PLEASE HELP!! 74 POINTS
An igneous rock has small, dark crystals. Which statement also accurately describes this rock?
It is an intrusive rock.
It has a coarse texture.
It was found on Earth’s surface.
It was formed from slow cooling.
---------------------------------------------------------
Which is one factor that contributes to the formation of polar, temperate, and tropical zones?
the angle of the Sun’s rays
the direction of seasonal winds
the presence of prevailing winds
the movement of wind near a mountain
Answers
Answer:
It was formed from slow cooling.
the angle of the Sun’s rays
Explanation:
Answer:
D. It was formed from slow cooling.
Related Questions
Which is the correct step(s) for this reaction mechanism?
O NO2 + CO2 + CO NO + CO2
O step 1: NO2 + NO2 → NO + NO3 (slow)
step 2: NO3 + CO → NO2 + CO2 (fast)
O step 1: NO + NO3-NO + NO3 (slow)
step 2: NO3 + CO → NO + CO2 (fast)
O step 1: 2N02 + NO 2NO + NO3 (slow)
step 2: NO3 + CO → 2NO2 + CO2 (fast)

Answers
The reaction mechanims shows the sequence of steps through which a reaction occurs. The reaction mechanism for this reaction is;
step 1: NO2 + NO2 → NO + NO3 (slow)
step 2: NO3 + CO → NO2 + CO2 (fast)
What is reaction mechanism?We have to note that not all reactions occur in a single reactive encounter. If a reaction occurs in a single reactive encounter then it is called an elementary reaction.
The reaction mechanim shows the reaction sequence of a non elementary reaction. The correct mechanims of this reaction is shown as;
step 1: NO2 + NO2 → NO + NO3 (slow)
step 2: NO3 + CO → NO2 + CO2 (fast)
Learn more about a reaction mechanism: https://brainly.com/question/26690612
The reactant concentration in a zero-order reaction was 8.00×10−2 M
after 140 s and 4.00×10−2 M after 400 s
. What is the rate constant for this reaction?
Answers
The rate constant for the reaction is either 7.14×10−3 s−1 or 2.50×10−3 s−1, depending on which rate was used to calculate it.
Determining the rate constantThe rate of the reaction is given by the equation:
Rate = -k[A]
where k is the rate constant and [A] is the concentration of the reactant.
Rate at t=140 s:
Rate = (8.00×10−2 M - 0 M) / (140 s - 0 s)
= 5.71×10−4 M/s
Rate at t=400 s:
Rate = (4.00×10−2 M - 0 M) / (400 s - 0 s)
= 1.00×10−4 M/s
Since this is a zero-order reaction, the rate of the reaction is constant, and we can use either rate to calculate the rate constant:
k = Rate / [A]
Using the rate at t=140 s:
k = 5.71×10−4 M/s / 8.00×10−2 M = 7.14×10−3 s−1
Using the rate at t=400 s:
k = 1.00×10−4 M/s / 4.00×10−2 M
= 2.50×10−3 s−1
The rate constant for the reaction is either 7.14×10−3 s−1 or 2.50×10−3 s−1.
Learn more on zero-order reaction https://brainly.com/question/21663229
#SPJ1
PLS HELP GIVING BRAINLIEST AND 100 POINTS
Which of the following describes air temperature and air pressure at higher altitudes?
Air temperature increase while air pressure decreases
Air temperature and air pressure both decrease
Air temperature and air pressure both increase
Air temperature decreases while air pressure increases
Answers
The air pressure is low at higher altitudes. The pressure decreases up the higher altitudes. Same with temperature, as we go up the higher altitudes the temperature decreases as molecules becomes more far apart.
altitude ↑ || pressure ↓
altitude ↑ || temperature ↓
HELP Which is a force that holds atoms together?
chemical bond
electron shell
valence electron
chemical formula
Answers
Answer:
Chemical Bond
Explanation:
A Chemical Bond holds atoms together.
All of the others only apply to the parts within the atom. A chemical formula is just a written formula.
Answer: chemical bond
Explanation: took quiz - right

ASAP PLASE I WILL GIVE BRAINLIEST!!!!!
There are several ways to model a compound. One type of model is shown.
