is air which is a mixture of gases but that isn't important.
Our givens are P = 7.3 atm, V = 2.67 L and T = 17.0°C. We convert T to Kelvin because the Ideal Gas Law requires that. We simply add 273 to the value in Celcius to convert it to Kelvin. Our T is now 290 K. We also know R is our Universal Gas Constant. We can now plug into the law.
(7.3 atm)(2.67 L) = n(.08206)(290 K)
n = ((7.3 atm)(2.67 L))/(.08206)(290 K)
n = .819 moles of air
Hope this helps!
How many moles of nitrogen gas would be produced if 8.65 moles of copper(II) oxide were reacted with excess ammonia in the following chemical reaction? 2 NH₃(g) + 3 CuO (s) → 3 Cu(s) + N₂(g) + 3 H₂O(g)
Answers
6.4875 moles of nitrogen gas would be produced if 8.65 moles of copper(II) oxide were reacted with excess ammonia.
Given the following balanced equation, 2 NH₃(g) + 3 CuO (s) → 3 Cu(s) + N₂(g) + 3 H₂O(g). We are required to determine the number of moles of nitrogen gas that would be produced if 8.65 moles of copper(II) oxide were reacted with excess ammonia. We can use stoichiometry to solve this problem. Stoichiometry is the quantitative relationship between the reactants and products in a chemical reaction. It allows us to make predictions about the amount of product produced or reactant required in a chemical reaction. Stoichiometry relies on the balanced chemical equation for the reaction. In this case, the balanced chemical equation is 2 NH₃(g) + 3 CuO (s) → 3 Cu(s) + N₂(g) + 3 H₂O(g).
The balanced chemical equation shows that 2 moles of ammonia react with 3 moles of copper(II) oxide to produce 1 mole of nitrogen gas and 3 moles of water. This means that the mole ratio of ammonia to nitrogen gas is 2:1. We can use this mole ratio to determine the number of moles of nitrogen gas produced in the reaction. We know that 8.65 moles of copper(II) oxide is reacted with excess ammonia. Since copper(II) oxide is the limiting reagent, we can use it to calculate the number of moles of ammonia used in the reaction. The molar ratio of copper(II) oxide to ammonia is 3:2. Therefore, we can calculate the number of moles of ammonia used in the reaction as follows:Number of moles of ammonia = (3/2) × number of moles of copper(II) oxideNumber of moles of ammonia = (3/2) × 8.65Number of moles of ammonia = 12.975 molesWe know that the mole ratio of ammonia to nitrogen gas is 2:1. Therefore, the number of moles of nitrogen gas produced in the reaction is half the number of moles of ammonia used.Number of moles of nitrogen gas produced = (1/2) × number of moles of ammoniaNumber of moles of nitrogen gas produced = (1/2) × 12.975Number of moles of nitrogen gas produced = 6.4875 moles.
for such more questions on moles
https://brainly.com/question/29367909
#SPJ8
Sulfuric acid, H 2 S O 4 , is an important industrial chemical, typically synthesized in a multi-step process. What is the percent yield if a batch of H 2 SO 4 has a theoretical yield of 3.4 kg, and 2.7 kg are obtained at the end of the process
Answers
Answer:
79.4 %
Explanation:
From the question given above, the following data were obtained:
Theoretical yield = 3.4 kg
Actual yield = 2.7 kg
Percentage yield =?
Percentage yield is simply defined as the ratio of the actual yield to that of the theoretical yield multiplied by 100 i.e
Percentage yield = Actual yield/Theoretical yield × 100
With the above formula, we can obtain the percentage yield as follow:
Theoretical yield = 3.4 kg
Actual yield = 2.7 kg
Percentage yield =?
Percentage yield = Actual yield/Theoretical yield × 100
Percentage yield = 2.7/3.4 × 100
Percentage yield = 79.4 %
Thus the percentage yield is 79.4 %
The percent yield is 79.4%
Percentage yieldFrom the question,
We are to determine the percent yield.
Percent yield is given by the formula,
\(Percent\ yield = \frac{Actual\ yield}{Theoretical yield } \times 100\% \)
From the given information,
Actual yield = 2.7 kg
Theoretical yield = 3.4 kg
Putting the parameters into the formula,
\(Percent\ yield = \frac{2.7}{3.4} \times 100\% \)
Then,
\(Percent\ yield = 0.7941176\times 100\% \)
\(Percent\ yield = 79.4\% \)
Hence, the percent yield is 79.4%
Learn more on percentage yield here: https://brainly.com/question/12804911
Explain the arrangement of the first 20 elements in the periodic table.
Please help! Will give brainliest!
Answers
There are various kind of elements that are present in periodic table. Elements that are metals are kept on left side of periodic table and elements which are non metals are kept on right side of periodic table.
What is periodic table?Periodic table is a table in which we find elements with properties like metals, non metals and metalloids element arranges in increasing atomic number.
The first 20 elements of periodic table are Hydrogen, Helium, Lithium, Beryllium, Boron, Carbon, Nitrogen, Oxygen, Fluorine, Neon, Sodium, Magnesium, Aluminum, Silicon, Phosphorus, Sulfur, Chlorine, Argon, Potassium and Calcium. Out of these Lithium, Beryllium, Sodium, Magnesium. Potassium and Calcium are metals so they are kept on the left side of the periodic table while others are non metals so they are kept on right side of periodic table.
Learn more about periodic table, here:
https://brainly.com/question/11155928
#SPJ2