Answers
Answer:
Explanation:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹⁴ 5d¹⁰ 6p⁶ 7s² 5f¹⁴ 6d⁸
The element is Darmstadtium . It is a d block element . Its symbol is Ds . Its atomic no is 110 .
The element having the given electronic configuration with atomic number 110 is Darmstadtium (Ds).
From the electronic configuration, the number of electrons present in the atom can be calculated.
The number of electrons in the given electronic configuration are :
2+2+6+2+6+2+10+6+2+10+6+2+14+10+6+2+14+8
The number of electrons in the given configuration = 110
The number of electrons in the atoms has been equal to the atomic number. The element having the atomic number 110 is Darmstadtium.
The element belongs to the d block with the symbol Ds. It has been an artificially produced element.
For more information about electronic configuration, refer to the link:
https://brainly.com/question/16762037
Related Questions
Write the chemical formula for the following structure. You can't write subscripts so just
do full size subscript numbers.

Answers
Answer:
H2O2
Explanation:
By numbering the atoms we know about that it’s Hydrogen Peroxide
1. What is the modern view of electrons in the quantum mechanical model?
Answers
Answer: An electron con only exist in a limited number of quantized energy levels.
Explanation:
6. MgCl2 is a strong electrolyte. If 5.25 g of MgCl2 is dissolved in 200.0 ml of solution calculate the concentration (molarity) of MgCl2
7. 6. MgCl2 is a strong electrolyte. If 5.25 g of MgCl2 is dissolved in 200.0 ml of solution calculate the concentration (molarity) of the magnesium ion
Answers
Molar concentration or molarity is a measure of the concentration of a solute in a solution that indicates the number of moles of solute that are dissolved in one liter of solution. This is, the molarity indicates the number of moles of solute that are dissolved in a given volume.
The molarity of a solution is calculated by the expression:
molarity= number of moles of solute÷ volume
Molarity is expressed in units moles/L.
Molarity of MgCl₂In this case, you have:
Mass of MgCl₂= 5.25 gMolar mass of MgCl₂= 95.21 g/moleMoles of MgCl₂= Mass of MgCl₂÷ Molar mass of MgCl₂= 0.055 molesVolume= 200 mL= 0.200 L (being 1000 mL= 1 L)Replacing in the definition of molarity:
molarity= 0.055 moles÷ 0.200 L
Solving:
molarity= 0.275 moles/L
Finally, 0.275 moles/L is the molarity of MgCl₂.
Molarity of the magnesium ionAn electrolyte that completely dissociates in solution is known as a strong electrolyte. This means that the concentrationof MgCl₂ is equal to the concentration of the magnesium ion.
Finally, the molarity of the magnesium ion is 0.275 moles/L.
Learn more about molarity:
brainly.com/question/10091778
brainly.com/question/27974698
brainly.com/question/20385036
#SPJ1
Many chemical processes involve substances in either the liquid or the gaseous state.
Which of the following statements is correct?
A. A specific mass of a gas has a fixed volume at room temperature
B. A specific mass of a liquid has a fixed volume at room temperature
C. 25 cm of gas contains more particles than 25 cm of liquid
D. Liquid particles placed inside a sealed container have fixed positions
[1 mark]

Answers
Answer:
B. A specific mass of a liquid has a fixed volume at room temperature
Explanation:
Matter exists in three states namely; gas, liquid and solid. Each of these states has its own unique property that distinguishes it from others.
- Gases are free moving and do not have a constant or fixed volume.
- Liquids have fixed volume and take on the shape of their container. This means that liquids have a fixed or constant volume at a room temperature.
Based on the above properties of gas and liquid states of matter, "A specific mass of a liquid has a fixed volume at room temperature" is the correct option.
Answer:
It could be the Bbbbbbbbbbb
What was the unknown metal?
Answers
Answer:
is there a picture or something?
Explanation:
Explanation:
Your question is incomplete please update it.
