Answers
Answer:
juvn hgf jb ujvi i junk food sux
Explanation:
Related Questions
WIL GIVE AS MANY POINTS POSSIBLE!!!!!! A. A U.S. state needs to increase its available electricity. Almost half of the state
is covered in desert, and there are strong winds most of the year. There are no
large or fast-moving rivers, and very few crops are grown.
i. Name two renewable energy sources, and state and explain whether the
state should use them based on the description above. (6 points)
1
ii. If the state wants to minimize environmental damage, what energy sources
should it consider using? Explain your position. (2 points)
Answers
it takes specific amount of energy to remove exactly one electron from an atom.
What is electron?
Electron is a stable sub atomic particle with a charge of negative electricity found in all atoms and acting as the primary Carrier of electricity in solid.
1-This is shown by the equation where the orbit of the electron around a proton is equal to the quantum. The quantities energy level, times a wavelength.
2-So this is this equation and then abruptly hypothesis links quantities. Energy states with a standing wave function so as that the energy of the election on increases its orbit must also increase.
3- And as considering the wavelength, we're assuming station is constant. Two pies are the only constants, and they aren't changing. It does.
4-As the energy or quantifies energy level of an electron increases its radius around the nucleus must increase.
5- Also, if its energy level decreases, its radius must also decrease. So this hypothesis ties together quantities, energy levels and the relationship and its relationship with the size of the circumference around it. A nucleus which is also the electron.
To know more about electron click-
https://brainly.com/question/25674345
#SPJ1
How many bromine atoms are present in 30.1 g of CH2Br2
Answers
Answer:
There are approximately 1.43 x 10^23 bromine atoms in 30.1 g of CH2Br2.
Explanation:
Calculate the molar mass of CH2Br2:
M(CH2Br2) = M(C) + 2M(H) + 2M(Br)
M(CH2Br2) = 12.01 g/mol + 2(1.01 g/mol) + 2(79.90 g/mol)
M(CH2Br2) = 252.82 g/mol
Calculate the number of moles of CH2Br2:
n = m/M
n = 30.1 g / 252.82 g/mol
n = 0.119 mol
Determine the number of bromine atoms:
There are 2 bromine atoms in each molecule of CH2Br2, so we can use Avogadro's number to convert moles to atoms:
N = n * N_A * 2
N = 0.119 mol * 6.022 x 10^23 atoms/mol * 2
N = 1.43 x 10^23 bromine atoms
which of the following will change the pressure in a reaction involving only gases at equilibrium? select all that apply. multiple select question.
Answers
A reactant or product being added to or removed Variations in the container's volume Addition of an inert gas
How would you define "gasses" in this situation?Dangerous gas emissions are also coming from the planet's surface. Human activity has caused the atmosphere's quantities of carbon dioxide, methane, tropospheric ozone, and nitrous oxide to rise dramatically above pre-industrial levels.
In the singular, what does the word "gas" mean?The plural version of the word "gas" is "gase," and it refers to a substance that expands to fill any container. Many distinct meanings can be assigned to third-person present tense verbs, such as "gasses."
To know more about Gases visit:
brainly.com/question/1369730
#SPJ4
Which molecule is butane?
H H H H
A. H-C-C-C-C-H
||||
H H H H
B.
C.
H3C
C=C
H
CH3
H
H
|
D. H-C=C-C-C-H
H H
H
|
Answers
Answer: A
Explanation:
The -ane suffix implies that the compound has only single bonds for carbon-carbon bonds. The but- prefix implies that the compound consists of four carbons. Since 4 bonds are required for each carbon, there will be a total of 10 hydrogen atoms: 3 on each carbon at the end of the chain and 2 for each carbon in the middle of the chain. Thus, butane is A.
which is the graph of the function g(x) = f(-x)
Answers
To graph the function g(x) = f(-x), you can start with the graph of f(x) and then reflect it about the y-axis.
What is a graph of the function g(x) = f(-x)?To find the graph of the function g(x) = f(-x), we can start with the graph of the function f(x) and then reflect it about the y-axis.
If the graph of f(x) is symmetric with respect to the y-axis, meaning it is unchanged when reflected, then g(x) = f(-x) will have the same graph as f(x).
