Intermolecular forces are all some kind of: lecture 9
Answers
All intermolecular forces include electrostatic attraction in some way.
Electrostatic interactions are the source of intermolecular forces; this means that they are created when positively and negatively charged species come into contact with one another. Intermolecular interactions, much like covalent and ionic bonds, are the result of the addition of components that are both attractive and repulsive. The electromagnetic forces of attraction or repulsion that act between atoms and other types of neighboring particles, such as atoms or ions, are examples of intermolecular forces, also known as secondary forces. An intermolecular force, also known as an IMF (or secondary force), is the force that mediates the interaction between molecules.
To know more about intermolecular forces click on the link below:
https://brainly.com/question/12243368
#SPJ4
Related Questions
HELPPP
What do isotopes depend on?
A- Atomic number
B- mass number
C-protons
D-neutrons
Answers
Answer: D
Explanation:
if two atoms have the same number of protons, but different numbers of neutrons we refer to them as isotopes.
Isotopes have the same nom of protons ( atomic number ) and electrons , but they have different no. of neutrons , they aslo have different mass no ; since mass no equals to protons + neutrons .
PLEASE GIVE BRAINLIEST
Compared to the nucleus 5626fe, what is the density of the nucleus 112 48cd?
Answers
For a comparison of the nucleus 5626fe, the density of the nucleus 112 48cd is mathematically given as the same.
n(Cd) / n(Fe)=1
What is the density of the nucleus 112 48cd?Generally, the equation for the density is mathematically given as
d=\frac{A}{4/3}\piR^3
Therefore
n(Cd) / n(Fe) = [A (Cd) / (A Fe) ] * [ R (Fe) / R (Cd)]^3
n(Cd) / n(Fe)= (112 / 56 ) * (1/1.26)3
n(Cd) / n(Fe)=1
In conclusion, The ratio of n(Cd) = n(Fe) is 1, hence same
Read more about density
https://brainly.com/question/14010194
Compared to the nucleus ⁵⁶₂₆Fe, the density of the nucleus ¹¹²₄₈Cd is same.
How do we calculate the density of nucleus?Density of any nucleus will be calculated by using the below equation as:
ρ = m/V, where
m = mass of the atom
V = volume = 4/3πR³
For the given question calculation will be proceed as:
n(Cd) / n(Fe) = [m(Cd)/m(Fe)].[R(Fe)/R(Cd)]³
From the given formulas of iron and cadmuim we put values on the above equation then we get,
n(Cd) / n(Fe) = (112 / 56 ) × (1/1.26)3
n(Cd) / n(Fe) = 1
Hence the density of nucleus ¹¹²₄₈Cd is same as of nucleus ⁵⁶₂₆Fe nucleus.
To know more about nucleus density, visit the below link:
https://brainly.com/question/5722114
#SPJ4
Can you help plzzz thank you
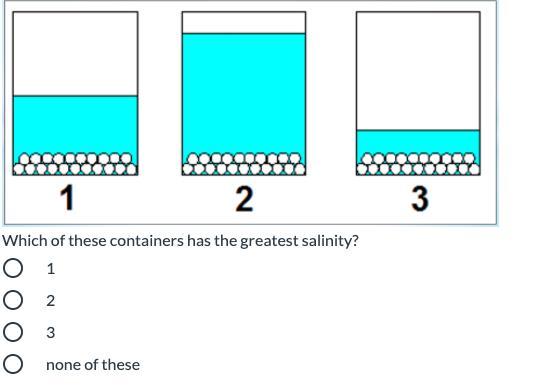
Answers
Answer:
It would be Container 3
Explanation:
Each of the containers has the same amount of salt. Salinity refers to salt level. Since the question is asking for the container with the greatest salinity, you are looking for the container with the least water (because it will be the saltiest out of all of them). Container 3 has the least water.
Hope this helps :)
3 has less water so its saltier
why does temperature affect voltage in a galvanic cell
Answers
It was predicted based on the Nernst equation experiment that the temperature would rise while the voltage generated by the electrolytic cell would drop.
What can you infer from the Nernst equation?The relationship between the ion concentrations on each side of a membrane that is completely selective for just that ion and the dc voltage (voltage) that will be recorded across that membranes under adaptation options is defined by this equation.
Simply put, what does the Nernst equation mean?The reduction potential of an atom or ion, as measured at any circumstances, may be equated with the reduction potential as measured under realistic conditions of 298K but one molar or one air pressure using the Nernst equation.
To know more about Nernst visit:
https://brainly.com/question/22724431
#SPJ4
Write complete reaction mechanisms for formation of 1-bromobutane and also show how 1-butene and di-n-butyl ether are formed. 2. Why is it necessary to remove water before weighing the 1-bromobutane
Answers
The formation of 1-bromobutane involves the following reaction mechanism: 1. Nucleophilic substitution (SN2): Butanol reacts with a bromide ion (Br-) to form 1-bromobutane and a hydroxide ion (OH-). Butanol + Br- → 1-bromobutane + OH-
The formation of 1-butene and di-n-butyl ether are side reactions in this process:
1. Elimination (E2) reaction: Butanol undergoes an elimination reaction to form 1-butene and water.
Butanol → 1-butene + H2O
2. Williamson ether synthesis: Two butanol molecules react with each other in the presence of a base, forming di-n-butyl ether and water.
2 Butanol + Base → di-n-butyl ether + H2O + Base
It's necessary to remove water before weighing the 1-bromobutane because water could alter the measured weight, leading to inaccurate results. Water can also promote side reactions like the formation of di-n-butyl ether, which could further impact the yield and purity of the desired product, 1-bromobutane.
More on Nucleophilic substitution: https://brainly.com/question/28787378
#SPJ11
Calculate the molality of 12 percent urea solution
Answers
Molality of a solution is defined as the number of moles of solute per kilogram of solvent. The formula for molality is given by the following equation:molality (m) = moles of solute / mass of solvent (in kg)In order to calculate the molality of a 12 percent urea solution, we need to first determine the mass of urea present in the solution.
We know that a 12 percent urea solution means that 12 grams of urea is present in 100 grams of the solution. Therefore, we can calculate the mass of urea in the solution as follows:Mass of urea = (12/100) x 200 g= 24 gNext, we need to convert this mass into moles. The molar mass of urea is 60.06 g/mol. Therefore, the number of moles of urea present in the solution can be calculated as follows:Number of moles of urea = Mass of urea / Molar mass of urea= 24 g / 60.06 g/mol= 0.3996 molFinally, we can use the formula for molality to calculate the molality of the solution as follows:molality (m) = moles of solute / mass of solvent (in kg)= 0.3996 mol / 0.2 kg= 1.998 mol/kgTherefore, the molality of the 12 percent urea solution is approximately 1.998 mol/kg.For such more question on moles
https://brainly.com/question/29367909
#SPJ8
If 5.9 × 10²⁵ molecules of CO₂ are produced in a combustion reaction, how many liters of CO₂ is produced? (The density of CO₂ is 1.98 kg/m³ and 1 L = 1 dm³).
Answers
Answer:
2.18 × 10³L
Explanation:

2178 litres of CO2 is produced in the combustion reaction.
HOW TO CALCULATE VOLUME
The volume of a substance can be calculated using the formula below:Density = mass ÷ volume
Firstly, we must calculate the number of moles present in 5.9 × 10²⁵ molecules by dividing by Avogadro number:no of moles. = 5.9 × 10²⁵ ÷ 6.02 × 10²³
no. of moles = 5.9/6.02 × 10²
no. of moles = 0.98 × 10²moles
= 98moles
Moles = mass ÷ molar mass
Mass of CO2 = moles × molar mass
Molar mass of CO2 = 12 + 16(2) = 44g/mol
Mass of CO2 = 98 × 44
Mass of CO2 = 4312g = 4.312kg
The volume of CO2 can be calculated as follows:Volume = 4.312kg ÷ 1.98kg/m³
Volume = 2.178m³
Since 1m³ = 1000L
2.178m³ = 2.178 × 1000
= 2178 liters.
Hence, 2178 litres of CO2 is produced in the combustion reaction.Learn more: https://brainly.com/question/15164682?referrer=searchResults
A customer is traveling to a branch office static ip.