How many L are required to make 3.5 M hydrochloric acid using 1.1 moles?
Answers
0.31 L
General Formulas and Concepts:Math
Pre-Algebra
Order of Operations: BPEMDAS Brackets Parenthesis Exponents Multiplication Division Addition Subtraction Left to RightEquality Properties
Multiplication Property of Equality Division Property of Equality Addition Property of Equality Subtraction Property of EqualityChemistry
Atomic Structure
MolesAqueous Solutions
Molarity = moles of solute / liters of solutionEquilibrium - Acid/Base
Strong AcidsWeak AcidsWriting AcidsExplanation:Step 1: Define
[Given] 3.5 M HCl
[Given] 1.1 moles
[Solve] Liters solution
Step 2: Solve
Substitute in variables [Molarity]: \(\displaystyle 3.5 \ M = \frac{1.1 \ moles}{x \ L}\)[Solution] Cross-Multiply [Equality Property]: \(\displaystyle x \ L = \frac{1.1 \ mol}{3.5 \ M}\)[Solution] Divide: \(\displaystyle x = 0.314286 \ L\)Step 3: Check
Follow sig fig rules and round. We are given 2 sig figs.
0.314286 L ≈ 0.31 L
what are all the ways that a substance can change state?
Answers
Explanation:
hdhehdbrhdns dhdjdjdhrjs dhfirjr rudjdbe dbdud d
Answer:
Matter can change from one state to another if heated or cooled. If ice (a solid) is heated it changes to water (a liquid). This change is called MELTING. If water is heated, it changes to steam (a gas).
Explanation:
The half reaction with a more positive standard reduction potential will
Answers
The half reaction with a more positive standard reduction potential will proceed spontaneously in a redox reaction
The half reaction with a more positive standard reduction potential will undergo reduction when compared to the half reaction with a more negative standard reduction potential.
Oxidation-reduction reactions, often known as redox reactions, are a set of chemical reactions that involve electron transfer between reactants. In a redox reaction, one reactant is oxidized, losing electrons, while the other reactant is reduced, gaining electrons.
The oxidation half-reaction is the process of losing electrons and increasing the oxidation number, whereas the reduction half-reaction is the process of gaining electrons and decreasing the oxidation number. The total reaction is referred to as the redox reaction.
Half-reaction:Half-reaction refers to the two parts of an oxidation-reduction reaction that happen separately. A half-reaction must always be either an oxidation reaction or a reduction reaction. It also describes the movement of electrons and hydrogen ions in an equation.
Know more about redox reaction here:
https://brainly.com/question/21851295
#SPJ8
2.
Which mixture could be a useful buffer in a solution?
acetic acid (CH3CO2H) and hydrochloric acid (HCl)
sodium hydroxide (NaOH) and elemental sodium (Na)
ammonia (NH3) and ammonium chloride (NH4Cl)
acetic acid (CH3CO2H) and ammonia (NH3)
Pls answer quickly
Answers
Ammonia (\(NH_3\)) and ammonium chloride (\(NH_4Cl\)) mixture could be a useful buffer in a solution. Option C
A buffer is a solution that can resist changes in pH when small amounts of acid or base are added. It consists of a weak acid and its conjugate base or a weak base and its conjugate acid. The buffer system works by the principle of Le Chatelier's principle, where the equilibrium is shifted to counteract the changes caused by the addition of an acid or a base.
In option A, acetic acid (\(CH_3CO_2H\)) is a weak acid, but hydrochloric acid (HCl) is a strong acid. This combination does not form a buffer because HCl is completely dissociated in water and cannot provide a significant concentration of its conjugate base.
Option B consists of sodium hydroxide (NaOH), which is a strong base, and elemental sodium (Na), which is a metal. This combination does not form a buffer as there is no weak acid-base pair involved.
Option D contains acetic acid (\(CH_3CO_2H\)), a weak acid, and ammonia (\(NH_3\)), a weak base. Although they are weak acid and base, they do not form a buffer system together as they are both weak acids or bases and lack the required conjugate acid-base pair.