However, if the graph of f(x) is not symmetric with respect to the y-axis, then g(x) = f(-x) will be a reflection of f(x) about the y-axis.
In either case, the resulting graph of g(x) = f(-x) will be symmetric with respect to the y-axis.
Learn more about the graph of functions at: https://brainly.com/question/17089414
#SPJ1

What happens to the amount of solution when we add food colour to it?
Answers
Answer:
We need more? What else is in the question? This is unanswerable.
Explanation:
Find the balance equation
_H2O+_O2=_H2O2
Answers
The balanced reaction equation of the reaction is; 2H2O + O2 → 2H2O2
How do we balance reaction equation?This is a chemical equation that represents the reaction between water (H2O) and oxygen (O2) to form hydrogen peroxide (H2O2). The coefficients in front of each substance indicate the relative number of molecules involved in the reaction.
Recall that we can balance the reaction equation by ensuring that the atoms of the elements on both sides of the reaction equation are the same as we have above.
Learn more about reaction equation:https://brainly.com/question/3588292
#SPJ1
determine the number of atoms of carbon in 15.0 grams of glucose
Answers
Answer:
Calculate the volume (in mL) of the 1.356 M stock NaOH solution needed to prepare 250.0 mL ... Glucose (molar mass=180.16 g/mol) is a simple, soluble sugar ... g of glucose in enough water to make 500.0 mL of solution. • Step 2: Transfer 18.6 mL of the glucose
Explanation:
What are the magnetic quantum numbers possible for a 6s subshell and for a 4f subshell? Drag the correct numbers into each of the columns.
Put responses in the correct input to answer the question. Select a response, navigate to the desired input and insert the response. Responses can be selected and inserted using the space bar, enter key, left mouse button or touchpad. Responses can also be moved by dragging with a mouse.
6s Subshell 4f Subshell
Answers
For the 6s subshell, the magnetic quantum number is zero while for the 4f subshell we have -3, -2, -1, 0, 1, 2, 3.
What is the magnetic quantum number?The magnetic quantum number takes on values between 0 to (n - l). It gives the orientations of the sublevels. The four types of quantum numbers are;
Principal quantum numberAzimuthal quantum numberMagnetic quantum numberSpin quantum numberFor the 6s subshell, the magnetic quantum number is zero while for the 4f subshell we have -3, -2, -1, 0, 1, 2, 3.
Learn more about magnetic quantum number:https://brainly.com/question/24204727
#SPJ1
A 54.2 g sample of polystyrene, which has a specific heat capacity of 1.880 J-gc, is put into a calorimeter (see sketch at
right) that contains 100.0 g of water. The temperature of the water starts off at 21.0 °C. When the temperature of the water stops
changing it's 34.3 °C. The pressure remains constant at 1 atm.
Calculate the initial temperature of the polystyrene sample. Be sure your answer is rounded to the correct number of significant
digits.
thermometer.
insulated
container
water
sample.
a calorimeter
Answers
Tthe initial temperature of the polystyrene sample is 39.4°C.
Given: Mass of polystyrene sample = 54.2 gSpecific heat of polystyrene = 1.880 J-g°CWater mass = 100.0 g Initial water temperature = 21.0°CWater final temperature = 34.3°CPressure remains constant at 1 atmFormula used:Heat gained by water = heat lost by polystyreneHence,Heat lost by polystyrene = Heat gained by water=> mcΔT = mcΔTwhere,m = mass of polystyrene or waterc = specific heat capacityΔT = change in temperatureThe temperature change is ΔT = 34.3°C - 21.0°C = 13.3°CNow we can use this temperature change to calculate the initial temperature of the polystyrene.Taking the water's specific heat capacity, c = 4.184 J/g°CHeat gained by water = (100.0 g)(4.184 J/g°C)(13.3°C) = 5574 JHeat lost by polystyrene = 5574 JTaking the polystyrene's specific heat capacity, c = 1.880 J/g° ) = 13.3°C Now let's calculate the mass of polystyrene using the specific heat capacity formula.5574 J = (54.2 g)(1.880 J/g°C)(13.3°C - Ti)Ti = 39.4°C
for more questions on polystyrene
https://brainly.com/question/15913091
#SPJ8
What are the differences between seismic waves and surface waves?