Answers
Answer:
A customer is travelling to a branch office, and the network administrator provides her with a static IP address for her laptop
Explanation:
I hope this helps!!!...
Brainliest please???
Which is the smallest particle into which a compound can be broken down
and still remain the same compound?
A) atom
B) mixture
C) element
D) molecule
Answers
Answer:
molcule
Explanation:
Determine the ratio of moles of iron actually used to moles of new product actually formed. Does this molar ratio agree with the ratio from the balanced chemical equation?
equation 1.50g Cu 1 mole cu ÷ 63.5g Cu Fe ÷1 Cu 55.8gFe ÷ 1 mole = 1.32g Fe

Answers
The mole ratio of iron to copper is 1 : 1, thus, 1.31 g of iron is required to produce 1.5 g of copper.
What is the equation of the reaction between iron and copper(ii) sulfate?The reaction between iron and copper(ii) sulfate is a redox displacement reactions in which copper is displaced from its salt by iron.
The equation of the reaction is given below:
\(CuSO_4 + Fe \rightarrow FeSO_4 + Cu\)
The mole ratio of both reactants and product is 1 : 1
moles = mass/molar massmolar mass of copper sulfate = 160 g
molar mass of Cu = 64
molar mass of iron = 56 g
mole ratio of iron to copper = 1 : 1moles of Cu in 1.5 g = 1.5/64 = 0.00234
mass of iron required = 0.00234 × 56 = 1.31 g of iron
Therefore, from the mole ratio of iron to copper, 1.31 g of iron is required to produce 1.5 g of copper.
Learn more about mole ratio at: https://brainly.com/question/19099163
What are 1A, 3B, and 7A examples of on the periodic table?
groups.
numbers
periods
O rows
Answers
Answer: Groups
Explanation:
I took the quiz
A weather balloon was initially at a pressure of 0.900 atm, and its volume was 35.0 L. The pressure decreased to 0.750 atm, without loss of gas or change in temperature. What was the change in the volume?
Answers
Boyle's Law
P₁V₁=P₂V₂
0.9 x 35 = 0.75 x V₂
V₂ = 42 L
Buckets of paint can be classified as what type of mixture?
Answers
Answer:
Homogeneous
Explanation:
Homogeneous mixtures are uniform in composition. They have the same proportion of components throughout. Homogeneous mixtures are called solutions. Sugar, paint, alcohol, gold are all examples of homogeneous mixtures because they look the same throughout.
what is the amount of molecules in 5 moles of nitrogen gas, N2?
Answers
The amount of molecules= 3.01 x 10²⁴
Further explanationGiven
5 moles of N2
Required
The number of molecules
Solution
The mole is the number of particles(molecules, atoms, ions) contained in a substance
1 mol = 6.02.10²³ particles
Can be formulated
N=n x No
N = number of particles
n = mol
No = Avogadro's = 6.02.10²³
For 5 moles :
= 5 x 6.02 x 10²³
= 3.01 x 10²⁴
The blood must keep moving round the body to keep the rate of diffusion high explains why
Answers
Answer:
red blood cells and diffusion.
Explanation:
this process involves red blood cells and diffusion. Red Blood cells (RBCs) are a type of blood cell whose role is to transport oxygen from the lungs to the peripheral tissues of the body. Oxygen diffuses from the alveoli of the lungs in to the RBC's, where it binds to hemoglobin.
Diffusion is a very important process within the human body and is essential to the transport of molecules within a number of organs including the lungs, kidneys, stomach and eyes. The lungs process millions of tiny air sacs that are called alveoli, each of which are in close contact with capillaries.
Rx Ephedrine sulfate (fz. pt = -0.13°C). 2%
Sodium chloride MW 58.5
Purified water qs ad. 30 mL
How much sodium chloride should be used to make this eye
solution isotonic with tears?
the answer is 22
Answers
The correct answer is the amount of sodium chloride needed to make this eye solution isotonic with tears is approximately 1.85 grams. Rounding it up to the nearest whole number gives us the answer as 2. Hence, the correct option is 22.
The given solution is a hypotonic solution as the solution's tonicity is lower than that of the tears. The tears contain 0.9% w/v of NaCl, which is isotonic with tears. So, to make the given solution isotonic, the amount of sodium chloride needs to be added.
The concentration of NaCl in tears is 0.9% w/v. Additional Information: We know that % w/v is the amount of solute present in grams per 100 ml of the solution. Therefore, 0.9% w/v means 0.9 grams of NaCl is present in 100 mL of tears.
To make 30 ml of isotonic solution, we can use the following formula: Equivalent weight of NaCl = 58.5/2 = 29.25 (as NaCl ionizes to give Na+ and Cl- ions)Moles of NaCl required to make 30 ml isotonic solution = 0.9 × 30 / 1000 = 0.027Moles of Na+ and Cl- ions present in 30 mL of isotonic solution = 2 × 0.027 = 0.054
A number of grams of NaCl needed to prepare 30 mL of isotonic solution is calculated as follows:0.054 g = (0.027 x 29.25 x X) / 1000Where X is the amount of NaCl required to make 30 mL isotonic solution. Solving this equation gives us: X = 1.85 g (approx). Therefore, the amount of sodium chloride needed to make this eye solution isotonic with tears is approximately 1.85 grams. Rounding it up to the nearest whole number gives us the answer as 2. Hence, the correct option is 22.
know more about hypotonic solution
https://brainly.com/question/122954
#SPJ11
In an ionic bond, how do you tell the number of electrons transferred from atom to atom. Would it be two ions formed in the reaction or no?
Example, Lithium + Chlorine
Answers
Answer:
Explanation:
In an ionic bond, atoms transfer electrons in order to achieve a stable electron configuration, also known as a full valence shell. The number of electrons transferred from one atom to another can be determined by the difference in their electron configurations.
In the example of lithium and chlorine forming an ionic bond, lithium (Li) has one valence electron and chlorine (Cl) has seven valence electrons. In order for both atoms to achieve a stable electron configuration, Li would transfer its one valence electron to Cl. This results in the formation of two ions: Li+ and Cl-.
It is important to note that for a reaction to be ionic, it must involve the transfer of electrons from one atom to another. In the case of Lithium and Chlorine it is ionic because electrons are transferred from Li to Cl.
The Kelvin temperature of sample of 650 cm sample of ammonia gas is doubled what is the new volume of the gas
Answers
Answer:
1300cm3.
Explanation:
Step 1:
Data obtained from the question.
Initial temperature (T1) = K
Initial volume (V1) = 650 cm3
Final temperature (T2) = double the original = 2K
Final volume (V2) =..?
Step 2:
Determination of the new volume of the gas.
The new volume of the gas can be obtained by using Charles' law as follow:
V1/T1 = V2/T2
650/K = V2/2K
Cross multiply
K x V2 = 650 x 2K
Divide both side by K
V2 = 650 x 2K /K
V2 = 650 x 2
V2 = 1300cm3.
Therefore, the new volume of the gas is 1300cm3
The new volume of the gas is 1300cm³.
Calculation for volume:Given:
Initial temperature (T₁) = K
Initial volume (V₁) = 650 cm3
Final temperature (T₂) = double the original = 2K
To find:
Final volume (V₂) =?
Determination of new volume:
V₁/T₁ = V₂/T₂
650/K = V₂/2K
K x V₂ = 650 x 2K
V₂ = 650 x 2K /K
V₂ = 650 x 2
V₂ = 1300cm³
Therefore, the new volume of the gas is 1300cm³.
Find more information about Volume here:
brainly.com/question/25736513
What is the balanced reduction half-reaction for the unbalanced oxidation-reduction reaction? Na(s) + Cl2lo) - NaCl(s) 1. Cla) + 2 - 2 C1"(s) 2. Cl2(g) 2 + 2 C1-(s) 3. Na(s) + +-Nat(s) 4. Na(s) - Na'(s) + 2 O 1
Answers
The balanced equation shows that two sodium atoms react with one chlorine molecule to form two molecules of sodium chloride.