Option C, ammonia (\(NH_3\)), is a weak base, and ammonium chloride (\(NH_4Cl\)) is its conjugate acid. This combination can form a buffer system. When ammonia reacts with water, it forms ammonium ions (NH4+) and hydroxide ions (OH-).
The ammonium ions act as the weak acid, while the ammonia acts as the weak base. The addition of a small amount of acid will be counteracted by the ammonium ions, and the addition of a small amount of base will be counteracted by the ammonia, thus maintaining the pH of the solution relatively stable.
Therefore, option C, consisting of ammonia (\(NH_3\)) and ammonium chloride (\(NH_4Cl\)), is the suitable mixture that could be a useful buffer in a solution.
For more such question on buffer visit:
https://brainly.com/question/13076037
#SPJ8
Balance the chemical equation below
Na HCO3 + H 3 C 6 H 5 07 → CO₂ + +H₂0 + Na ₂ C6 H ₂ O 7
Answers
The skeletal equation is:
NaHCO3
Na2CO3 + H2O + CO2 • To equalize sodium, multiply NaHCO3 by 2 2NaHCO3 - Na2CO3 + H2O + CO2
The equation is balanced.
An atom's Lewis dot structure has four dots. Which of the following elements could it be, and why?
O Aluminum, because it is a metal with four electrons in period 3.
O Beryllium, because it is in period 2 and has four total electrons.
O Carbon, because it is in group 14 and has four valence electrons.
O Calcium, because it is an alkaline earth metal in period 4.
Answers
Answer:
Beryllium, because it is in period 2 and has four total electrons.
Explanation:
Answer:
Its carbon
Explanation:
lphins... Acid. (b) Chlorine reacts with red hot iron powder to give Iron(III) Chloride but not Iron (II) Chloride. Explain. (1Mark)
Answers
(a) Because acid is caustic, dolphins can perish from exposure to it. Acids are compounds that give other things protons (H+). Acid can react with the proteins and lipids in dolphins' skin when they come into touch with it, leading to chemical burns and damage to the underlying tissue. Systemic consequences from this include death.
(b) Because chlorine is a potent oxidizer, it interacts with red-hot iron powder to produce Iron(III) chloride (FeCl3) rather than Iron(II) chloride (FeCl2). FeCl3 is created when chlorine at high temperatures rapidly accepts electrons from iron atoms. Contrarily, iron interacts with HCl, a less potent oxidizer than chlorine, to produce FeCl2.
Learn more about chlorine at :
https://brainly.com/question/31560014
#SPJ1
2. If 1.204 X 1024 atoms of sodium is 20% of the total sodium atoms in sodium
chloride NaCl), how many grams of salt do you have ?
Answers
Answer:
584.2 g
Explanation:
Hello there!
In this case, since we know the 20% of the atoms correspond to sodium, we can compute the total atoms as shown below:
\(\frac{1.204 x10^{24}}{0.2}=6.02x10^{24}atoms\)
Which are also equal to 1 mol and the Avogadro's number of sodium chloride with a molar mass of 58.44 g/mol; thus, the grams of salt turn out to be:
\(6.02x10^{24}atoms*\frac{1mol}{6.022x10^{23}atoms} *\frac{58.44g}{1mol}\\\\=584.2g\)
Best regards!
Carbon dioxide contributes to atmospheric warming by
Answers
for the reaction below identify the structure of the product with appropriate formal charges and the appropriate number of hydrogens on the nitrogen atom
Answers
The given question is incomplete, hence, can not be answered. Still, adding some relevant information for your reference.
What are formal charges?
Formal charge (FC) is the charge associated with the atoms in a molecule, assuming that the electrons of all chemical bonds are shared equally between atoms, regardless of their relative electronegativities. In determining the optimal Lewis structure (or dominant resonance structure) of a molecule, structures are chosen such that the formal charge of each atom is as close to zero as possible.
