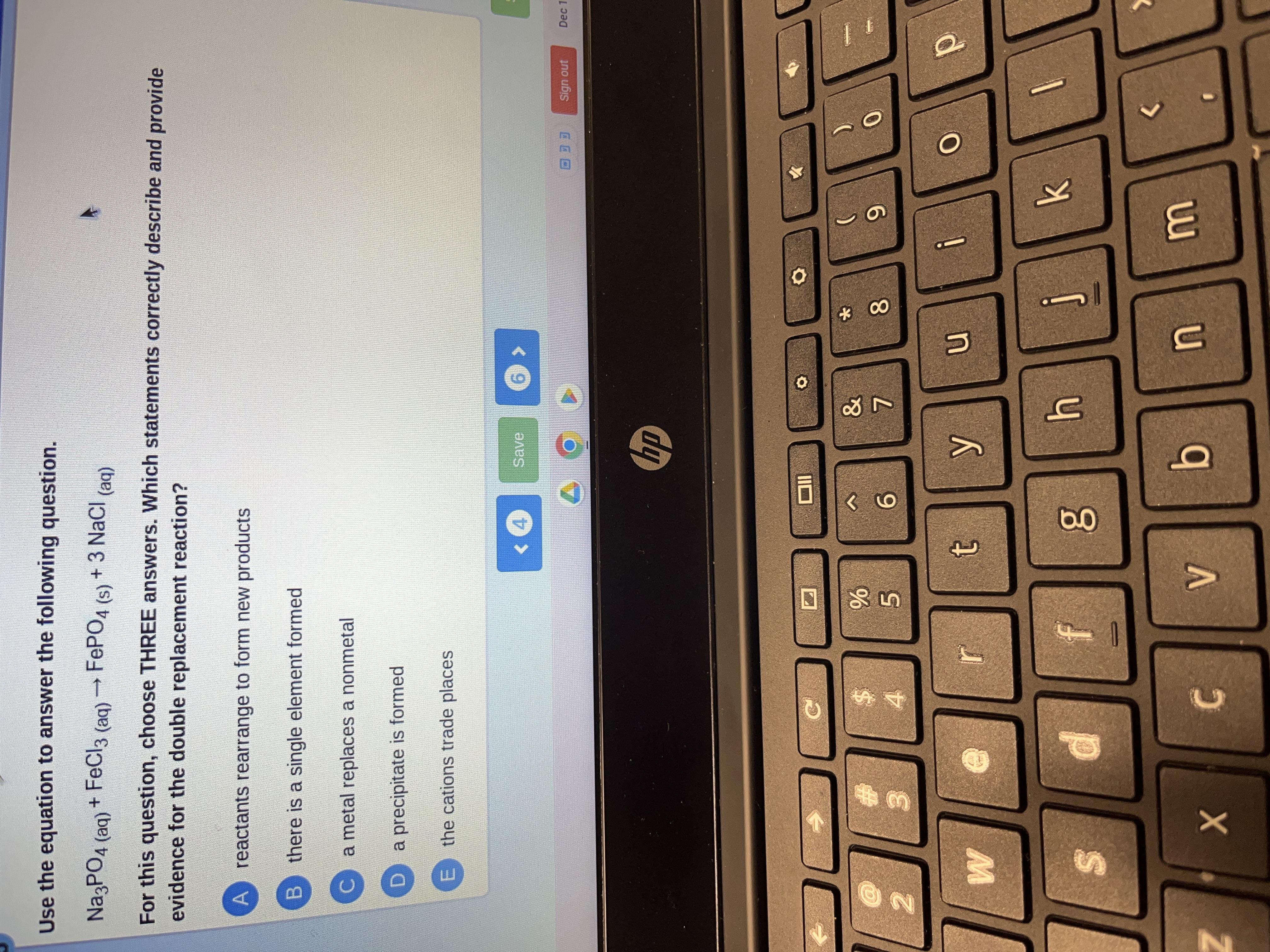
Answers
The statements which correctly describe and provide evidence for the double replacement reaction are:
reactants rearrange to form new productsmetal replaces a nonmetalthe cations trade placesThe correct options are A, D, and E.