The balanced reduction half-reaction for the unbalanced oxidation-reduction reaction Na(s) + Cl2(g) → NaCl(s) can be found by identifying the species being reduced. In this case, it is the chlorine molecule (Cl2) that is being reduced to form chloride ions (Cl-). The reduction half-reaction for this process can be written as follows:
Cl2(g) + 2e- → 2Cl-(aq)
This equation represents the balanced reduction half-reaction for the given oxidation-reduction reaction. To balance the full reaction, we need to combine it with the oxidation half-reaction, which represents the oxidation of sodium atoms (Na) to form sodium ions (Na+). The oxidation half-reaction can be written as:
Na(s) → Na+(aq) + e-
By combining the two half-reactions, we get the balanced oxidation-reduction reaction:
2Na(s) + Cl2(g) → 2NaCl(s)
This reaction represents the balanced reduction half-reaction and oxidation half-reaction combined. The reduction half-reaction involves the gain of electrons by chlorine atoms, while the oxidation half-reaction involves the loss of electrons by sodium atoms. The balanced equation shows that two sodium atoms react with one chlorine molecule to form two molecules of sodium chloride.
learn more about atoms
https://brainly.com/question/1566330
#SPJ11
Which of the following is an ionic compound?
(A) CO₂
B) NH3
C) KBr
D) C12

Answers
Answer:
co2
Explanation:
it is an ionic compound
Nitrogen gas becomes converted into biologically usable chemical forms by what type of organism?.
Answers
Nitrogen gas becomes converted into biologically usable chemical forms by specific bacteria called aerobic bacteria.
In atmosphere, nitrogen gas is present in gaseous form while in soil nitrogen in not present in natural form.
There are specific oxygen consuming or aerobic bacteria also called 'nitrogen fixing bacteria' which use atmospheric nitrogen and transform it into biologically usable chemical compounds such as ammonia.
Later, fixed ammonia is used by plants as rich nitrogen source.
To put it simply, nitrogen cycle is a process during which atmospheric nitrogen moves through environment to the living things in the form ammonia.
If you need to learn more about nitrogen gas click here:
https://brainly.com/question/25669014
#SPJ4
Cellular respiration occur within ______ of a cell.
Answers
Answer:
The mitochondria
Explanation:
Its where cellular respiration occurs
Balance the following equations.
a. _CH4 + _O2 → _CO2 + _H2O
b. _Zn + _HCl → _ZnCl2 + _H2
c. _H2SO4 + _HI → _H2S + _I2 + _H2O
d. _NaCl + _SO2 + _H2O + _O2 → _Na2SO4 + _HCl
Answers
Answer:
a. CH4+202→CO2+2H2O
b.Zn+2HCl→ZnCl2+H2
c.H2SO4+8HI→H2S+4I2+4H2O
d.4NaCl+2SO2+2H2O+O2→2Na2SO4+4HCl
The organelle that move proteins and other substances through the cell is the
Answers
Answer:
Endoplasmic reticulum
Explanation:
what is the predominant alloying element in all of the stainless steels
Answers
The predominant alloying element in all of the stainless steels is chromium(Cr).
Why Chromium element is the predominant?Because the addition of chromium gives stainless steels their corrosion-resistant properties. Typically, stainless steels contain at least 10.5% chromium by mass, although some high-performance stainless steels may contain up to 26% chromium. The chromium reacts with oxygen in the air to form a thin, invisible layer of chromium oxide on the surface of the steel, which protects it from corrosion.
Other alloying elements, such as nickel, molybdenum, and nitrogen, may also be added to stainless steels to enhance their properties.
Learn more about stainless steels here https://brainly.com/question/28448322
#SPJ11
What are examples of the solar system
Answers
Answer:(:
Explanation:
An example of the solar system is the eight planets including Earth that revolve around the sun. The sun together with the eight planets and all other celestial bodies that orbit the sun. A system of planets or other bodies orbiting a star. That portion of our galaxy which is subject to the gravity of the sun
Which of the following correctly orders the types of radiation from the LONGEST wavelength to the SHORTEST wavelength?
A. Green Visible Light, Red Visible Light, Blue Visible Light, Ultraviolet
B. Microwave, Orange Visible Light, Ultraviolet, Violet Visible Light
C. Red Visible Light, Infrared, Microwaves, Radio waves
D. Microwave, Blue Visible Light, Ultraviolet, Gamma
Answers
Answer:
For the wavelenth of light spectrum, I believe the order would be red visible light, infrared, microwaves and radio waves.
if 44.5 l of nitrogen at 848 mm hg are compressed to 976 mm hg at constant temperature. what is the new volume?
Answers
Answer:
....................
Explanation:
.....................
After nitrogen compression from 848 mm Hg to 976 mm Hg at a constant temperature, the new volume is approximately 38.2 L.
What is the new volume of nitrogen?Hi! To find the new volume of nitrogen when 44.5 L at 848 mm Hg is compressed to 976 mm Hg at a constant temperature, you can use Boyle's Law, which states that the product of the initial pressure and volume (P1V1) is equal to the product of the final pressure and volume (P2V2).
Given:
Initial volume (V1) = 44.5 L
Initial pressure (P1) = 848 mm Hg
Final pressure (P2) = 976 mm Hg
Boyle's Law formula:
P1V1 = P2V2
Step 1: Plug the given values into the formula:
(848 mm Hg)(44.5 L) = (976 mm Hg)(V2)
Step 2: Solve for the final volume (V2):
V2 = (848 mm Hg)(44.5 L) / (976 mm Hg)
V2 ≈ 38.2 L
So, when the nitrogen is compressed from 848 mm Hg to 976 mm Hg at constant temperature, the new volume is approximately 38.2 L.
Learn more about nitrogen compression
brainly.com/question/15483405
#SPJ11
Recently, the number of elks has increased significantly in a forest. These elks often enter the nearby grassland and eat up all the grass. This is indirectly affecting the livestock industry in that area. Which method might solve the nuisance caused by these excess elks in the fores
Answers
The method might solve the nuisance caused by these excess elks in the forest is research. Therefore, option C is correct.
What is forest ?A forest is a land area dominated by trees. Hundreds of definitions of forest are used around the world, with factors such as tree density, tree height, land use, legal standing, and ecological function all being considered.
The word forest comes from the Old French forest (also forès), which means "forest, vast expanse covered by trees"; forest was first used in English to describe wild land set aside for hunting that did not necessarily have trees on it.
A forest is a complex ecological system in which trees predominate. A forest is nature's most efficient ecosystem, with a high rate of photosynthesis that affects both plant and animal systems in a complex web of organic relationships.
Thus, option C is correct.
To learn more about the forest, follow the link;
https://brainly.com/question/22279329
#SPJ1
Your question is incomplete, probably your question was
Recently, the number of elks has increased significantly in a forest. These elks often enter the nearby grassland and eat up all the grass. This is indirectly affecting the livestock industry in that area. Which method might solve the nuisance caused by these excess elks in the forest?
A- establishing WMA
B- hunting
C- research
D-habitat manipulation
4. Where is the magnitude of force around this object the greatest?
C
A.The magnitude is greatest close to the poles.
B.The magnitude is greatest far from the poles.
C. The magnitude is equal at all points on the field.
D. The magnitude is greatest halfway between poles.
Answers
\(\mathfrak{\huge{\orange{\underline{\underline{AnSwEr:-}}}}}\)
Actually Welcome to the Concept of the Magnetism.
Since any magnetic object like true magnets, permanent magnets and solenoids always have a magnetic field lines around them that , originate from North Pole and Drive to South Pole, hence,
The most intensive and strong magnetic field are close to the poles.
A. is the correct answer.
===> A. The magnitude is greatest close to the poles.
If the centers of two objects are close to each other then greater will be the magnitude of the force of gravity. Hence the gravitational force will be greater in poles than the equator. The correct option is A.
What is gravitational force?The gravitational force is regarded as the universal force which attracts all the physical forms of the earth having mass. It is considered as an attractive force exerted on all objects.
The expression for gravitational force is:
F = G m₁m₂ / r²
The gravitational force is directly proportional to the masses of the objects and inversely proportional to the distance between them.
The gravitational force is maximum at poles than the equator. It is because the poles are found to be closer to the earth than the equator due to the oblateness of earth.
The distance of the equator from the center of the earth is found to be greater than that of the poles. Hence the maximum force is at poles.
Thus the correct option is A.
To know more about force, visit;
https://brainly.com/question/13014979
#SPJ6