4 C's are connected in a line by 3 black lines. The C on the left has 3 H's attached to it each by a black line. The next C has 1 H attached by a black line and 1 O attached by 2 black lines. The next C has 2 H's attached to it by 1 black line each. The last C has 2 H's attached to it by 1 black line each and 1 O attached by a single black line. That O has an H attached to it by 1 black line.
What is the chemical formula for the molecule represented by the model?
CHO
C4H9O2
C4H8O
C3H8O2
Answers
According to the given description the chemical formula for the molecule represented by the model is C₄H₉O₂.
What is chemical formula?Chemical formula is a way of representing the number of atoms present in a compound or molecule.It is written with the help of symbols of elements. It also makes use of brackets and subscripts.
Subscripts are used to denote number of atoms of each element and brackets indicate presence of group of atoms. Chemical formula does not contain words. Chemical formula in the simplest form is called empirical formula.
There are four types of chemical formula:
1)empirical formula
2) structural formula
3)condensed formula
4)molecular formula
Learn more about chemical formula ,here:
https://brainly.com/question/29031056
#SPJ1
Why does nitrogen not show allotropy?
Answers
Answer:
Nitrogen does not show allotropy because of its small size and high electronegativity. The single N-N bond is weaker than P-P bond because of high inter electronic repulsions among non-bonding electrons due to the small bond distance. Hence it does not show allotropy.
Answer:
The nitrogen atom has short inter-bond distance, hence highly electronegative in terms of magnitude. This creates no relation in energy varieties hence no allotropes formed.
Nitrogen atom is also very small.
A certain ore is 21.3% nickel by mass. How many kilograms of this ore would you need to dig up to have 20.0 g of nickel?
Answers
If a certain ore is 21.3% nickel by mass. The number of kilograms of this ore would you need to dig up to have 20.0 g of nickel is: 93.897 kg.
How to find the mass of ore?Using this formula to determine the mass of ore
Mass of ore = Mass of nickel / Percentage of nickel by mass
Convert the percentage to decimal form by dividing it by 100
0.213 = 21.3%.
So,
Mass of ore = (20.0 g) / (0.213)
Mass of ore = 93.897 kg
Therefore the mass of ore is 93.897 kg.
Learn more about mass of ore here:https://brainly.com/question/10306443
#SPJ1
If you found a Carbon 13 atom, you would know that
Answers
Why is honesty important?
Answers
Honesty leads to a fulfilling, free life. Honesty is not just about telling the truth. It's about being real with yourself and others about who you are, what you want and what you need to live your most authentic life. ... Honesty sharpens our perception and allows us to observe everything around us with clarity.
(brainliest plz)
2. Show the calculation supporting the claim that atmospheric pressure near sea level corresponds to the pressure exerted by a column of mercury that is about 760 mm high. Considering the density of mercury = 13.6 g/cm³.
Answers
The pressure exerted by a column of mercury that is about 760 mm high corresponds to approximately 0.987 atm.
To calculate the pressure exerted by a column of mercury, we can use the formula:
Pressure = density * gravity * height
Given:
Density of mercury = 13.6 g/cm³
Height of the mercury column = 760 mm = 76 cm
Acceleration due to gravity = 9.8 m/s²
First, we need to convert the height of the mercury column from centimeters to meters:
Height = 76 cm * (1 m / 100 cm) = 0.76 m
Now, we can calculate the pressure:
Pressure = 13.6 g/cm³ * 9.8 m/s² * 0.76 m
To ensure consistent units, we need to convert the density from grams per cubic centimeter (g/cm³) to kilograms per cubic meter (kg/m³):
Density = 13.6 g/cm³ * (1 kg / 1000 g) * (1 cm³ / (1e-6 m³))
Density = 13600 kg/m³
Plugging in the values into the pressure formula:
Pressure = 13600 kg/m³ * 9.8 m/s² * 0.76 m
Pressure = 99992.8 Pa
We can express the pressure in terms of atmospheric pressure:
1 atm = 101325 Pa (approximately)
To compare the pressure with atmospheric pressure, we can convert 99992.8 Pa to atm:
Pressure in atm = 99992.8 Pa / 101325 Pa/atm
Pressure in atm ≈ 0.987 atm
The pressure exerted by a column of mercury that is about 760 mm high corresponds to approximately 0.987 atm. Since atmospheric pressure near sea level is approximately 1 atm, this calculation supports the claim that atmospheric pressure near sea level is equivalent to the pressure exerted by a column of mercury about 760 mm high.
for more such questions on pressure
https://brainly.com/question/24719118
#SPJ11
Balancing Chemical Equations, I need help with them

Answers
Answer: (look at the photo)
Explanation: I hope this helps have a nice day

Calculate the number of gold atom
present in 0.6g of 18 carat gold (24 carat
gold is taken as 100%
where and amu of
gold in 197
Teacher's Signature.
Answers
The number of gold atom : 1.204 x 10²¹
Further explanationGiven
0.6 g of 18 carat gold
mass number of gold = 197
24 carat as 100% gold
Required
The number of gold atom
Solution
Percentage of gold in 18 carat :
\(\tt \dfrac{18}{24}\times 100\%=75\%\)
Mass of gold in 0.6 g of 18 carat :
\(\tt 75\%\times 0.6~g=0.45~g\)
mol :
\(\tt mol=\dfrac{mass}{MW}=\dfrac{0.45}{197}=0.0023\)
A mole is a number of particles(atoms, molecules, ions) in a substance
1 mole = 6.02.10²³ particles
So the number of gold atom :
\(\tt 0.0023\times 6.02\times 10^{23}=1.204\times 10^{21}\)
Complete the table. Then explain how wavelength and frequency are mathematically related.
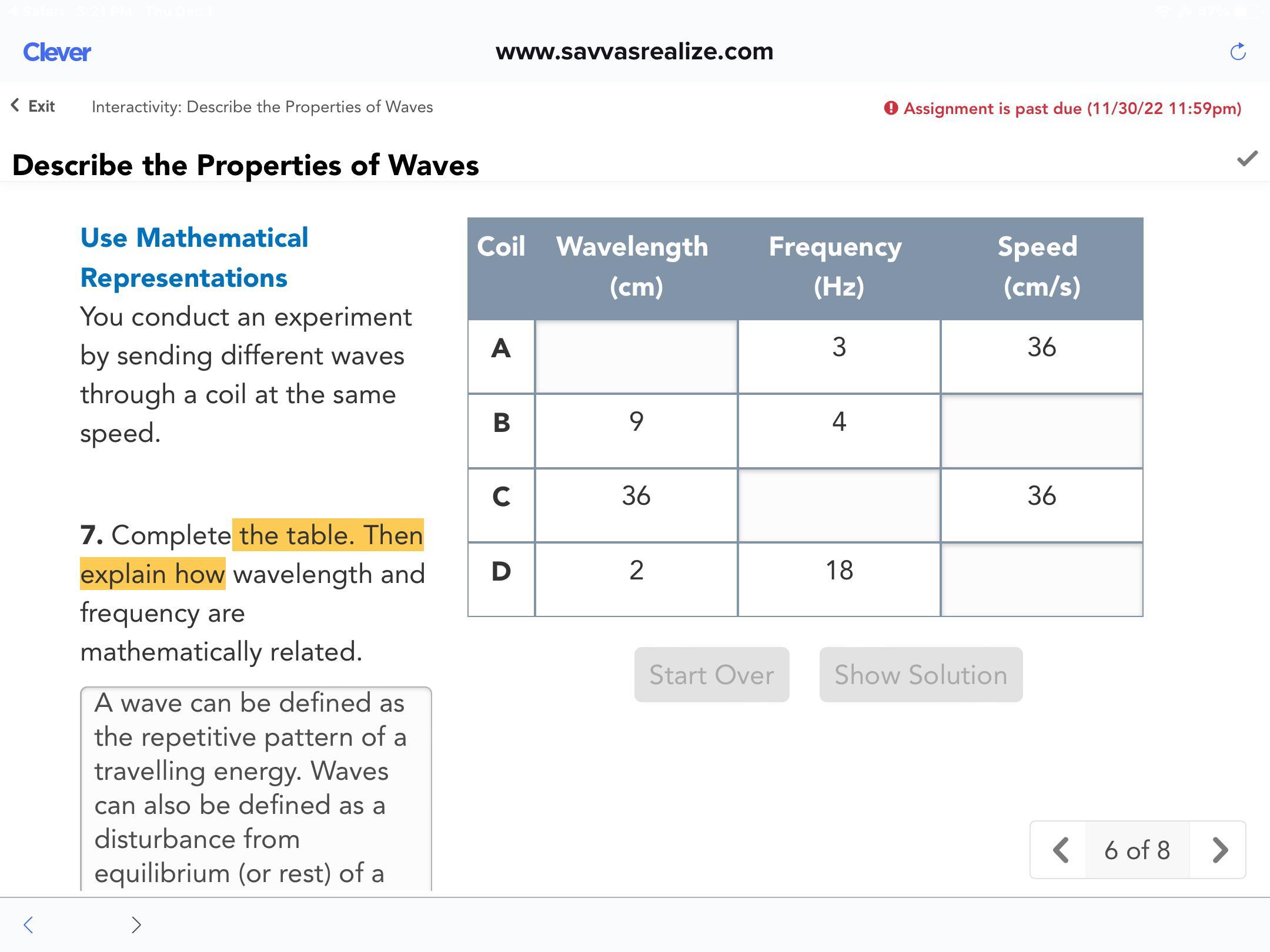
Answers
The wavelength is same.