Answers
Answer:
Seismic waves can travel through the Earth's inner layers, but surface waves can only move along the surface of the planet like ripples on water.
ammonia gas occupies a volume of 57.9 L at a pressure of 532.4atm. if the pressure were lowered to 256.8atm,what would the new volume be
Answers
Answer:
683838447745 anmmlllkkkkk
_____ Al(NO3)3 + _____ (NH4)3PO4 → _____AlPO4 + _____ NH4NO3
Answers
Answer:
1 Al(NO3)3 + 1 (NH4)3PO4 → 1 AlPO4 + 3 NH4NO3
Explanation:
To balance Al(NO3)3 + (NH4)3PO4 → AlPO4 + NH4NO3 you'll need to be sure to count all of the atoms on each side of the chemical equation. Once you know how many of each type of atom you can only change the coefficients (the numbers in front of atoms or compounds) to balance the equation.
Important tips for balancing chemical equations:
Only change the numbers in front of compounds (the coefficients). Never change the numbers after atoms (the subscripts). The number of each atom on both sides of the equation must be the same for the equation to be balanced.Select the structure that corresponds
to the molecule name:
aniline
B.
A.
-NH₂
C. both
-NH₂
Enter

Answers
Answer:
B- \(C_{6} H_{5} NH_{2}\)Explanation:
Aniline is an organic compound with the formula C6H5NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine.


Fill in the following chart: 100 points!
**If there is a + sign that means there are less electrons. If there is a sign, there are more electrons.

Answers
Answer:
Calcium atom:
20 protons, 20 electrons
Calcium 2+ ion:
It is a calcium atom but with 2 fewer electrons, so
20 protons, 18 electrons
Oxygen atom:
8 protons and 8 electrons
Oxygen 2- ion:
It is an oxygen atom but with 2 more electrons, so
8 protons, 10 electrons
Calcium oxide:
Add protons and electrons of of Ca with O to get:
20 + 8 protons = 28 protons, 20 + 8 electrons: 28 electrons.
Alternatively, add protons and electrons of Ca{2+} and O{2-} to get the same result.
The correct number of protons and electrons of the given ions are as follows:
Ca atom = 20 protons, 20 electronsCa2+ ion = 20 protons, 18 electronsO atom = 8 protons, 8 electronsO2- ion = 8 protons, 10 electronsHow to calculate protons and electrons?The number of protons and electrons in an atom or ion determines the overall charge of the ion or atom.
In a neutral atom, the number of protons and electrons are equal, hence, the overall charge of the atom is 0. However, in a charged atom (ion), the difference between the proton and electron number is the base.
It is important to note that the number of protons is the atomic number of an atom, hence, does not change. However, the number of electrons changes depending on whether electrons are gained or lost.
Learn more about protons and electrons at: https://brainly.com/question/19027296
#SPJ1
To identify a diatomic gas ( X2 ), a researcher carried out the following experiment: She weighed an empty 6.4- L bulb, then filled it with the gas at 1.30 atm and 27.0 ∘C and weighed it again. The difference in mass was 9.5 g . Identify the gas.
Answers
The diatomic gas ( X2 ) is N₂ dinitrogen.
Dinitrogen is a chemical compound fashioned from the covalent bonding of two nitrogen atoms. it's far a colorless, odorless gas at room temperature and pressure, which makes up about seventy-eight % of the Earth's environment.
Diatomic gas is a chemical compound formed from the covalent bonding of two nitrogen atoms. it's miles drab, odorless gasoline at room temperature and stress, which makes up about seventy eight % of the Earth's surroundings.
Volume = 6.4 L
Pressure = 1.3 atm
Temperature = 25 C = 298 K
R = 0.08206 L.atm/mol.K
P * V = n * R * T
1.3 atm * 6.4 L = n * 0.08206 L.atm/mol.K * 298 K
n = 0.34 moles
difference of mass is the mass of gas = 9.5 g
Molar mass = Mass / No. of moles = 9.5 g / 0.34 moles = 27.9 g/mol
diatomic gas with molar mass 28 g/mol is N2
Hence the diatomic molecule is N2
Learn more about dinitrogen here:-https://brainly.com/question/11651796
#SPJ9
Sample Response: Repetition refers to performing multiple trials throughout an experiment. Repetition reduces mistakes and increases one’s confidence in the results. Replication refers to the ability of a process to be repeated by another individual. When a scientist replicates the experiment of another, the experiment should produce the same results.