In chemistry, the formal charge in the covalent view of a chemical bond is assigned to the atoms in a molecule assuming that all chemical bond electrons are equally shared between atoms, regardless of their relative electronegativities. is the virtual charge that Simply put, the formal charge is the difference between the number of valence electrons of an atom in the neutral free state and the number assigned to that atom in the Lewis structure. In determining the optimal Lewis structure (or dominant resonance structure) of a molecule, structures are chosen such that the formal charge of each atom is as close to zero as possible.
The given question is incomplete, hence, can not be answered. Still, adding some relevant information for your reference.
To know more about Formal charge, visit:
https://brainly.com/question/28446255
#SPJ4
Convert the following measurement
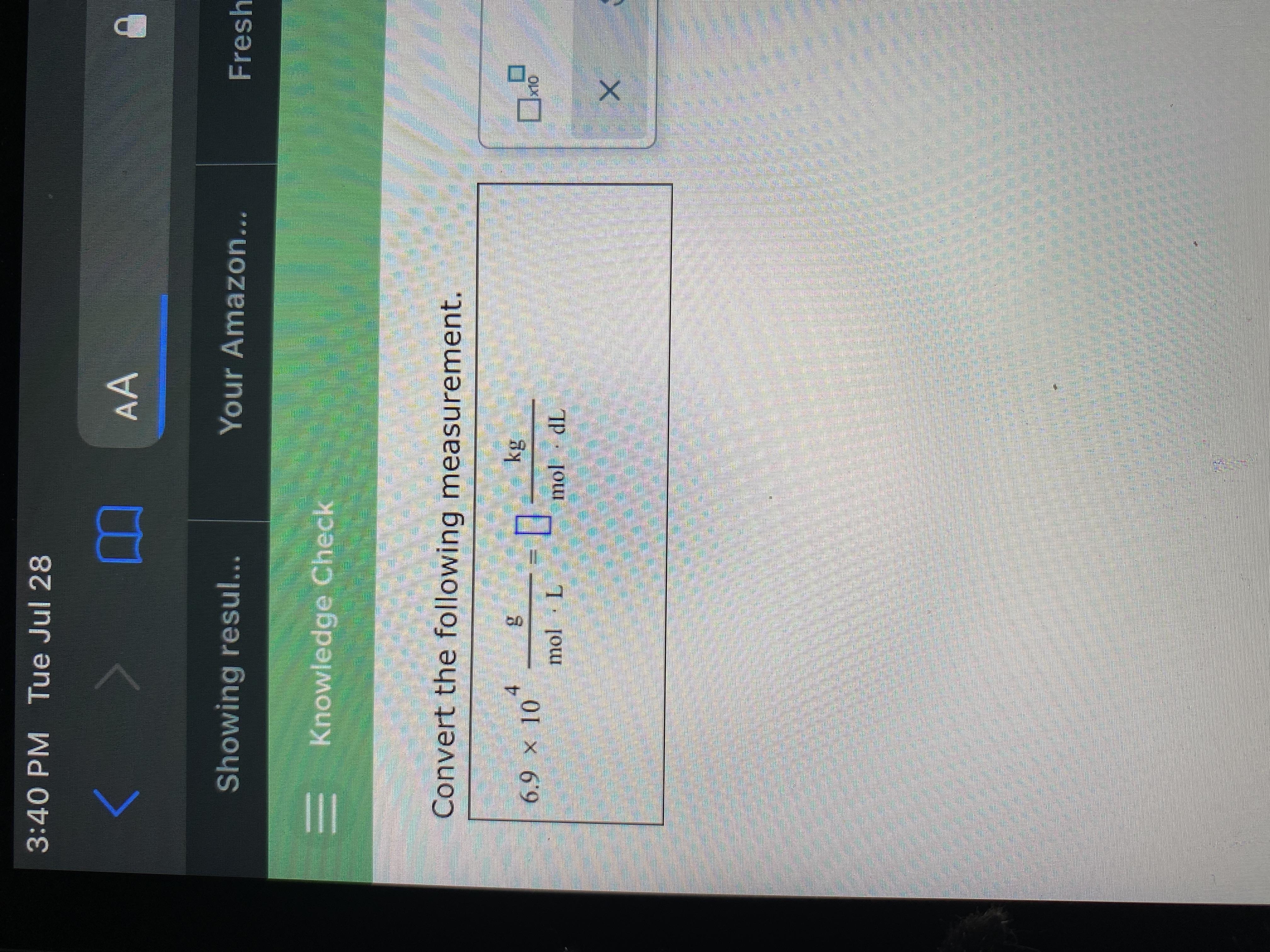
Answers
Answer:
6.9 Kg/mol•dL
Explanation:
To convert 6.9×10⁴ g/mol•L to kg/mol•dL,
First, we shall convert to kg/mol•L.
This can be achieved by doing the following:
Recall: 1 g = 1×10¯³ Kg
1 g/mol•L = 1×10¯³ Kg/mol•L.
Therefore,
6.9×10⁴ g/mol•L = 6.9×10⁴× 1×10¯³
6.9×10⁴ g/mol•L = 69 Kg/mol•L
Finally, we shall convert 69 Kg/mol•L to Kg/mol•dL.
This is illustrated below:
Recall: 1 L = 10 dL
1 Kg/mol•L = 1×10¯¹ Kg/mol•dL
Therefore,
69 Kg/mol•L = 69 × 1×10¯¹
69 Kg/mol•L = 6.9 Kg/mol•dL
Therefore, 6.9×10⁴ g/mol•L is equivalent to 6.9 Kg/mol•dL.
Will a precipitate form if solutions of magnesium acetate and strontium chloride are combined
Answers
A precipitate is an insoluble solute formed in a solution. No precipitate will form when magnesium acetate and strontium chloride react as they produce a soluble salt.
What is a precipitate?A precipitate is an insoluble particle that is formed when the ionic compounds react to produce insoluble salt.
Reaction when magnesium acetate and strontium chloride reacts is shown as:
Mg(CH₃COO)₂ (aq) +SrCl (aq) → MgCl₂ (aq) +Sr(CH₃COO)₂ (aq)
The substance containing acetate in the compound is always soluble and hence cannot make precipitate. Also, chloride gets soluble with magnesium.
Therefore, no precipitate will be formed.
Learn more about precipitate here:
https://brainly.com/question/5895133
#SPJ1
Liquid hexane CH3CH24CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. Suppose 5.2 g of hexane is mixed with 33.0 g of oxygen. Calculate the minimum mass of hexane that could be left over by the chemical reaction. Be sure your answer has the correct number of significant digits.
Answers
Answer:
The leftover is 15 g of oxygen rather than liquid hexane as the fuel limits the reaction.
Explanation:
Hello!
In this case, since the described combustion reaction is:
\(C_6H_{14}(l)+\frac{19}{2} O_2(g)\rightarrow 6CO_2(g)+7H_2O(g)\)
Thus, since 5.2 g of hexane (86.2 g/mol) is reacted with 33.0 g of oxygen (32.0 g/mol) we can compute the mass of hexane that was actually consumed via stoichiometry with oxygen (1:19/2 mole ratio):
\(m_{C_6H_{14}}^{consumed \ by\ O_2}=33.0gO_2*\frac{1molO_2}{32.0gO_2}*\frac{1molC_6H_{14}}{\frac{19}{2}gO_2 } *\frac{86.2gC_6H_{14}}{1molC_6H_{14}} \\\\m_{C_6H_{14}}^{consumed \ by\ O_2}=9.36gC_6H_{14}\)
It is proved then than the hexane won't have any leftover but oxygen does, as shown below:
\(m_{O_2}^{consumed \ by\ C_6H_{14}}=5.2gC_6H_{14}*\frac{1molC_6H_{14}}{86.2gC_6H_{14}} *\frac{\frac{19}{2}molO_2 }{1molC_6H_{14}} *\frac{32.0gO_2}{1molO_2} \\\\m_{O_2}^{consumed \ by\ C_6H_{14}}=18g\)
It means the leftover of oxygen is:
\(m_{O_2}^{leftover}=33g-18g\\\\m_{O_2}^{leftover}=15g\)
Regards!
please help :(
i have no idea what this even means lol

Answers
Here are the answers for A and B.
Answer:
(a) 80% are metals.
(b) (1.) Metals are malleable and ductile. (2.) Metals are dense.
Explanation:
(a) If you look at the picture, there are faint lines dividing the circle into ten parts. Metals take up 8 sections and non-metals take up 2. This means that the number of metals on the periodic table takes up 80% and non-metals take up 20%.
(b) There are many physical properties of metals, but two of them are that metals are malleable & ductile (these relate to how metal can be shaped) and that metals are dense (density = mass ÷ volume).
Convection currents in the atmosphere influence many weather patterns. What property of the air has the most influence on convection currents?
1. The direction of the wind
2. The velocity of the wind
3. The temperature of the air
4. The mass of the air
Answers
Convection currents are largely influenced by air temperature. A low-pressure area is produced when air is heated because it becomes less dense and rises.
What effect on the climate does convection have?For instance, the air above the Earth heats and rises as its surface warms from the sun. This air may continue to rise while cooling and producing cumulus clouds if the conditions allow.
How do air masses respond to convection currents?Climate-affecting wind patterns are driven by convection currents. Convection currents are used to shift air masses. There are two different kinds of air masses: warm and cold. From higher latitudes to lower latitudes, cold air masses travel.
To know more about temperature visit:-
https://brainly.com/question/30450112
#SPJ1
50.0ml each of 1.0M Hcl and 1.0M Naoh at room temperature (20.0c) are mixed the temperature of the resulting Nacl solutions increase to 27.5c
the density if the resulting Nacl solutuion 1.02 g/ml
the specific heat of the resulting Nacl solutions is 4.06j/gc
calculate the heat of neutralisation of hcl and naoh in kj/mol nacl products
Answers
Answer:
62.12kJ/mol
Explanation:
The neutralization reaction of HCl and NaOH is:
HCl + NaOH → NaCl + H₂O + HEAT
You can find the released heat of the reaction and heat of neutralization (Released heat per mole of reaction) using the formula:
Q = C×m×ΔT
Where Q is heat, C specific heat of the solution (4.06J/gºC), m its mass and ΔT change in temperature (27.5ºC-20.0ºC = 7.5ºC).
The mass of the solution can be finded with the volume of the solution (50.0mL of HCl solution + 50.0mL of NaOH solution = 100.0mL) and its density (1.02g/mL), thus:
100.0mL × (1.02g / mL) = 102g of solution.
Replacing, heat produced in the reaction was:
Q = C×m×ΔT
Q = 4.06J/gºC×102g×7.5ºC
Q = 3106J = 3.106kJ of heat are released.
There are 50.0mL ×1M = 50.0mmoles = 0.0500 moles of HCl and NaOH that are reacting releasing 3.106kJ of heat. That means heat of neutralization is:
3.106kJ / 0.0500mol of reaction =
62.12kJ/mol is heat of neutralization
Describe the trend of the reactivity of the elements in group VII
Answers
The non-metal elements in Group 7 – known as the halogens – get less reactive as you go down the group
Answer & Explanation:
The reactivity of elements in Group VII, also known as Group 17, decreases with increasing atomic radius. This is because halogens have high electronegativities and a proclivity to gain electrons in noble gas configurations. Myths are traditional stories or beliefs that explain cultural or societal beliefs, customs, or natural phenomena. They can be passed down through generations and can be based on true or fictitious events. Mythology, on the other hand, is the collection of myths associated with a specific culture or religion. Mythology can be amplified through retelling, incorporation into religious practices; association with significant events or figures, and adaptation into other media forms such as literature, film, or art.
How many moles of atoms are in each elemental sample?
(a) 4.6 X 1024 Pb atoms
(b) 2.87 × 10- He atoms
(c) 7.91 × 102 K atoms
(d) 4.41 × 10- Ca atoms
Answers
(a) For 4.6 x 10^24 Pb atoms, the number of moles is: 4.6 x 10^24 Pb atoms / 6.022 x 10^23 atoms/mol = 0.764 mol Pb
(b) For 2.87 x 10^-2 He atoms, the number of moles is: 2.87 x 10^-2 He atoms / 6.022 x 10^23 atoms/mol = 4.77 x 10^-26 mol He
(c) For 7.91 x 10^2 K atoms, the number of moles is: 7.91 x 10^2 K atoms / 6.022 x 10^23 atoms/mol = 1.31 x 10^-21 mol K
(d) For 4.41 x 10^-2 Ca atoms, the number of moles is: 4.41 x 10^-2 Ca atoms / 6.022 x 10^23 atoms/mol = 7.30 x 10^-26 mol Ca
Please help me am I doing right? If no so, what is the answer?

Answers
Answer:
C, O and H
Explanation:
CO is a combination of C and O, and H2O is both H and O
how do scientists qualitahtive observations to gather empirical evidence
Answers
Answer:
A hypothesis can be tested and accurately evaluated using different types of data collection, such as experiments that aim to produce a quantifiable or observable reaction, trials that replicate experiments to test their effectiveness (such as medication trials), or other methods of data collection.
Explanation:
There are no predetermined standards for observational evidence, often known as "empirical evidence," in matter science, other than the fact that it is helpful. The standards have evolved over time, just as science itself has. In fact, science only began to be effective with the advent of observation and test by observation.
Utilizing the century-old but still relevant notion of observation as a statistical hypothesis test, like any other hypothesis test, against observed parameters is one method to arrange observational data. A basic model like this may be used to deduce many things, but for our needs we can extract the following criteria (Iredia has previously addressed some of them):
ObservableQuantifiableTestableUseful uncertaintyRepeatableFor each science and type of test, a different quality criterion is used to evaluate the uncertainty sigma factor. But broadly speaking, consider this comparison:
Astronomy often use 7 sigma for observations, since they look at lots of systems and hence need to be vary of “once off” circumstances.Particle physics often use 5 sigma for observations, since they look at lots of energies and hence need to be vary of “look elsewhere” effects.Physics often use 3 sigma for observations, since they look at simple systems.Biologists often use 2 sigma for observations, since they look at complicated systems.Medicine may often have 80 % correct diagnosis and 60 % efficiency of treatments, which translates to 1–2 sigma uncertainty in outcome.It is useful as long as we maintain above 50% uncertainty with some degree of repeatability.
Thank you,
Eddie
Give me some examples of why is it important to understand how ocean currents flow.
Answers
Answer:
By moving heat from the equator toward the poles, ocean currents play an important role in controlling the climate. Ocean currents are also critically important to sea life. They carry nutrients and food to organisms that live permanently attached in one place, and carry reproductive cells and ocean life to new places.
Explanation:
:)
fill in the blank. if dissociation of mgcl2 in water were 100%, the van`t hoff factor would be___; however, for real solutions the van`t hoff factor for mgcl2 is (greater than, less than) this value.
Answers
If dissociation of mgcl2 in water were 100%, the van`t hoff factor would be 3 (three); however, for real solutions the van`t hoff factor for mgcl2 is (greater than, less than) this value.
MgCl2 dissociates into three ions when it dissolves in water, giving it a theoretical van't Hoff factor of 3. The van't Hoff factor for MgCl2 is normally tested at a value of no more than 2.7.
The van't hoff factor for magnesium chloride is 3. The letter I stands for the Van't Hoff factor. One magnesium cation and two chloride ions make up the last three ions formed when magnesium chloride separates. Consequently, the magnesium chloride van't hoff factor is equal to 3.
A solute's impact on associated properties, such as osmotic pressure, relative vapor pressure reduction, boiling-point elevation, and freezing-point depression, is measured by the van 't Hoff factor i.
To know about the van't hoff factor
https://brainly.com/question/21854669
#SPJ4
These organic solvents are used to clean compact discs:Solvent Density(g/mL)at 20∘ CChloroform 1.492Diehtyl ehter 0.714Ethanol 0.789Isopropanol 0.785Toluene 0.867(a) If a 15.00-mL sample of CD cleaner weighs 11.775 g at 20∘ C, Which solvent is most likely to be present? (b) The chemist analyzing the cleaner calibrate her equpment and finds that tha pipet is accurant ±0.02 mL, and the balance is accurate to ±0.003g. Is this equipment precise enough to distinguish between ethanol and isopropanol?
Answers
These organic solvents are used to clean compact discs: 0.785Toluene. so, option (d) is correct.
What is organic solvents?
The ability to dissolve or disperse one or more other compounds makes organic solvents a carbon-based chemical. Organic solvents may be neurotoxic, reproductively unsafe, and carcinogenic. Benzene, carbon tetrachloride, and trichloroethylene are examples of organic solvents that are carcinogenic.
What is ethanol ?
Ethanol (ethyl alcohol, CH3CH2OH) is one of a class of chemical substances (alcohols) whose molecules contain a hydroxyl group (OH) attached to a carbon atom. Fermentation of agricultural crops including sugarcane, corn, and manioc, among others, results in the production of ethanol.
Therefore, organic solvents are used to clean compact discs: 0.785Toluene. so, option (d) is correct.
Learn more about organic solvents from the given link.
https://brainly.com/question/9630611
#SPJ1
Hope Sergil
Check the Science and Interpreting Graphs
A group of students buit electromagnets using, wre, nails, and a 6V lentern battery. They decided design an
one set of investigations Use the graph to complete the organizer
nvestigation to test factors that they could chance to make a stronger electromagnet. The follow dete is from
Number of Wire Wraps and Magnetic Strength
Paperclips Magnet Picks Up
Independent Variable
Dependent Variable
12
10
8
6
4
Possible Research Question
What trends do you notice in the
data?
Write a claim based on this data
What evidence from the chart or
graph supports your claim?
25
vat
50
Number of Coils Wrapped Around
000
75
Answers
Answer:
Independent Variable: Number of Coils Wrapped Around
Dependent Variable: Number of Paperclips Magnet Picks Up
Possible Research Question: How does the number of wire wraps affect the strength of an electromagnet?
Trends noticed in the data: As the number of wire wraps (coils) around the nail increases, the magnetic strength of the electromagnet also increases. There is a positive correlation between the number of wire wraps and the number of paper clips the magnet picks up.
Claim based on the data: The magnetic strength of an electromagnet increases as the number of wire wraps (coils) around the nail increases.
Evidence from the chart or graph supporting the claim: The graph shows that as the number of wire wraps increases from 4 to 12, the number of paper clips the magnet picks up also increases from 6 to 25. This suggests a positive correlation between the two variables.
B) State right (V) or wrong (x): 1. The internal energy in the cyclic process has constant value. 2. An adiabatic system is one which cannot transfer energy but transfer matter to and from its surrounding. 3. Heat of combustion could be used in calculation of the heat of formation. 4. An isochoric process takes place at constant concentration.
Answers
Answer:
1. False. The internal energy of a system can vary during a cyclic process.
2. False. An adiabatic system is a system that does not exchange heat with its surroundings. It may still be able to transfer matter or energy in other ways.
3. True. The heat of combustion is the amount of heat produced when a substance is burned. It can be used to calculate the heat of formation, which is the heat absorbed or released when a substance is formed from its elements.
4. False. An isochoric process is a process that takes place at constant volume. It does not necessarily involve a change in concentration.
Explanation: