what is the missing product from this reaction? 22888 ra → _____ 42 he
Answers
The missing product from the reaction ²²⁸₈₈Ra → _____ + ⁴₂He is ²²⁴₈₆Rn.
In this nuclear reaction, ²²⁸₈₈Ra (radium-228) undergoes alpha decay, releasing an alpha particle (⁴₂He). To determine the missing product, we need to subtract the atomic and mass numbers of the alpha particle from those of the radium-228 nucleus:
Mass number: 228 - 4 = 224
Atomic number: 88 - 2 = 86
Thus, the missing product is an isotope with a mass number of 224 and an atomic number of 86, which is radon-224 (²²⁴₈₆Rn).
The complete nuclear reaction is ²²⁸₈₈Ra → ²²⁴₈₆Rn + ⁴₂He.
To know more about nuclear reaction, click here
https://brainly.com/question/16526663
#SPJ11
Related Questions
What volume of an hcl solution with a ph of 1. 3 can be neutralized by one dose of milk of magnesia?.
Answers
480 mL of the HCl solution with a pH of 1.3 can be neutralized by one dose of milk of magnesia assuming the concentration of magnesium hydroxide is 0.2 M.
To determine the volume of \(HCl\) solution that can be neutralized by milk of magnesia, we need to know the concentration of the milk of magnesia.
Assuming milk of magnesia is a suspension of solid magnesium hydroxide in water, we need to know the concentration of magnesium hydroxide \((Mg(OH)2)\) in the suspension.
Let's assume that the concentration of magnesium hydroxide in milk of magnesia is 0.2 M.
The balanced chemical equation for the neutralization reaction between \(HCl\) and\(Mg(OH)2\)is:
\(2HCl + Mg(OH)2 - > MgCl2 + 2H2O\)
From the equation, we can see that two moles of \(HCl\) react with one mole of \(Mg(OH)2\).
To determine the volume of \(HCl\) solution, we need to calculate the number of moles of \(Mg(OH)2\) in one dose of milk of magnesia:
0.2 M = 0.2 moles / liter
Let's assume one dose of milk of magnesia is 30 mL, or 0.03 L. Then the number of moles of \(Mg(OH)2\) in one dose is:
0.2 moles / L x 0.03 L = 0.006 moles Mg(OH)2
Therefore, this amount of \(Mg(OH)2\) would require:
2 x 0.006 = 0.012 moles of \(HCl\) for complete neutralization
Now, let's calculate the volume of \(HCl\) solution needed to provide 0.012 moles of \(HCl\).
The volume of \(HCl\) solution can be calculated using the balanced chemical equation and the molarity of the \(HCl\) solution:
2 moles HCl / 1 mole \(Mg(OH)2\) x 0.012 moles \(Mg(OH)2\) / 1 = 0.024 moles HCl
\(pH = -log[H+]1.3 = -log[H+]\\[H+] = 5 x 10^-2 M\)
Now we can calculate the volume of the HCl solution using the equation:
moles = concentration x volume
0.024 moles = \(5 x 10^-2 M x volume\)
volume = 0.48 L or 480 mL
To know more about milk of magnesia refer to-
https://brainly.com/question/22066653
#SPJ11
What is the net ionic equations of Cu (s) + 2 AgC2H3O2 (aq) --> Cu(C2H3O2)2(aq) + 2 Ag(s) ?
Answers
Answer:
Explanation:
Cu(s) + 2Ag+ ======> Cu^(2+) + Ag(s)
Notice what happened. Silver started out as an ion. It took on 2 electrons that were donated by the copper.
The silver then became a solid ppte. This equation wil take on much more meaning when you take up oxidation and reduction. Until then, you need only know who gave up elections and who took them up.
What is the concentration of a sodium hydroxide solution if 28.1 cm^3 of it are exactly neutralized by 30.0 cm^3 of a 0.32 M hydrochloride acid solution

Answers
In order to find the concenctration of this base we will use the titration formula, which is:
Ma * Va = Mb * Vb
Where:
Ma = concentration of acid
Va = volume of acid
Mb = concentration of base
Vb = volume of base
0.32 * 30.0 = Mb * 28.1
9.6 = 28.1Mb
Mb = 0.34
Therefore the concentration of Sodium hydroxide is 0.34M
0.0045 x 0.000789 sig fig
Answers
Answer:
The answer IS 0.0000035505. Hope it helps!