What did you include in your response? Check all that apply.
Repetition refers to performing multiple trials throughout an experiment.
Repetition reduces mistakes and increases one’s confidence in the results.
Replication refers to the ability of a process to be repeated by another individual.
When a scientist replicates the experiment of another, the experiment should produce the same results.
Answers
The difference between repetition and replication include the following:
Repetition refers to performing multiple trials throughout an experiment. Repetition reduces mistakes and increases one's confidence in the results. Replication refers to the ability of a process to be repeated by another individual. When a scientist replicates the experiment of another, the experiment should produce the same results.What is Replication?This involves the process of copying or duplicating something from a source material and the result is usually the same.
On the other hand repetition refers to performing activities multiple times and doesn't involve duplication which is why the results gotten may be different depending on the conditions involved.
Read more about Repetition here https://brainly.com/question/3601186
#SPJ1
Question is in picture below

Answers
The dissociation of cadmium chloride is as follows: CdCl₂(s) → Cd⁺²(aq) + 2Cl⁻(aq)
What is dissociation?Dissociation is the process by which a compound body breaks up into simpler constituents; said particularly of the action of heat on gaseous or volatile substances.
It is a chemical reaction in which a compound breaks apart into two or more components. The general formula for a dissociation reaction follows the form:
AB → A + B
According to this question, cadmium chloride undergoes dissociation into cadmium and chlorine ions as follows:
CdCl₂(s) → Cd⁺²(aq) + 2Cl⁻(aq)
Learn more about dissociation at: https://brainly.com/question/28952043
#SPJ1
Milk of magnesia, which is an aqueous suspension of magnesium hydroxide, is used as an antacid in the reaction below. How many molecules of HCl would have to be present to form 34.52 g of MgCl₂?
Mg(OH)₂(s) + 2 HCl(aq) → 2 H₂O(l) + MgCl₂(aq)
Answers
Approximately 4.37 x 10^23 molecules of HCl would be required to form 34.52 g of MgCl₂.
To determine the number of molecules of HCl required to form 34.52 g of MgCl₂, we need to use the molar mass and stoichiometry of the balanced equation:
Mg(OH)₂(s) + 2 HCl(aq) → 2 H₂O(l) + MgCl₂(aq)
The molar mass of MgCl₂ is 95.21 g/mol.
First, we need to calculate the number of moles of MgCl₂ formed:
Moles of MgCl₂ = mass of MgCl₂ / molar mass of MgCl₂
Moles of MgCl₂ = 34.52 g / 95.21 g/mol
Moles of MgCl₂ = 0.363 mol
According to the balanced equation, the stoichiometric ratio between HCl and MgCl₂ is 2:1. Therefore, the moles of HCl required can be calculated as follows:
Moles of HCl = 2 * Moles of MgCl₂
Moles of HCl = 2 * 0.363 mol
Moles of HCl = 0.726 mol
To calculate the number of molecules, we need to use Avogadro's number, which is approximately 6.022 x 10^23 molecules/mol.
Number of molecules of HCl = Moles of HCl * Avogadro's number
Number of molecules of HCl = 0.726 mol * 6.022 x 10^23 molecules/mol
Number of molecules of HCl = 4.37 x 10^23 molecules
Therefore, approximately 4.37 x 10^23 molecules of HCl would be required to form 34.52 g of MgCl₂.
For more such questions on molecules
https://brainly.com/question/1351818
#SPJ8
CAN SOMEONE HELP WITH THIS QUESTION?