What is wavelength?
Wavelength can be defined as the distance between two successive by throughs of a wave.
The question wants you to determine the energy that is the incoming photon by the must have in order to the accept the electron that melted it tob the jump to 2 to 1.
A good starting point here will be to calculate the energy of to the photon emitted when the electron falls from 1 to 2
by applying the Rydberg equation.
1/π = R
λ
si the wavelength of the emittted photon R
is the Rydberg constant, equal to 1.907.
This means that you haveλ=
4.10
So, you know that when an electron falls from the
to
a photon of wavelength by the
410 nm
is emitted. This implies by that in order to the for the electron by jump to the 2 to 1 .
it must absorb a photon of the same wavelength.
To know more about wavelentgh click-
https://brainly.com/question/10750459
#SPJ1
7. Which of the following medications should most likely not be taken prior to
operating heavy machinery?
a. Penicillin (antibiotic)
b. Famotidine (stomach)
C. Diphenhydramine (Benadryl)
d. Hydrochlorothiazide (diuretic)
Answers
Diphenhydramine medications should be taken by person who operate heavy machinery.
What is Medications?Medications refers to drugs or substances taken into the body to relieve of pains or symptoms of diseases.
What is Diphenhydramine?Diphenhydramine is also called Benadryl and is antihistamine take to relieve symptoms of pains, allergy, hay fever, and the common cold symptoms like cough, sneezing, sore throat, cold , cattarh and so on. It is used to treat dizziness, nausea, vomiting and motion sickness. Diphenhydramine can be taken to relax the body and fall asleep.
Therefore, Diphenhydramine medications should be taken by person who operate heavy machinery because it will help relive pains and symptoms of cold which can be due to vigorous activity of operating heavy machinery.
Learn more about Diphenhydramine from the link below.
https://brainly.com/question/247403520
A 1.90 g sample of elemental sodium, Na(s) is reacted with water, yielding sodium
hydroxide, NaOH, and hydrogen. 2Na(s) + 2H2O(l)→ 2NaOH(aq) + H2(g); The H2(g) is collected
over water at 18 oC. What are the partial pressures of the two gases (hydrogen and water
vapor) when contained in a 1.00 l container at 25
oC? What is the total pressure?
Answers
The partial pressure of hydrogen is 0.98 atm while the partial pressure of water is 0.02 atm. The total pressure of the system is 1 atm.
What is the pressure?We have seen the reaction as it has been shown in the equation that is attached to the question above. Now, we must have to find the number of moles of the hydrogen that was obtained.
Number of moles of sodium = 1.90 g/23 g/mol = 0.08 moles
2 moles of sodium produces 1 mole of hydrogen
0.08 moles of sodium would produce 0.08 moles * 1 mole/ 2 moles
= 0.04 moles
Given that;
P = pressure
V = volume
n = Number of moles
R = gas constant
T = temperature
PV = nRT
P = nRT/V
P = 0.04 * 0.082 * 298/1
P = 0.98 atm
The partial pressure of the hydrogen = 0.98 atm
The partial pressure of the water = SVP of water at 18°C = 0.02 atm
Total pressure of the system = 0.98 atm + 0.02 atm = 1 atm
The system has a total pressure of 1 atm.