Explanation:
Answer: if you put it in a caculator you would get 0.0045 x 0.000789= 0.0000035505
Explanation:
magnesium has three naturally occuring isotopes. the masses and bundances of the isotopes are given in the table below. what is the average atomic mass of mg?
Answers
According to the question,Average atomic mass of Mg = 24.3050.
What is the mass ?Mass is the measure of the amount of matter in an object. It is related to the concept of weight, which is the measure of the gravitational force that acts on an object. Mass can be measured in a variety of ways, including weighing, using a beam balance, or by using an instrument such as a spring scale. Mass is usually measured in units of kilograms or grams. Mass is an important concept in physics and is one of the properties used to describe an object's motion.
24Mg (78.99%)
25Mg (10.00%)
26Mg (11.01%)
Average atomic mass of Mg = 24.3050
24Mg*(78.99%) + 25Mg*(10.00%) + 26Mg*(11.01%) = 24.3050
To learn more about mass
https://brainly.com/question/837939
#SPJ4
This Venn diagram is used to compare the products and reactants of a reaction of baking soda and vinegar. What could be put into the intersection? Precipitates Chemical compounds Solid compounds Gases
Answers
I think and I'm sure it is gases
Answer:
gasses
Explanation:
i took the quiz
Consider the balanced chemical equation . How many moles of H 2O are produced when 1.0 mole of C 2H 6 reacts with excess O 2
Answers
Answer:
3.0 mol H₂O
Explanation:
Step 1: Write the balanced combustion equation
C₂H₆ + 3.5 O₂ ⇒ 2 CO₂ + 3 H₂O
Step 2: Establish the appropriate molar ratio
The molar ratio of C₂H₆ to H₂O is 1:3.
Step 3: Calculate the moles of H₂O formed from 1.0 mole of C₂H₆
1.0 mol C₂H₆ × 3 mol H₂O/1 mol C₂H₆ = 3.0 mol H₂O
A block weighing 13.15 grams has sides of 11.5 cm, 5.75 cm, and 8.44 cm. What is the density of the block in g/cm?
0.519 g/cm3
0.0236 g/cm3
42.4 g/cm
1.95 g/cm
Answers
Answer:
B. or .0236
Explanation:
Multiply the 3 sides to get 558.095, then divide the mass and volume to get .0236
the shrinking of a strawberry from water loss after being sprinkled with sugar is an example of
Answers
The shrinking of a strawberry from water loss after being sprinkled with sugar is an example of osmosis.
Osmosis is the movement of water from an area of high water concentration to an area of low water concentration. When sugar is sprinkled on a strawberry, the sugar molecules draw water out of the strawberry cells. This causes the strawberry to shrink.
Osmosis is a common process that occurs in living cells. It is important for the transport of nutrients and waste products in cells. Osmosis can also be used to preserve food.
For example, fruits and vegetables can be preserved by soaking them in a solution of sugar and water. The sugar solution will draw water out of the cells of the fruit or vegetable, making it difficult for bacteria to grow.
To know more about osmosis, refer here:
https://brainly.com/question/31028904#
#SPJ11
Which best describes a mixture? It has a single composition, and it has a set of characteristics that does not change. It can have different compositions, but it has a set of characteristics that does not change. It has a single composition, but it has a set of characteristics that does change. It can have different compositions, and it has a set of characteristics that does change.
Answers
Answer:
option 4 - it can have different compositions, and it has a set of characteristics that change.
Mixture is defined to have different compositions, and it has a set of characteristics that does change.There are 2 types of mixtures:1) homogenous mixtures 2)heterogenous mixtures
What are heterogenous mixtures?
Heterogenous mixtures is defined as a type of mixture where in the composition is not uniform throughout the mixture.It consists as two or more phases. The phases are chemically distinct from each other.
As there are two or more phases present in heterogenous mixtures they can be separated by solvent extraction where in one phase is miscible with the solvent and the other phase is immiscible.
Components of a heterogenous mixtures are distinctly visible . There are two types of heterogenous mixtures, the colloids and the suspensions.These two vary with each other with respect to particle size.
Learn more about heterogenous mixtures,here:
https://brainly.com/question/24898889
#SPJ6
which set of trials should be used as experiment a (expa) and experiment b (expb) to study the impact of the initial concentration of y?
Answers
The correct answer is: Trial 1 (expA) and Trial 2 (expB).
What is a reaction rate?The rate at which a chemical reaction occurs is known as the reaction rate. It is described as the shift in a reactant or product's concentration over time. The rate of a chemical reaction depends on several factors, including the concentration of reactants, temperature, pressure, and the presence of catalysts or inhibitors.
To study the impact of the initial concentration of Y on the reaction rate, we need to select two experiments with the same initial concentrations of reactants except for Y. This will allow us to compare the effect of changing the initial concentration of Y while keeping other factors constant.
Looking at the table, we can see that the initial concentrations of reactants in Trials 1 and 2 are the same, while the initial concentration of Y is different. Therefore, we can use these two trials as experiment A (expA) and experiment B (expB) to study the impact of the initial concentration of Y.
To know more about inhibitors, visit:
https://brainly.com/question/14800463
#SPJ1
What is the heaviest element that was created in the Big Bang?
Hint: The early universe remained hot enough for fusion for only a short time.
a. Deuterium
b. Magnesium
c. Lithium
d. Iron
Answers
The heaviest element that was created in the Big Bang is lithium (option c).
During the early stages of the universe, known as Big Bang nucleosynthesis, the conditions were hot and dense enough for the fusion of protons and neutrons to form light elements. While hydrogen and helium were the most abundant elements produced, a small amount of lithium-7 was also synthesized. However, the production of heavier elements through fusion processes required the later formation of stars and subsequent stellar nucleosynthesis.
Hence, the correct option is option c.
Learn more about the big bang here:
https://brainly.com/question/17209127
#SPJ 4
(Please help)
What is the most common isotope for element X

Answers
Answer:
Isotope 2
Explanation:
Isotope 2 is the most abundant. Its percent abundance is 78.68% which is the largest percentage, and therefore the most common of the isotopes presented.
Good luck!
What is the formula to determine the mass in grams of 3.6 mols of H2SO4
Answers
The molecular mass of 3.6 moles of H₂SO₄.
So let us first calculate the molecular mass of 1 mole of H₂SO₄ and then when we calculate the molecular mass of 1 mole of H₂SO₄, we will calculate the molecular mass of 3.6 moles of H₂SO₄ by multiplying the molecular mass of 1 mole of H₂SO₄ by 3.6
Molecular mass of 1 mole of H₂SO₄ = 2*(molecular mass of Hydrogen) + (molecular mass of Sulphur ) + 4*(molecular mass of oxygen )
Molecular mass of 1 mole of H₂SO₄ = 2*1 + 32 + 4*16 = 98 grams
Mass of 3.6 moles H₂SO₄ = 3.6*98 =352.8 grams
To learn more about the mole concept, please refer to the following link -https://brainly.com/question/9758790
52. In the following reaction, which are the Spectator ions? *
Mg(NO3)2 (aq) + Na₂CO3 (aq) → 2NaNO3 (aq) + MgCO3 (s)
Answers
Answer:
nitrate and sodium are spectator ions .The reactions that form an aqueous solution are often a spectator ions.
fill in the blanks pls

Answers
Le Chatelier's principle predicts that if NH3 is introduced to the reaction, the equilibrium will move towards products (P) to partially balance the addition of reactant.
Since the concentration of the product will decrease if NO is eliminated from the reaction, the equilibrium will likewise change in favour of the products (P). Water being a product, the addition of H2O would change the reaction's direction towards reactants (R).
The equilibrium would shift towards reactants (R) if O2 were taken out of the process to make up for its loss. As the temperature rises, the equilibrium will move in favour of the reactants (R), or endothermic reactions. Reduced pressure will cause the equilibrium to shift in favour of the side that contains more gas molecules, in this example, towards products.
Learn more about le chatlier principle at :
https://brainly.com/question/14967447
#SPJ1
Susan is investigating physical changes. To do this, she places some ice into a large bowl and seals it with a lid. She leaves the bowl on the counter for several hours until all of the ice has melted. Using a balance, Susan determines that the mass of both the water and the ice are equal. Why is the mass of the ice and the water the same? (SC.8.P.9.1)
Answers
Answer:
No mass loss
Explanation:
The mass of ice and the water in the different state are equal because the same of quantity of matter is present in both state of matter of matter.