What are double replacement reactions?Double replacement reactions are reactions where there is an exchange of radicals between two compounds when their solution is mixed together.
In double replacement reactions, a precipitate is always formed.
Also, the metallic ions trade places such that the cation of one compound is exchanged with the cation of the other compound.
A typical double replacement reaction is illustrated below:
AB + CD ---> AD + CB
where;
A and C are cations
B and D are anions.
Learn more about double replacement reactions at: https://brainly.com/question/23918356
#SPJ1
Complete question:
Which statements correctly describe and provide evidence for the double replacement reaction?
reactants rearrange to form new productsthere is a single element formeda metal replaces a nonmetala precipitate is formedthe cations trade placesRelated Questions
When cleaning a buret
Answers
Answer:
1. Rinse with distilled water.
2. Avoid dropping it
3. Use a stopper and fill with water and shake
4. Dry
The list of materials you can use for the doghouse roof is below. Can you sort them based on their physical properties? Drag and drop the following building material from highest to lowest based on their heat capacity. Use your prior knowledge to think through the tasks.
Answers
Answer:
Hi, there!
The list of materials you can use for the doghouse roof is below. Can you sort them based on their physical properties?
The Answer Is Oak, Clay, Concrete, Slate UwU
xXxAnimexXx
Have a great day!
Answer:
The Answer Is Oak, Clay, Concrete, Slate
Hope this Helps you!
PLSSSSS I NEED HELP MY AMPLIFY SIM WON’T WORK I DONT KNOW WHAT TO DO FOR AMPLIFY TAB 3.5.!! IM IN THE PURPLE GROUP !! The lead chemist wants you to determine what is happening to the freedom of movement of an object’s molecules when you smell something. Is it possible to smell a chocolate bar when it is a solid? Launch the Sim and investigate.
Use the Sim to determine if the molecules of a substance can be in two different phases at the same time.
Go through each substance and see if you can get it to exist in two phases at once.
Record as much evidence as you can in the table below.
Answers
When a chocolate bar is solid, it is able to smell it. It is also important to note that a substance 's molecules cannot be in two distinct phases at the a time.
Why is this the case ?In general, it is not possible for a the molecules of the chocolate to be in two distinct phases at the same time.
It must be noted that smelling a solid, such as a chocolate bar, however, mean the release of loose molecules from the solid, which may then move through the air and reach our olfactory receptors in our nose, allowing us to sense the fragrance.
Learn more about phase transition:
https://brainly.com/question/29795670
#SPJ1
If the plant population decreased, the amount of carbon in the atmosphere would _______.
Increase
Stay the same
Decrease
Answers
Answer:
increase answer
Explanation:
i hope that is right
What doesn’t change the resistance of a wire
Answers
The factor that doesn’t change the resistance of a wire is pressure. option A.
What is resistance of a wire?Resistance is a conductor's capacity to thwart the passage of current. It is controlled by the interplay of the applied voltage and the electric current passing through it. The amount of opposition any object applies to the flow of electric current is referred to as resistance.
The ohm, a unit of measurement for resistance, is represented by the Greek letter omega. According to Ohm's law, the voltage across two places is precisely proportional to the current flowing through a conductor between them.
Hence option A is correct.
Learn more about resistance at
https://brainly.com/question/17563681
#SPJ1
missing part;
The pressure
The length of the resistor.
The thickness of the resistor.
The temperature of the conductor.
What is the boiling point in °C of a 0.32 molal aqueous solution of NaCl?
BP (water) = 100.00 °C Kb (Water) = 0.512 °C/m
Answers
Answer:
the boiling point of solution at 3 decimal point is 100.329०C Ans.
Explanation:
given data -
molality of Nacl = 0.321 m
molal boiling point elevation constant (Kb) =0.512०C/m
# formula of change of boiling point of sample =
∆ Tb =i × Kb × m
Kb = molal boiling point of elevation constant
m = molality
i = vont's hoff factor.