Learn more about partial pressure:https://brainly.com/question/15075781
#SPJ1
When solving a problem it is important to identify your given and needed units, but it is also important to understand the relationship between those units so you will know how to set up your equation in order to solve the problem. Review the data sets below and use the steps of the problem-solving method to determine whether the given measurements would be appropriate for calculating mass, volume, or density.
a. 432 g of table salt occupies 20.0 cm^3 of space
b. 5.00 g 0T balsa wood, density of balsa wood : 0.16 g/cm^3
c. 32 cm^3 sample of gold density of gold 19.3 =g/cm^3
d. 150 g of iron, density of Iron = 79.0 g/cm^3
Answers
Answer:
See Explanation
Explanation:
Given
(a) to (d)
Required
Determine whether the given parameters can calculate the required parameter
To calculate either Density, Mass or Volume, we have
\(Density = \frac{Mass}{Volume}\)
\(Mass = Density * Volume\)
\(Volume = \frac{Mass}{Density}\)
(a) 432 g of table salt occupies 20.0 cm^3 of space
Here, we have:
\(Mass = 432g\)
\(Volume = 20.0cm^3\)
The above can be used to calculate Density as follows;
\(Density = \frac{Mass}{Volume}\)
\(Density = \frac{432g}{20.0cm^3}\)
\(Density = 21.6g/cm^3\)
(b) 5.00 g of balsa wood, density of balsa wood : 0.16 g/cm^3
Here, we have:
\(Mass = 5.00g\)
\(Density = 0.16g/cm^3\)
This can be used to solve for Volume as follows:
\(Volume = \frac{Mass}{Density}\)
\(Volume = \frac{5.00g}{0.16g/cm^3}\)
\(Volume = 31.25cm^3\)
(c) 32 cm^3 sample of gold density of 19.3 g/cm^3
Here, we have:
\(Volume = 32cm^3\)
\(Density = 19.3g/cm^3\)
This can be used to calculate Mass as follows:
\(Mass = Density * Volume\)
\(Mass = 32cm^3 * 19.3g/cm^3\)
\(Mass = 617.6g\)
(d) 150 g of iron, density of Iron = 79.0 g/cm^3
Here, we have
\(Mass = 150g\)
\(Density = 79.0g/cm^3\)
This can be used to calculate volume as follows:
\(Volume = \frac{Mass}{Density}\)
\(Volume = \frac{150g}{79.0g/cm^3}\)
\(Volume = 1.90 cm^3\) Approximated
Of the following, ________ is the most volatile. Select one: C2F6 C2H6 C2I6 C2Cl6 C2Br6
Answers
Answer:
C2Br6
because bromine is a liquid volatile
According to the forces of attraction and boiling point C₂Br₆ is most volatile as its boiling point is very low.
What are forces of attraction?Forces of attraction is a force by which atoms in a molecule combine. it is basically an attractive force in nature. It can act between an ion and an atom as well.It varies for different states of matter that is solids, liquids and gases.
The forces of attraction are maximum in solids as the molecules present in solid are tightly held while it is minimum in gases as the molecules are far apart . The forces of attraction in liquids is intermediate of solids and gases.
The physical properties such as melting point, boiling point, density are all dependent on forces of attraction which exists in the substances.
Learn more about forces of attraction,here:
https://brainly.com/question/14365107
#SPJ2
When the molecular reaction is correctly balanced, what is the stoichiometric coefficient for potassium nitrate?
Co(NO3)3 + K2S --->
Answers
The balanced equation for the reaction between cobalt(III) nitrate (\(Co(NO_3)_3\)) and potassium sulfide (\(K_2S\)) is:
\(3 Co(NO_3)3 + 2 K_2S - > 3 CoS + 6 KNO_3\)
In the balanced equation, the stoichiometric coefficient for potassium nitrate (\(KNO_3\)) is 6.
A molecular reaction is a chemical reaction in which two or more molecules combine to form a new compound. Molecular reactions include a wide range of natural and synthetic phenomena, ranging from enzyme-catalyzed transformations to protein folding and drug binding. In a chemical equation, stoichiometric coefficients reflect the relative number of molecules of each reactant and product.
Potassium nitrate is a chemical compound that is commonly used in fertilizers, fireworks, and rocket propellants. It is an ionic salt with the formula \(KNO_3\). Potassium nitrate is a white crystalline substance that is soluble in water. It is also known as saltpeter.What is the balanced chemical equation for the reaction of potassium nitrate and potassium sulfide?\(Co(NO_3)_3 + K_2S → CoS + 3KNO_3\)
This is the balanced chemical equation for the reaction of potassium nitrate and potassium sulfide. The stoichiometric coefficient for potassium nitrate is 3, which means that three molecules of KNO3 are required to react with one molecule of \(K_2S\). On the other hand, the stoichiometric coefficient for \(Co(NO_3)_3\) is 1, which means that only one molecule of \(Co(NO_3)_3\) is required to react with one molecule of \(K_2S\) .