In essence, the mass of the physical change process is conserved. When mass is conserved, matter is neither created nor destroyed but can be changed from one form to the other. This is in compliance with the law of conservation of matter. So, no mass was lost in the physical change process and the mass will remain the same.How many electrons does calcium have in its outermost shell
Answers
Yes, calcium is defined as a metal because of both its physical and chemical traits. They all have an outer shell with two electrons and are very reactive. Those elements in the second column have two electrons ready to make compounds. It shouldn't surprise you that calcium has a valence of 2.
If you have a density of 2.5 g/mL and a mass of 5 g, what is the volume?
Answers
Answer:2 ml.
Explanation:Volume. 2.5 g/ml = 5g/Volume. Volume = 5g/(2.5 g/ml). = 2 ml.
Answer:
2ml
Explanation:
Study the image.
Which phrase describes this plate boundary?
does not occur in oceans
may form rift valleys
is a type of convergent boundary
is a region where earthquakes occur
Answers
The phrase that describes the plate boundary shown is D. is a region where earthquakes occur .
What is the plate boundary shown ?A transform boundary is the name given to this tectonic plate boundary. Two or more plates slip past one another in such a border. This shift is typically accompanied by some highly active earthquakes.
At a transform plate border, the grinding motion between the plates causes shallow earthquakes, significant lateral rock displacement, and a wide zone of crustal deformation. Essentially While no new crust is produced, subducted, or formed, and no volcanoes are formed, the fault produces earthquakes.
Find out more on plate boundaries at https://brainly.com/question/22005072
#SPJ1
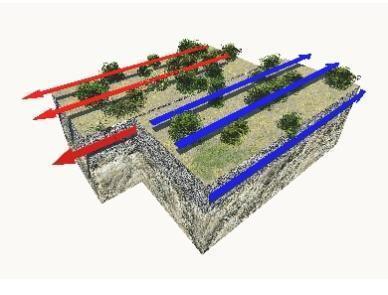
Describe three characteristics of the outer core and explain why the outer core is so important to humans on earth.
Answers
Answer:
The outer core is the only layer that is liquid. It is also mainly made from nickel and iron. The main job of the outer core is that it's responsible for the Earth's magnetic field. As the earth spins, the liquid inside this layer spins aswell, keeping it balanced.
one gallon milk is equal to how many milliliters of milk?
Answers
Answer:
3785.411784 mL
Explanation:
PLZ I NEED HELP I WILL GIVE 100 points for helping me
Silver nitrate reacts with barium chloride. If 39.02 grams of barium chloride are
reacted, how many grams of silver chloride are produced?
Answers
Answer:
4.2 g good luck with the answer
To find the range, identify the largest value and
the smallest value in the data set and find the
difference.
1, 2, 3, 3, 3, 4, 4, 4, 5, 7
What is the range of the data?
A. The largest value is 7 and the smallest value is 1. Find
the difference. 7-1-6 The range is 6.
B. The smallest value is 1. So the range is 1.
C. The largest value is 7. So the range is 7.
Answers
Answer:
A. The largest value is 7 and the smallest value is 1. Find the difference. 7 - 1 = 6.
Explanation:
How many atoms of silver are in a bracelet that has a mass of 13.94 grams?
Answers
Number of Silver atoms : N = 7.8 x 10²² atoms
Further explanationGiven
mass of bracelet : 13.94 g
Required
Number of atoms
Solution
Assuming the bracelet consists entirely of silver
mol of silver :
= mass : molar mass
= 13.94 g : 107,8682 g/mol
= 0.129
Number of atoms :
N = n x No(No = Avogadro's number)
N = 0.129 x 6.02 x 10²³
N = 7.8 x 10²² atoms
Given that the pka of acetic acid is 4.8, what would be the likely result of adding 1 mL of 0.1 M HCl to a 0.1 Macetate buffer at a pH of 5.0?
A. The pH would drop slightly.
B. The pH would increase significantly.
C. The pH would drop significantly.
D. The pH would increase slightly.
Answers
The pKa of acetic acid is 4.8, the result of adding 1 mL of 0.1 M HCl to a 0.1 M acetate buffer at a pH of 5.0 is:
C. The pH would drop significantly.
Acetic acid (CH₃COOH) is a weak acid that partially dissociates in water, releasing hydrogen ions (H⁺). In an acetate buffer solution, acetic acid and its conjugate base, acetate (CH₃COO⁻), are present in equilibrium:
CH₃COOH ⇌ H⁺ + CH₃COO⁻
The equilibrium is governed by the acid dissociation constant, represented as Ka. The pKa of acetic acid is given as 4.8. The pKa is the negative logarithm of the Ka value.