Answers
1a. The theoretical percentage of water in nickel(II) sulfate heptahydrate is 44.9%
1b. The theoretical percentage of water in aluminum chloride hexahydrate is 44.7%
1a. How do I determine the percentage of water?The percentage of water in nickel(II) sulfate heptahydrate, NiSO₄.7H₂O can be obtained as follow:
Molar mass of NiSO₄.7H₂O = 280.7 g/moleMass of H₂O in NiSO₄.7H₂O = 7H₂O = 7 × 18 = 126 gPercentage of water, H₂O =?Percentage of water, H₂O = (mass of H₂O / mass of NiSO₄.7H₂O) × 100
Percentage of water, H₂O = (126 / 280.7) × 100
Percentage of water, H₂O = 44.9%
1b. How do I determine the percentage of water?The percentage of water in aluminum chloride hexahydrate, AlCl₃.6H₂O can be obtained as follow:
Molar mass of AlCl₃.6H₂O = 241.5 g/moleMass of H₂O in AlCl₃.6H₂O = 6H₂O = 6 × 18 = 108 gPercentage of water, H₂O =?Percentage of water, H₂O = (mass of H₂O / mass of AlCl₃.6H₂O) × 100
Percentage of water, H₂O = (108 / 241.5) × 100
Percentage of water, H₂O = 44.7%
Learn more about percentage composition:
https://brainly.com/question/11952337
#SPJ1
Select the correct answer.
A bicycle traveled 150 meters west from point A to point B. Then it took the same route and came back to point A. It took a total of 2 minutes for
the bicycle to return to point A. What is the average speed and average velocity of the bicycle?
ОА.
The average speed is 2.5 meters/second, and the average velocity is
2.5 meters/second east.
OB
The average speed is O meters/second, and the average velocity is
2.5 meters/second east.
Ос.
The average speed and average velocity are both O meters/second.
OD. The average speed is 2.5 meters/second, and the average velocity is
O meters/second.
Answers
How has the increase in human population impacted earths resources?
Answers
Answer:
The increase in human population has impacted Earth's resources in several major ways:
• Increased demand for food, water, and shelter. A larger population requires massive increases in food, water, and living spaces which strains natural resources and infrastructure. Producing enough food alone is a significant challenge.
• Accelerated consumption of resources. As population grows, the use of resources like forests, minerals, fossil fuels also increases rapidly to meet demands. This accelerated depletion of resources threatens long term sustainability.
• Increased pollution. A bigger population produces more pollution, waste, emissions, and environmental degradation as a byproduct of energy usage, transportation, industrialization, and land/resource use. This pollution harms ecosystems and contaminates the air, water and land.
• Biodiversity loss. As natural habitats are destroyed or fragmented to enable more human use, many plant and animal species lose their homes and face a higher risk of extinction. Tropical rainforests, in particular, have been heavily impacted.
• Inequality. While resources are limited, population growth often exacerbates inequality in access to and distribution of resources. Poor or developing regions typically have the highest populations but fewest resources per capita.
• Migration and conflict. Shortages of resources in certain areas or regions may lead to migration, economic troubles, social unrest, and in some cases even resource conflicts or wars.
• Slower development. Extremely rapid population growth rates make it difficult for governments, organizations and societies to effectively manage development, improve standards of living, advance technology, and make other progress at an optimal pace. Slower, more stabilized population growth may enable a higher overall quality of life.
So in many profound and troubling ways, increased population size has created immense pressures on Earth's resources and made it more difficult to meet present and future needs in a sustainable manner. Most experts agree that slowing population growth is critical to ensuring resource security for future generations.
Explanation:
You have three crystal substances (x, y, and z) whose properties are listed in the table below. Identify the types of bonds in each substance and explain your answer.
Answers
X contains metallic bond
Y contains ionic bond
Z contains covalent bond
What is the crystal structure?We know that the crystal structure of a compound would have to do with the way that the atoms and the elements that compose the compound could be said to have been arranged. Thus if a compound is said to have a crystal structure then we can say that the compound would be crystalline in nature as we can see from the table that we have in the question here.
The kind of bonds that we have in the compound is about one of things that can be able to determine whether or not the compound has a crystalline structure as we can see.
Looking at the properties of the compounds that have been shown in the bale that has been attached to the answer that we have here, we can be able to make a decision regarding each of the substances shown.
Learn more about crystal substances:https://brainly.com/question/28728101
#SPJ1

U Activity 2. Lights On, Lights Off Write On if the process pertains to light-dependent reaction and writes OFF if the process pertains to the light-independent reaction. Write your answer on a separate sheet of paper.