The stoichiometric coefficient for CoS is also 1, which means that one molecule of CoS is produced for every molecule of \(K_2S\)that reacts.
For more such questions on stoichiometric coefficient
https://brainly.com/question/6666875
#SPJ8
Anthony is experimenting with how the amount of air inside a basketball
affects how high it bounces. He uses logical reasoning to conclude that he
should drop each ball from the same height. Why is this important?
A. Dropping the balls from different heights would take longer.
B. Dropping the balls from different heights would introduce error.
C. Dropping the balls from the same height will create more data for
the experiment.
D. Dropping the balls from the same height is less fun.
Answers
It is important to drop the balls from the same height because : ( B ) Dropping the balls from different heights would introduce error
The experiment Anthony is trying to perform is to determine the effect of the air inside a basketball on how high the ball will bounce, therefore other variables such as height should be kept constant ( i.e the same for every ball to be used for the experiment ).
If anthony drops the balls at different heights that will have a direct effect on the measurement of the bounce heights attained by each ball therefore creating room for error in the experiment .
Therefore we can conclude that It is important to drop the balls from the same height because Dropping the balls from different heights would introduce error
Learn more about experimental errors : https://brainly.com/question/2764830
(b) A 10.32g of AlCl3 are allowed to vapourize in 1dm³ vessel at 80°C a pressure of 1.7 x 10 NM2 develops. What is the degree of association into AICI3 into Al2Cl6?
Answers
The degree of association of AlCl3 into Al2Cl6 is 0.663. The degree of association of AlCl3 into Al2Cl6 can be determined using the ideal gas law and the van't Hoff factor.
Firstly, we need to calculate the number of moles of AlCl3 present in the vessel using the formula n = m/M, where m is the mass of AlCl3 and M is the molar mass of AlCl3.
n = 10.32g / 133.34 g/mol = 0.0774 mol
Next, we can use the ideal gas law equation PV = nRT to calculate the number of moles of particles in the gas phase. Rearranging this equation, we get:
n = PV/RT
where P is the pressure, V is the volume, R is the gas constant and T is the temperature in Kelvin.
n = (1.7 x 10 N/m²) x 1 dm³ / (8.31 J/mol/K x 353 K) = 7.55 x 10⁻⁴ mol
The van't Hoff factor (i) is the ratio of the actual number of particles in solution to the number of formula units dissolved. For a completely dissociated compound, the van't Hoff factor is equal to the number of ions produced. In the case of AlCl3, it undergoes a degree of association to form Al2Cl6, so the van't Hoff factor is less than 1.
We can now use the formula i = 1 + (α - 1)β, where α is the degree of association and β is the number of particles in solution per formula unit. For AlCl3, β = 4 (AlCl3 contains one Al and three Cl atoms), and assuming a degree of association of x, we get:
i = 1 + (x - 1) x 4 = 4x - 3
Substituting the values for n and i into the equation n = iC, where C is the concentration in mol/dm³, we get:
7.55 x 10^-4 mol = (4x - 3) C
Solving for x, we get:
x = 0.663
Therefore, the degree of association of AlCl3 into Al2Cl6 is 0.663.
To know more about van't Hoff factor, visit:
https://brainly.com/question/30905748
#SPJ1
. A potassium dichromate solution was standardized
with the following results: 0.1017, 0.1019, 0.1016,
0.1015 N.
(a) Within what range are you 95% confident that the
true value falls?
(b) Determine if there is a statistical difference between
the obtained mean concentration and the prepared
concentration of 0.1012 N
Answers
A) Calculate the lower and upper bounds of the 95% confidence interval:
Lower bound = X - ME
Upper bound = X + ME
B) you would compare the obtained mean concentration (0.101675 N) with the prepared concentration (0.1012 N) and calculate the t-value. The t-value is then compared to the critical t-value at a certain significance level (e.g., a = 0.05) and the degrees of freedom (n-1) to determine if there is a statistically significant difference.
(a) To determine the range within which you can be 95% confident that the true value falls, you can calculate the confidence interval using the obtained results.
First, calculate the mean (X) of the measured concentrations:
X = (0.1017 + 0.1019 + 0.1016 + 0.1015) / 4 = 0.101675 N
Next, calculate the standard deviation (s) of the measured concentrations:
s = sqrt(((0.1017 - X)² + (0.1019 - X)² + (0.1016 - X)² + (0.1015 - X)²) / (4 - 1))
Then, calculate the standard error of the mean (SE) using the formula:
SE = s / sqrt(n)
where n is the number of measurements (in this case, n = 4).
With the standard error, you can calculate the margin of error (ME) for a 95% confidence interval using the t-distribution. The t-value for a 95% confidence interval with 3 degrees of freedom is approximately 3.182.
ME = t * SE
Finally, calculate the lower and upper bounds of the 95% confidence interval:
Lower bound = X - ME
Upper bound = X + ME
(b) To determine if there is a statistical difference between the obtained mean concentration (0.101675 N) and the prepared concentration (0.1012 N), you can perform a t-test. The t-test compares the means of two sets of data to determine if they are significantly different from each other.
Using the formula for a two-sample t-test, you would compare the obtained mean concentration (0.101675 N) with the prepared concentration (0.1012 N) and calculate the t-value. The t-value is then compared to the critical t-value at a certain significance level (e.g., α = 0.05) and the degrees of freedom (n-1) to determine if there is a statistically significant difference.
If the calculated t-value is greater than the critical t-value, it suggests a statistically significant difference between the obtained mean concentration and the prepared concentration.
Please note that the actual calculations and interpretation may vary depending on the statistical software or method used, and it's always recommended to consult a statistician or refer to appropriate statistical references for accurate analysis.
For more question on significant
https://brainly.com/question/24630099
#SPJ8
In a chemical reaction
Zn(NO3)2 + NO2 + H₂O
a) What is redox reaction?.
b) Balance the reaction by oxidation number or ion electron method.
Answers
A redox reaction is a reaction in oxidation or the loss of electrons occurs simultaneously with reduction involving a gain of electrons.
The balanced equation of the redox reaction by the oxidation number method is as follows: Zn + 2HNO₃ ----> Zn(NO₃)₂ + 2NO₂ + H₂O
What is the balanced equation of the redox reaction?The complete equation of the redox reaction is given below as follows:
Zn + HNO₃ ----> Zn(NO₃)₂ + NO₂ + H₂OTo balance the chemical reaction by oxidation number, we need to ensure that the total change in oxidation numbers for each element is zero on both sides of the equation.
Let's assign the oxidation numbers to the elements:
In Zn(NO₃)₂, the oxidation number of Zn is +2, and the oxidation number of each NO₃ group is -1.
In HNO₃, the oxidation number of H is +1, the oxidation number of N is +5, and the oxidation number of each O in NO₃ is -2.
On the product side, the oxidation number of Zn is +2, and the oxidation number of each NO₃ group is -1. The oxidation number of N in NO₂ is +4, and the oxidation number of each O is -2. The oxidation number of H in H₂O is +1, and the oxidation number of O is -2.
Now, let's balance the reaction by considering the changes in oxidation numbers:
Zn: 0 → +2
H: +1 → 0
N: +5 → +4
O: -2 → -2
To balance the oxidation numbers, we need two NO₂ molecules on the product side. The balanced equation is:
Zn + 2HNO₃ ----> Zn(NO₃)₂ + 2NO₂ + H₂O
Learn more about redox reactions at: https://brainly.com/question/21851295
#SPJ1
How is carbon used by living things
Answers
Answer: Carbon is used by plants to build leaves and stems, which are then digested by animals and used for cellular growth.
Explanation:
Which of the following happens during a chemical change but does not happen during a physical change? (1 pO The mass of the substance changes.O Atoms of one or more substances rearrange into a new substance.O The state of the substance changes.O The appearance of a substance changes, but it is still the same substance.
Answers
Answer:
Atoms of one or more substances rearrange into a new substance. (second )
Explanation:
When a chemical change occurs, the original substance before the change is different from the substance after the change, in this case, that change is due to the fact that the atoms of one or more substances are rearranged into a new substance, since the other options include a physical change.
Form a group and discuss the possible reasons why EA, is a positive quantity for oxygen atom.
Answers
Answer:
Possible reasons why EA, or electronegativity, is a positive quantity for oxygen atom include:
Explanation:
What is the molarity of Ca(NO3)2 in a solution resulting from mixing 150.0 mL of 0.200 M HNO3 with 150.0 mL of 0.0100 M Ca(OH)2
Answers
The molarity of the Ca(NO3)2 solution would be 0.005 M
First, let us look at the balanced equation of the reaction:
\(2HNO_3+Ca(OH)_2 ---> Ca(NO_3)_2+2H_2O\)
The mole ratio of HNO3 to Ca(OH)2 is 2:1.
Mole of HNO3 = molarity x volume
= 0.2 x 150/1000
= 0.03 mole
Mole of Ca(OH)2 = 0.01 x 150/1000
= 0.0015
Thus, there is no limiting or excess reactant.
Also from the equation, mole ratio of Ca(OH)2 to Ca(NO3)2 is 1:1. Hence, the mole of Ca(NO3)2 would also be 0.0015 mole.
The total volume of the resulting solution would be: 150 +150 = 300 mL
Thus, the molarity of the resulting Ca(NO3)2 would be:
Molarity = mole/volume
= 0.0015/0.3
= 0.005 M
More on molarity can be found here: https://brainly.com/question/12127540
A burning match will burn more vigorously in pure oxygen than in air because _________ . Select one: a. oxygen is a catalyst for combustion b. nitrogen is a reactant in combustion and its low concentration in pure oxygen catalyzes the combustion c. oxygen is a product of combustion d. nitrogen is a product of combustion and the system reaches equilibrium at a lower temperature e. oxygen is a reactant in combustion and pure oxygen increases the reactant concentration
Answers
Answer:
e. oxygen is a reactant in combustion and pure oxygen increases the reactant concentration
Explanation:
The reaction of a burning match is combustion. In this combustion, the organic components of the match (such as cellulose, C₆H₁₀O₅) react with oxygen, producing water and carbon dioxide:
C₆H₁₀O₅(s) + 6O₂(g) → 5H₂O(g) + 6CO₂(g)Seeing as oxygen is a reactant and not a catalyst nor product, and that nitrogen plays no part in the reaction, the only correct answer is option e.
1. Which statement BEST explains why coal is used to generate electricity
despite the amount of pollution produced by burning coal?
A Compared to other fuels, coal produces more energy per kilogram.
B. Compared to other fuels, coal is relatively cheap and plentiful.
C. Coal burns without producing greenhouse gases.
D. Coal is a renewable source of energy.
Answers
Answer:
B
Explanation:
How many bonding electrons are in the Lewis structure of PCl₃?
5
6
4
2
Answers
Answer:
B.) 6
Explanation:
Attached below is the Lewis structure of PCl₃ . Since phosphorus (P) has 5 valence electrons and chlorine (Cl) has 7 valence electrons, there should be 26 valence electrons (5 + 7(3) = 26) in the Lewis structure.
Bonding electrons are the electrons present in the chemical bonds between two atoms.
There are 2 electrons shared in every single bond. Within PCl₃, there are 3 single bonds. As such, there are 6 bonding electrons in the Lewis structure of PCl₃.

How many atoms are in 12 g of Carbon-12 (12C)?
Answers
There are approximately 6.022 × 10^23 atoms in 12 grams of Carbon-12 (12C).
The number of atoms in a given amount of a substance can be calculated using Avogadro's number, which represents the number of atoms or molecules in one mole of a substance. Avogadro's number is approximately 6.022 × 10^23.
Carbon-12 is a specific isotope of carbon, with an atomic mass of 12 atomic mass units (amu). One mole of Carbon-12 has a mass of 12 grams. Since one mole of any substance contains Avogadro's number of particles, in the case of Carbon-12, it contains 6.022 × 10^23 atoms.
Therefore, if we have 12 grams of Carbon-12, which is equal to one mole, we can conclude that there are approximately 6.022 × 10^23 atoms in this amount of Carbon-12.
In summary, 12 grams of Carbon-12 contains approximately 6.022 × 10^23 atoms. Avogadro's number allows us to relate the mass of a substance to the number of atoms or molecules it contains, providing a fundamental concept in chemistry and enabling us to quantify and understand the microscopic world of atoms and molecules.
for such more questions on atoms
https://brainly.com/question/6258301
#SPJ8