Nacl is strong electrolyte and its 100% dissociate so the value of i for Nacl is 2
put value in the formula
∆ Tb = 2 × 0.512 ०C/m × 0.321m
= 0.3287
= 0.329०C
∆Tb = T'b - Tb
T'b = boiling point of solution
Tb= boiling point of solvent( water)
0.329०C = T'b - 100०c ( boiling point of water = 100०C)
T'b = 0.329०C + 100०C
= 100.329०C
hope this helps
Cellular respiration is a chemical process in cells that releases energy the cells need to function. What statement below is true about this reaction.
A. The process of cellular respiration releases energy because the energy that is released when the bonds are formed in CO2 and water is equal to the energy required to break the bonds of sugar and oxygen.
B. The process of cellular respiration releases energy because the energy that is released when the bonds that are formed in CO2 and water is lost when bonds of glucose and oxygen are broken.
C. The process of cellular respiration releases energy because the energy that is released when the bonds are formed in CO2 and water is less than the energy required to break the bonds of sugar and oxygen.
D. The process of cellular respiration releases energy because the energy that is released when the bonds are formed in CO2 and water is greater than the energy required to break the bonds of sugar and oxygen.
Answers
The process of cellular respiration releases energy because the energy that is released when the bonds are formed in CO\(_2\) and water is equal to the energy required to break the bonds of sugar and oxygen. Therefore, the correct option is option A.
Cells turn sugars into energy through a process called cellular respiration. Cells need fuel or an electron acceptor to power the chemical process that converts energy into usable forms such as ATP along with additional kinds of energy that can be utilised to power cellular reactions.
All multicellular species, including eukaryotes, as well as certain single-celled organisms, generate energy by aerobic respiration. Utilising oxygen, which is the strongest electron acceptor found in nature, is called aerobic respiration. The process of cellular respiration releases energy because the energy that is released when the bonds are formed in CO\(_2\) and water is equal to the energy required to break the bonds of sugar and oxygen.
Therefore, the correct option is option A.
To know more about Cellular respiration, here:
https://brainly.com/question/31342305
#SPJ1
**Find the speed of an object that travels 500 miles in 5 hours. S = d/t
Answers
Answer:
I believe that would be 100 mph.
Explanation:
Your distance (d) is 500 miles and your time (t) is 5 hours.
So when you replace the variables with numbers you bet 500/5 which is 100.
Therefore, you speed (S) would be 100mph.
Air is a solution and a mixture of gases, what is the solvent in the solution we call air?
a. Oxygen gas
b. Carbon dioxide gas
c. Nitrogen gas
d. Water vapor
Answers
Answer:
c. nitrogen gas
Explanation:
Our air is a homogeneous mixture of many different gases and therefore qualifies as a solution. Approximately 78% of the atmosphere is nitrogen, making it the solvent for this solution.
The carbonyl group consists of
A) a carbon-oxygen-hydrogen structure.
B) a carbon-oxygen single bond.
C) a carbon-oxygen double bond.
D) a carbon-oxygen triple bond.
Answers
A double bond connecting a carbonyl carbon and an oxygenyl carbon forms a carbonyl group.
A carbon atom is double-bonded to an oxygen atom to form the chemically organic functional group known as a carbonyl group. [C=O] Aldehydes and ketones are the most basic carbonyl groups, and they are typically joined to another carbon molecule. Many aromatic compounds have these structures, which contribute to flavor and fragrance. A functional group called the carbonyl group has a carbon atom double-bonded to an oxygen atom, or C=O. Another meaning of the word "carbonyl" is a metal-bound, neutral carbon monoxide ligand. Tetracarbonyl nickel, or Ni(CO)4, is one illustration. A double bond connecting a carbonyl carbon and an oxygenyl carbon forms a carbonyl group. Two of the carbonyl oxygen's six valence electrons are shared with the carbonyl carbon atom. Two sets of electron lone pairs make up the remaining four valence electrons.
Learn more about carbonyl here :
https://brainly.com/question/21440134
#SPJ4
If heat is added to 25.0g of copper, calcium, aluminum, iron, lead what will have the highest final temperature
Answers
Lead will have the highest temperature .
What is Specific Heat ?
Specific Heat is defined as the amount of heat required to raise the temperature of a 1 gm of substance by 1 degree Celsius.
If heat is added to 25.0g of copper, calcium, aluminum, iron, lead
The missing given specific heat is
Copper, 0.377
Calcium, 0.656
Aluminum, 0.895
Iron, 0.448
Lead, 0.129
Higher specific heat means more temperature required to bring the same amount of change in temperature
Here the temperature change is fixed , The metal with the lowest Specific heat will have the highest temperature.
From the data it can be seen that
Specific Heat of Lead, 0.129
Therefore Lead will have the highest temperature.
To know more about Specific Heat
https://brainly.com/question/11297584
#SPJ1
I need help with question 5

Answers
how many moles of oxygen are in 0.2 g of aspirin, C,H, 0,?
Answers
The number of the atoms of oxygen that we have is 3.97 * 10^21 atoms.
What is the number of moles of oxygen in aspirin?Mole is a unit of measurement used to quantify the amount of a substance. It is a fundamental concept in the field of chemistry and plays a crucial role in stoichiometry, which is the study of the quantitative relationships between substances involved in chemical reactions.
We have to note that we have 0.2 g of the aspirin and the number of moles of the aspirin would be given as;
Moles = 0.2 g/180 g/mol
= 0.0011 moles
Then we have that the number of the atoms of oxygen is;
6 * 0.0011 * 6.02 * 10^23
= 3.97 * 10^21 atoms
Learn more about moles:https://brainly.com/question/30885025
#SPJ1
The measure of the length of events and the duration of intervals between events
Answers
The measure of the length of events and the duration of intervals between events is time.
What is time?The duration of events or the gaps between them can be measured, compared, or even ordered using time. The lengthy period of time that the Earth's geologic history takes up is known as geologic time. Starting at the beginning of the Archean Eon formal geologic time runs until the present. Geology is defined as the "Science of the Earth."
Geology is the fundamental Earth science that examines how the earth created, its structure and composition, and the various forces acting on it. It is sometimes known as geoscience or earth science.
Learn more about time at;
https://brainly.com/question/479532
#SPJ1
Iron filings are attracted by a magnet. This is a physical property of iron but not of most other materials, including sand. How could you use this difference in physical properties to separate a mixture of iron filings and sand?
Answers
Answer:
Direct a magnet towards the mixture of iron and sand. You can observe that only the iron filling is attracted to the magnet and will come in contact with it. Therefore, after this process, you'll be left with iron filling on the surface of the magnet whereas the sand particles are left on the dish
To separate a mixture of iron filings and sand, using the physical property of iron is by using a magnet because a magnet attracts iron, but not sand.
What are physical properties?
Physical properties are those properties that can be seen by seeing an object or a substance. These properties do not change with the change in the composition of matter.
Physical properties are appearance, texture, color, odor, melting point, boiling point, density, solubility, polarity, and many others.
Magnetic properties are the property in which the object attracts to magnets and or iron-containing substances, here it can be used to separate sand and iron.
Thus, using a magnet and the magnetic property of iron—which attracts iron but not sand—one can separate a combination of iron filings and sand.
To learn more about physical properties, refer to the link:
https://brainly.com/question/18327661
#SPJ2
Question 4 of 10
Based on information from the periodic table, what does this image
represent?
= 9 Protons
= 10 Neutrons
= 10 Electrons
A.Neutral fluorine
B. A positively charged fluoride ion
C. A negatively charged fluoride ion
D. A neutral neon atom
Answers
So we can cross out D.
Next, we can figure out a charge of an ion by looking at protons and electrons. Neutrons doesn’t matter since they’re neutral and only contribute to the mass. Protons and positive and electrons are negative. It’s like doing kindergarten math. John has 9 apples and he eats 10 (I know it’s not possible), how many apples does he have left? The answer is -1. The number of electrons are greater than the number of protons, so the ion is negative.
The answer is therefore C. A negatively charged fluoride ion.
Mg(s) + Ni2+(ag) -> Mg2+ (aq) + Ni(s) What is the total number of moles of electrons lost by Mg(s) when 2.0 moles of electrons are gained by Ni2+(ag)? * 10 ( 1.0 mol ,20 mol ,3.0 mol, 4.0 mol
Answers
The total number of moles of electrons lost by Mg(s) when 2.0 moles of electrons are gained by Ni2+(ag) is also 2.0 moles of electrons.
How to find the number of moles?This is because in a chemical equation, the number of moles of electrons gained by the reducing agent (in this case Ni2+) is equal to the number of moles of electrons lost by the oxidizing agent (in this case Mg(s)).
In this redox reaction, Mg is being oxidized because it loses electrons and Ni is being reduced because it gains electrons. The oxidation and reduction process are occurring simultaneously, so the number of electrons lost by Mg(s) is equal to the number of electrons gained by Ni2+(ag).
Learn more about moles of electrons in brainly.com/question/512038
#SPJ1
The electrons that are gained by the \(Ni^{2+}\) ion is 2.0 moles of electrons.
What is the number of the electrons gained?We know that when there is a redox reaction, there would be the loss or gain of electrons in the process. The process is a simultaneous one so the electrons that are lost by one specie must as a matter of necessity be gained by another specie.
In this case, as we look at the reaction equation we can see that there are two electrons that have been lost by the magnesium atom and these two electrons would be gained by the Nickel II ion.
Learn more about redox reaction:https://brainly.com/question/13293425
#SPJ1
Solid potassium (K) conducts an electric current, whereas solid potassium iodide (KI) does not
because a. solid potassium is a nonmetal
b. solid potassium has a delocalized sea of electrons and KI has its cations and anions
too close together
c. KI has covalent bonds
d. Kl needs to be reacted with another compound first
Answers
Solid potassium (K) conducts an electric current, whereas solid potassium iodide (KI) does not because solid potassium has a delocalized sea of electrons and KI has its cations and anions.
How does potassium iodide work?Positively charged potassium and negatively charged iodine combine to form the straightforward inorganic ionic chemical potassium iodide. It is solid at room temperature and resembles sodium chloride or table salt in terms of crystalline structure.
The major uses of potassium iodide are to treat hyperthyroidism and as a dietary supplement for those who are iodine deficient. Additionally, it is utilized as an expectorant to treat asthma and other long-term respiratory disorders, as well as to shield the thyroid gland from radiation.
When potassium iodide is dissolved in water, it splits into cations and anions, enabling free electron movement across the solution. It does not conduct electricity when it is in a dry crystal state, though, like other ionic compounds.
To know more about potassium iodide, visit:
https://brainly.com/question/12050170
#SPJ13
6. How many moles are in 8.30 x 1023 molecules of CO₂?
a.
b.
C.
d.
1.37
2.8
55.5
100
Answers
Please help!
Fe2O3 + C → CO + Fe

Answers
How many mL of a 0.75 N KOH solution
should be added to a 500 mL flask to make
500 mL of a 0.300 M KOH solution?
Answers
The amount of volume of KOH solution that should be added to make 500mL of a 0.300M solution is 200mL.
How to calculate volume?The volume of a solution given the concentration can be calculated using the following expression;
CaVa = CbVb
Where;
Ca = initial concentrationVa = initial volumeCb = final concentrationVb = final volumeAccording to this question, we are to calculate how many mL of a 0.75 M OH solution that should be added to a 500 mL flask to make 500 mL of a 0.300 M KOH solution.
0.75 × Va = 500 × 0.3
0.75Va = 150
Va = 150/0.75
Va = 200mL
Learn more about volume at: https://brainly.com/question/14710169
#SPJ1
Answer fast please!!
A calorimeter contains 600 g of water at 25°C. You place a hand warmer containing 300 g of liquid
sodium acetate inside the calorimeter. When the sodium acetate finishes crystallizing, the temperature of
the water inside the calorimeter is 36.4°C. The specific heat of water is 4.18 J/g-°C. What is the enthalpy
of fusion (AH) of the sodium acetate? (Show your work.) Where necessary, use q = mH₁.
Answers
The enthalpy of fusion of sodium acetate is approximately -93.64 J/g.
To calculate the enthalpy of fusion (ΔH) of sodium acetate, we can use the principle of energy conservation. The heat lost by the hand warmer during the crystallization process is equal to the heat gained by the water in the calorimeter.
First, let's calculate the heat gained by the water in the calorimeter using the formula q = m × c × ΔT, where q is the heat gained or lost, m is the mass of the water, c is the specific heat capacity of water, and ΔT is the change in temperature.
q_water = m_water × c_water × ΔT_water
Given:
m_water = 600 g
c_water = 4.18 J/g-°C
ΔT_water = 36.4°C - 25°C = 11.4°C
q_water = 600 g × 4.18 J/g-°C × 11.4°C
q_water = 28092 J
Since the heat lost by the hand warmer during crystallization is equal to the heat gained by the water, we can write:
q_water = q_handwarmer
Now, let's calculate the heat lost by the hand warmer using the same formula:
q_handwarmer = m_handwarmer × c_handwarmer × ΔT_handwarmer
Given:
m_handwarmer = 300 g
c_handwarmer = unknown (specific heat capacity of sodium acetate)
ΔT_handwarmer = 36.4°C - initial temperature of sodium acetate
Since the sodium acetate undergoes crystallization, its temperature remains constant during this phase change. The temperature at which crystallization occurs is known as the freezing point of sodium acetate, which is approximately 58°C. Therefore:
ΔT_handwarmer = 36.4°C - 58°C = -21.6°C
Now, we can substitute the known values into the equation:
q_water = q_handwarmer
28092 J = 300 g × c_handwarmer × -21.6°C
To solve for c_handwarmer, we rearrange the equation:
c_handwarmer = -28092 J / (300 g × -21.6°C)
c_handwarmer ≈ 5.47 J/g-°C
The specific heat capacity of sodium acetate (c_handwarmer) is approximately 5.47 J/g-°C.
The enthalpy of fusion (ΔH) can be calculated using the equation ΔH = q_handwarmer / m_handwarmer:
ΔH = -28092 J / 300 g
ΔH ≈ -93.64 J/g
For more such questions on fusion visit:
https://brainly.com/question/3992688
#SPJ8
Volume (V) can be measured in ______or____.O grams (g) or liters (l)O milliliters (ml) or centimeters cubed (cm3)O milliliters (ml) or grams (g)O kilograms (kg) or milligrams (mg)
Answers
According to this question, we must know that everything related to grams, means a mass of something.
So, we cannot use grams (g) or kilograms (kg) as a unit of volume.
For volume, we use liters (L), milliliters (mL), and cm^3 as we see on the written question. Also, we have plenty of units for volume in different systems used to measure.
Answer: milliliters (mL) or centimeters cubic (cm^3)
What is the smallest part of a gold ring that is still gold?
an atom
O an electron
O a molecule
O a compound
Answers
Answer: an atom
Explanation: he smallest particle of an element that still has the element's properties is an atom. All the atoms of an element are alike, and they are different from the atoms of all other elements. For example, atoms of gold are the same whether they are found in a gold nugget or a gold ring
how does ease of ion pair formation depend on concentration.
Answers
200.0 mL of a 0.600 M solution of KCl is diluted to 600.0 mL. What is
the new concentration of the solution?
Answers
Answer:
0.200 M
Explanation:
Begin by finding how many moles of KCl there are by using the molarity and given volume:
\(\frac{0.600 mol}{1000 mL} x \frac{200.0 mL}{1} = 0.12 mol KCl\)
(Remember that molarity is mol/L, which is equal to mol/1000 mL.)
Next, divide the moles of KCl by the new volume (after dilution):
\(\frac{0.12 mol KCl}{600.0 mL} x \frac{1000 mL}{1 L} = \frac{0.200 mol KCl}{1 L} = 0.200 M\)
The new concentration is 0.200 M.
The new concentration of the KCL solution is 0.200M
HOW TO CALCULATE CONCENTRATION:
The concentration or molarity in mol/L or M of a solution can be calculated using the following formula:C1V1 = C2V2
Where;
C1 = initial concentration of the solution
C2 = final concentration of the solution
V1 = initial volume of the solution
V2 = final volume of the solution
According to the information given in this question, we have the following:V1 = 200mL
V2 = 600mL
C1 = 0.600 M
C2 = ?
C2 = C1V1 ÷ V2
C2 = (0.600 × 200) ÷ 600
C2 = 120 ÷ 600
C2 = 0.2M
Therefore, the new concentration of the KCL solution is 0.200M.Learn more: https://brainly.com/question/4401006?referrer=searchResults
Peter woke up in the middle of night in darkness. Without being able to see, he was able to make his way to the bathroom without running into any objects in his path. Which of the following best describes how he is able to do this?
Answers
The answer choice that best describes this is C. Peter's brain still received messages from his other sensory receptors, which his brain combined with stored memories in order to move through the darkness.
How is Peter able to move in the dark?Here, the sensory organs in Peter's eyes, ears, nose, mouth, and internal organs are specialized organs that allow each receptor type to convey a different sensory modality to eventually integrate into a single perceptual frame.
As a result of these sensory organs, Peter will eventually go stuttering when he wakes up in the middle of the night in complete darkness. However, he was able to make his way to the bathroom without bumping into any objects in his path.
Read more about memory here:
https://brainly.com/question/14288553
#SPJ1
Peter woke up in the middle of night in darkness. Without being able to see, he was able to make his way to the bathroom without running into any objects in his path.
Which of the following best describes how he is able to do this?
A. Peter's brain received visual information from sensory receptors other than his eyes, so there was no change in how his body responded to the darkness around him.
B. Peter's brain used only stored memories as input when he moved through the darkness, as all sensory receptors are directly tied to the visual system.
C. Peter's brain still received messages from his other sensory receptors, which his brain combined with stored memories in order to move through the darkness.
D. Peter's brain used only messages received from his other sensory receptors when he moved through the darkness, as the visual system is not an important part of the central nervous system.
How many moles are in 15 grams of Li?
Answers
Determine the molecular formulas and then draw Line structures for the following condensed structures

Answers
No, these three structures do not represent the same molecule. They all have different arrangements of atoms and functional groups.
Define functional groups ?
Functional groups are specific groups of atoms or bonds within molecules that determine the chemical properties and reactivity of those molecules. They often determine the physical and chemical properties of a molecule, such as its solubility, acidity, and chemical reactions.
(a) Molecular formula: \(C_{13}H_{25}BrO\)
Line structure:
\(CH_3\)
|
\(CH_3-C-CH_2-CH_2-CO-CH_2-CHBr-CH(CH_3)_2\)
|
\(CH_3\)
(b) Molecular formula: \(C_{13}H_{25}BrO\)
Line structure:
\(CH_3\)
|
\(CH_3-C-CH-CH-CH-OH\)
| | |
\(CH_3 Br CH_2CH(CH_3)_2\)
(c) Molecular formula: \(C_{12}H_{23}BrO\)
Line structure:
\(CH_3\)
|
\(CH_3-C-CH-Br\)
|
\(CH_2CH(CH_3)_2-CO-CH_2-CH(CH_3)_2\)
No, these three structures do not represent the same molecule. They all have different arrangements of atoms and functional groups. Although they have the same molecular formula of C13H25BrO, the isomers differ in the way the atoms are connected to each other, leading to different chemical and physical properties.
To learn more about isomers visit the link :
https://brainly.com/question/12796779
#SPJ1
Patrick is a 16 year old boy whose body has stopped producing osteoclasts. What does this mean for his bones? What other parts of his body will be affected by this?
Answers
Hope this helps!
Have an AWESOME day! :)