1. It is also known as the dark reaction of photosynthesis.
2. Primary acceptor of carbon is Photosystem I and II.
3. Site of the process is in the stroma. 4. Photolysis of water does not occur.
5. Process type is both cyclic and non-cyclic processes.
6. It is a release of oxygen that gives off aldehydes and hydrogen upon dehydrogenation
7. It is a process that converts solar energy into chemical energy.
8. It is a light dependent process.
9. Process type is cyclic only,
10. Primary acceptor of carbon is Rubiscobisphosphate.
Answers
1. It is also known as the dark reaction of photosynthesis: Off.
A dark reaction of photosynthesis takes place outside of the thylakoids and do not require light to proceed, so it is a light-independent reaction.2. Primary acceptor of carbon is Photosystem I and II: On.
Photosystem I (P700) and Photosystem II (P680) are large membrane protein complexes that accepts carbon during a light-dependent reaction.3. Site of the process is in the stroma: Off.
The stroma is the site for series of biochemical redox reactions called Calvin cycle, which is a light-independent reaction.4. Photolysis of water does not occur: Off.
Since the photolysis of water doesn't occur, the reaction is a light-independent reaction.5. Process type is both cyclic and non-cyclic processes: On.
A non-cyclic processes forms ATP, so it is a light-dependent reaction.6. It is a release of oxygen that gives off aldehydes and hydrogen upon dehydrogenation: On.
A light-dependent reaction causes a release of oxygen that gives off aldehydes.7. It is a process that converts solar energy into chemical energy: On.
The conversion of solar energy into chemical energy is typically a light-dependent reaction.8. It is a light dependent process: On.
A light-dependent reaction takes place in the presence of light.9. Process type is cyclic only: Off.
A cyclic process is a light-independent reaction because it doesn't require light.10. Primary acceptor of carbon is Rubiscobisphosphate: Off.
When Rubiscobisphosphate is the primary acceptor of carbon, the reaction is a light-independent reaction.Read more: https://brainly.com/question/12827458
Nitrogen and hydrogen combine at a high temperature, in the presence of a catalyst, to produce ammonia.
N2(g)+3H2(g)⟶2NH3(g)
There are four molecules of nitrogen and nine molecules of hydrogen present in the diagram.
When the reaction is complete, how many molecules of NH3 are produced?
What is the limiting reactant?
How many molecules of each reactant are remain after the reaction is complete?
Answers
After the reaction is complete, no nitrogen and no hydrogen molecules remain, and 8.00 x 1014 molecules of NH3 are produced.
In the equation, nitrogen and hydrogen react at a high temperature, in the presence of a catalyst, to produce ammonia, according to the balanced chemical equation:N2(g)+3H2(g)⟶2NH3(g)The coefficients of each molecule suggest that one molecule of nitrogen reacts with three molecules of hydrogen to create two molecules of ammonia.
So, to determine how many molecules of ammonia are produced when four nitrogen and nine hydrogen molecules are present, we must first determine which of the two reactants is the limiting reactant.
To find the limiting reactant, the number of moles of each reactant present in the equation must be determined.
Calculations:
Nitrogen (N2) molecules = 4Hence, the number of moles of N2 = 4/6.02 x 1023 mol-1 = 6.64 x 10-24 mol
Hydrogen (H2) molecules = 9Hence, the number of moles of H2 = 9/6.02 x 1023 mol-1 = 1.50 x 10-23 mol
Now we have to calculate the number of moles of NH3 produced when the number of moles of nitrogen and hydrogen are known, i.e., mole ratio of N2 and H2 is 1:3.
The mole ratio of N2 to NH3 is 1:2; thus, for every 1 mole of N2 consumed, 2 moles of NH3 are produced.
The mole ratio of H2 to NH3 is 3:2; thus, for every 3 moles of H2 consumed, 2 moles of NH3 are produced.
From these mole ratios, it can be observed that the limiting reactant is nitrogen.
Calculation for NH3 production:
Nitrogen (N2) moles = 6.64 x 10-24 moles
The mole ratio of N2 to NH3 is 1:2; therefore, moles of NH3 produced is 2 × 6.64 × 10−24 = 1.33 × 10−23 moles.
Now, to determine how many molecules of NH3 are produced, we need to convert moles to molecules.
1 mole = 6.02 x 1023 molecules
Thus, 1.33 x 10-23 moles of NH3 = 8.00 x 1014 molecules of NH3 produced.
To find the amount of each reactant remaining after the reaction is complete, we must first determine how many moles of nitrogen are consumed, then how many moles of hydrogen are consumed, and then subtract these from the initial number of moles of each reactant.
The moles of nitrogen consumed = 4 moles × 1 mole/1 mole N2 × 2 mole NH3/1 mole N2 = 8 moles NH3
The moles of hydrogen consumed = 9 moles × 2 mole NH3/3 mole H2 × 2 mole NH3/1 mole N2 = 4 moles NH3
Thus, the moles of nitrogen remaining = 6.64 × 10−24 mol – 8 × 2/3 × 6.02 × 10^23 mol-1 = 5.06 × 10−24 mol
The moles of hydrogen remaining = 1.50 × 10−23 mol – 4 × 2/3 × 6.02 × 10^23 mol-1 = 8.77 × 10−24 mol
Finally, the number of molecules of each reactant remaining can be calculated as follows:
Number of N2 molecules remaining = 5.06 × 10−24 mol × 6.02 × 10^23 molecules/mol = 3.05 × 10−1 molecules ≈ 0 molecules
Number of H2 molecules remaining = 8.77 × 10−24 mol × 6.02 × 10^23 molecules/mol = 5.28 × 10−1 molecules ≈ 0 molecules.
For more such questions on molecules
https://brainly.com/question/24191825
#SPJ8
someone help me please i need to finish this
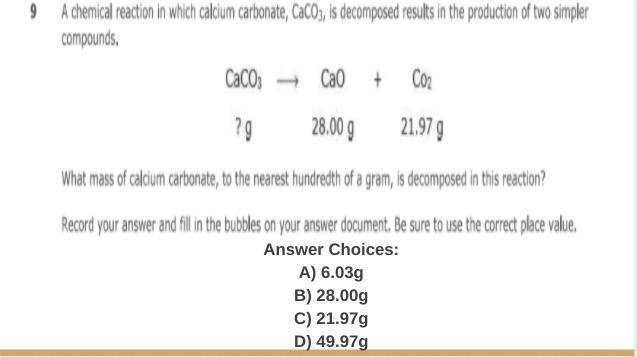
Answers
the answer is D hope it helps :)
Which action demonstrates a CHEMICAL CHANGE?
A- Long hair is cut and dried.
B- A wooden pencil is sharpened and breaks
C- An ice cube melts and becomes a clear liquid.
D- An iron nail becomes orange and flaky on the surface.
Answers
Genetic diversity is the variation in the genes of an entire species. Each circle represents a population of a particular species in different habitats. Select the population with maximum genetic diversity.
Answers
The second circle has the greatest number of species diversity .
What is genetic diversity?The term genetic diversity has to do with the existence of different species in a habitat. We know that due to natural selection, only the organisms that have traits that enable them to survive longer and reproduce are better adapted in the ecosystem. Thus, other species may begin to dwindle and even disappear in the ecosystem.
Genetic diversity ensures that there are different species in the habitat. In each circle, we see that the colors represent the different species of the organisms in the habitat. The habitat with the most number of different colors is the habitat that has the greatest magnitude of species diversity.
Hence, the second circle has the greatest number of species diversity as indicated in the image attached to this answer.
Learn more about species diversity:https://brainly.com/question/13258090
#SPJ1

If a material is ductile, it is mostly likely a
nonmetal
metal
metalloid
Gas
Answers
Answer:
Explanation:
Which of the following is most likely to be ductile?
a. Metal
b. Nonmetal
c. Metalloid
d. Gas
Answer: a. MetalMetal
What is the mass of 1.78 moles of O2
Answers
Answer:
56.96 grams
Explanation:
To find the mass of 1.78 moles of O2, we need to use the molar mass of O2, which is the mass of one mole of O2.
The chemical formula for O2 is O-O or simply O2. The molar mass of O2 is the sum of the atomic masses of two oxygen atoms, which can be found on the periodic table.
The atomic mass of oxygen (O) is approximately 16.00 g/mol. So the molar mass of O2 is:
Molar mass of O2 = 2 x atomic mass of O
= 2 x 16.00 g/mol
= 32.00 g/mol
Therefore, the mass of 1.78 moles of O2 is:
Mass = number of moles × molar mass
= 1.78 mol × 32.00 g/mol
= 56.96 g
So the mass of 1.78 moles of O2 is 56.96 grams.