When a strong acid, such as HCl, is added to the acetate buffer solution, it introduces a large concentration of additional H⁺ ions. This disturbs the equilibrium between acetic acid and acetate, shifting it towards the formation of more undissociated acetic acid. Consequently, the concentration of H⁺ ions increases, causing a significant drop in pH.
Since the initial pH of the acetate buffer is 5.0, which is higher than the pKa of acetic acid (4.8), the buffer is in a partially deprotonated state. Adding HCl, a strong acid, disrupts the balance between the acid and its conjugate base, resulting in a more acidic solution and a significant decrease in pH.
Therefore, option C, "The pH would drop significantly," is the likely result of adding 1 mL of 0.1 M HCl to the 0.1 M acetate buffer at a pH of 5.0.
To know more about acetic acid here
https://brainly.com/question/15202177
#SPJ4
Chemical composition and function of iodized salt
Answers
Answer:
What is the chemical composition of iodized salt?
Iodized salt is a very cheap and good way of getting the necessary iodine. Ordinary table (cooking) salt is a chemical compound called "sodium chloride" (its chemical formula is NaCl). "Sodium iodide" (NaI) is added to the NaCl to make iodized salt.
Salt that has been iodized contains trace levels of sodium or potassium iodide. It's just regular salt sprinkled with potassium iodate. It has the same appearance and flavor! The bulk of table salt used currently is iodized, which has a number of advantages.
function of iodized salt
Iodized salt aids in the production of hormones that control heart rate and blood pressure. It also aids in the burning of unwanted fat deposits that may lead to heart disease. Salt maintains a healthy amount of hydration and electrolyte balance.
Jason shot a bb straight up in the air with a velocity of 105 m/s.what will the velocity of the bb when it is at a height of 203 m?
Answers
Answer:
The velocity of the bb when it reaches a height of 203 m can be determined using the laws of projectile motion. Since the bb is moving vertically upwards, its velocity at that height will be zero.
brainlest?
Answer: v = 83.96 m/s
Assuming the acceleration due to gravity is approximately 9.8 m/s^2, we can use the principles of projectile motion and energy conservation.
Using the equation for the vertical displacement of an object in free fall:
Δy = (v₀² - v²) / (2g)
Δy = vertical displacement (203m)
v₀ = initial velocity (105 m/s)
v = final velocity (not known yet)
g = accerlation due to gravity (9.8 m/s^2)
Lets rearrange the equation to solve for the final velocity:
v = v = √(v₀² - 2gΔy)
Substituting the given values:
v = √(105² - 2 * 9.8 * 203)
v ≈ √(11025 - 3979.6)
v ≈ √(7054.4)
v ≈ 83.96 m/s
Therefore, when the BB pellet is at the height of 203m, its velocity will be approximately 83.96 m/s.
How many moles of Helium atoms are present in 11.2 L of He gas at STP
Answers
There are 0.5 moles of helium atoms present in 11.2 L of Helium gas at Standard Temperature Pressure.
At STP (Standard Temperature and Pressure), which is defined as 0°C (273.15 K) and 1 atm of pressure, one mole of any gas occupies a volume of 22.4 L. Therefore, we can use the following formula to calculate the number of moles of helium atoms present in 11.2 L of He gas at STP:
n = V/ Vm
where n is the number of moles of helium atoms, V is the volume of the gas (11.2 L), and Vm is the molar volume of the gas at STP (22.4 L/mol).
Substituting the values, we get:
n = 11.2 L / 22.4 L/mol
n = 0.5 mol
Therefore, there are 0.5 moles of helium atoms present in 11.2 L of He gas at STP.
learn more about atoms here:
https://brainly.com/question/29695801
#SPJ4
Based on the video you just watched, what is contributing to the change in sea level in the Northern Patagonian Ice Field. Why is this area important for climate science research?
Answers
Answer:
The area is important for climate science research because they can look at the glaciers and see over the years what is happening with them, are they multiplying or are they decreasing and if so at what rate.
Answer:
Melting glaciers are causing sea levels to rise. It’s important to study natural conditions in the Northern Patagonian Ice Field because the landscape there is dynamic and the effects of climate change can easily be seen. The retreat of the Nef Glacier and the disappearance of icebergs on the lake are evidence of rising temperatures and climate change.
Explanation: