Answers
Answer:
0.273 Molarity
Explanation:
Super simple, since Molarity = moles of solute/L of solute just take .761 moles/2.78 L = .273 molarity
Related Questions
Two asteroids are 75,000 m apart one has a mass of 8 x 10^7 N what is the mass of the other asteroid
Answers
The mass of the asteroid is C. 1.2 x \(10^{12}\) Kg
To find the mass of the other asteroid, we can rearrange the equation for the gravitational force between two objects:
F = (G * m1 * m2) / \(r^{2}\)
where F is the force of gravity, G is the gravitational constant, m1 and m2 are the masses of the two asteroids, and r is the distance between them.
Given that the distance between the asteroids is 75000 m, the force of gravity between them is 1.14 N, and one asteroid has a mass of 8 x \(10^{7}\) kg, we can substitute these values into the equation and solve for the mass of the other asteroid (m2):
1.14 N = (6.67430 × \(10^{-11}\) N \(m^{2}\)/\(Kg^{2}\) * 8 x \(10^{7}\) kg * \(m2\)) / \((75000 m)^{2}\)
Simplifying and solving the equation, we find that the mass of the other asteroid (m2) is approximately 1.2 x \(10^{12}\) kg. Therefore, Option C is correct.
The question was incomplete. find the full content below:
Two asteroids are 75000 m apart one has a mass of 8 x \(10^{7}\) kg if the force of gravity between them is 1.14 what is the mass of the asteroid
A. 3.4 x \(10^{11}\) kg
B. 8.3 x \(10^{12}\) kg
C. 1.2 x \(10^{12}\) kg
D. 1.2 x \(10^{10}\) kg
Know more about gravitational force here:
https://brainly.com/question/72250
#SPJ8
Based on context clues what is the meaning of tête-à-tête
Answers
Answer: A private conversation between two people.
Explanation: Hope this Helps.
300×175
Express your answer using the correct number of significant figures.
Answers
Answer:
300×175=52500
Explanation:
caculator
heating curve iron
at what temperature does the substance begins to boil
at what temperature does a substance begin to melt
at what temperature is a substance for a liquid and a gas
at what temperature is the substance both a solid and a liquid

Answers
The substance begins to boil at 2750⁰C, the substance begins to melt at 1500⁰C, the temperature at which the substance is both a liquid and a gas at 2750⁰C, temperature is the substance both a solid and a liquid at 1500⁰C.
Heating curves are the graphical correlations between heat added to a substance. When viewed from a cooling perspective, ie. loss of heat, it is the cooling curve.
The gradient of the cooling curve is related to the heat capacity, the thermal conductivity of the substance, and the external temperature. The more heat is required to change the temperature of the substance, the slower it cools, so the smaller the gradient of the curve. The higher the thermal conductivity, the faster heat is transferred, so the faster the substance cools.
Learn more about Heating curve, here:
https://brainly.com/question/29592874
#SPJ1
If the half-life of 14C is 5730 years, there will only be ½ of a given sample left after 11,460 years.
Answers
After 11,460 years (which is exactly two half-lives of 14C), only 1/4 of the initial amount of 14C will remain, since half of the remaining 14C will have decayed twice.
What is Half Life?
Half-life is a term used to describe the time it takes for half of the atoms in a radioactive substance to decay. It is a characteristic property of each radioactive isotope, and it is used to measure the rate of decay of the isotope.
The process of radioactive decay involves the spontaneous transformation of an unstable atom into a more stable atom by emitting radiation in the form of particles or electromagnetic waves. The rate of decay of a radioactive substance is proportional to the number of atoms present, and it can be described mathematically using an exponential decay equation.
The half-life of a radioactive substance is the time it takes for half of the initial amount of the substance to decay. In the case of 14C, its half-life is approximately 5730 years. This means that after 5730 years, half of the 14C in a given sample will have decayed, and after another 5730 years, half of the remaining 14C will have decayed, and so on.
Therefore, after 11,460 years (which is exactly two half-lives of 14C), only 1/4 of the initial amount of 14C will remain, since half of the remaining 14C will have decayed twice. This means that there will only be 1/2 of a given sample left after 5730 years, and 1/4 of a given sample left after 11,460 years.
Learn more about Half Life from given link
https://brainly.com/question/1160651
#SPJ1
if 100s of heat is added to a system and 0.100ks of work is done by the system, what is the value of DE
Answers
If 100 s of heat is added to a system and 0.100 ks of work is done by the system, then the value of ΔE or DE (change in internal energy) is 200 J, which is in option A. This is as per the first rule of thermodynamics.
ΔE = Q - W
ΔE (change in internal energy)
Given that 100 J (joules) of heat is added to the system (Q = 100 J) and 0.100 kJ (kilojoules) of work is done by the system (W = -0.100 kJ), we need to ensure that the units are consistent.
Since 1 kJ = 1000 J, we can convert the work done to joules:
W = -0.100 kJ = -0.100 × 1000 J = -100 J
Now one can substitute the values into the equation:
ΔE = Q - W
= 100 J - (-100 J)
= 100 J + 100 J
= 200 J
Therefore, the value of ΔE (change in internal energy) is 200 J.
Learn more about energy here
https://brainly.com/question/12479265
#SPJ1
complete question is below
if 100s of heat is added to a system and 0.100ks of work is done by the system, what is the value of DE
A. 200 j
B.50 j
C.2 j
D. 10000j
Which bonds form in the reaction shown in the diagram? 2 2H, + 0 H-H O=0 H-H → 2H 0 H-0-H H-OH A. The bonds between the two hydrogen atoms and between the two oxygen atoms B. The bonds between the two hydrogen atoms C. The bonds between the oxygen and hydrogen atoms D. The bonds between the two oxygen atoms

Answers
The water molecule is formed by the covalent bonding between oxygen and hydrogen atoms. Therefore, option C is correct.
What is covalent bonding ?Covalent bonding is a type of chemical bonding that involves the sharing of electrons between two atoms. In covalent bonding, the two atoms share a pair of electrons to fill their outermost electron shell and form a stable molecule.
This type of bonding usually occurs between non-metal atoms, which have a high electronegativity and tend to attract electrons strongly. In a covalent bond, the shared electrons are attracted to the positively charged nuclei of both atoms, creating a strong bond.
The strength of the bond depends on the number of shared electrons and the distance between the nuclei. Covalent bonds can be either polar or nonpolar, depending on the electronegativity difference between the two atoms.
In water the bond is formed between oxygen atom and two hydrogen atoms. Hence, C is correct.
Find more on covalent bonding:
https://brainly.com/question/12661797
#SPJ7
Use the standard reduction potentials located in the 'Tables' linked above to calculate the standard free energy change in kJ for the reaction: Zn2+(aq) + 2Cu+(aq) ----> Zn(s) + 2Cu2+(aq) K for this reaction would be ____ than one
a. greates
b. less
Answers
Option (a) is correct. The standard free energy change in KJ for the reaction, 2Cu+(aq.) ----> Zn(s) + 2Cu2+(aq.) is greater then one.
The standard cell potential, E∘ cell and the standard Gibbs free energy, ΔG∘ are related to each other. The standard cell potential is the potential difference between the cathode and anode. The standard Gibbs free energy of formation of a compound is the change of Gibbs free energy that accompanies the formation of 1 mole of a substance in its standard state from its constituent elements in their standard states. It is expressed by the equation,
ΔG∘ = nFE∘cell
where n is the number of transferred electrons
F is the Faraday's constant.
Since a spontaneous reaction has a negative ΔG∘, a spontaneous redox reaction must have a positive ΔE∘cell. The opposite is true for non-spontaneous reactions.
To learn more about Standard Reduction potential please visit:
https://brainly.com/question/4135123
#SPJ4
Complete the paragraph to describe precipitation reactions.
Chemical precipitation occurs when a product is
and precipitates out of a solution. By comparison, precipitation reactions require the reactants to be
in the solvent.
Answers
Chemical precipitation occurs when a product is insoluble and precipitates out of a solution. By comparison, precipitation reactions require the reactants to be soluble in the solvent.
What is Chemical precipitation ?Chemical precipitation is a type of chemical reaction in which a solid substance is formed from the reaction of two or more dissolved chemicals. In a chemical precipitation reaction, ions from the reactant solutions combine to form a new compound that is insoluble in water, causing it to "precipitate" or settle out of the solution as a solid. Chemical precipitation reactions are often used in the laboratory to purify and isolate compounds, as the solid precipitate can be easily separated from the liquid solution using techniques such as filtration or centrifugation. The formation of a solid precipitate is typically indicated in a chemical equation by the presence of a (s) notation after the product.To learn more about Chemical precipitation refer :
https://brainly.com/question/11194650
#SPJ1
Answer: 1.) insoluble 2.) soluble
Explanation: EDMENTUM
Tom lost money on his peach crops because they kept freezing in the winter. How could Tom most likely solve the problem of his peach plants dying over the winter?
A.
He could breed his peach plants with peach plants that grow slowly.
B.
He could breed his peach plants with peach plants that can survive the frost.
C.
He could breed his peach plants with peach plants that are very small.
D.
He could breed his peach plants with peach plants that need more sunlight.
Answers
Answer:
B. He could breed his peach plants with peach plants that can survive the frost.
Explanation:
Tom can best solve his problem by breeding his peach plants with the ones that can survive the frost.
This is known cross breedingThe goal of cross breeding is to produce offspring that contains the traits of both of the parents or an improved hybrid. If Tom can cross breed the failing peaches with the successful winter resistant ones, he can ensure better production. The new hybrid will therefore will be genetically superior and adapted to the prevailing environmental changes.what would the result have been if there were five exons and four introns? sketch what this would look like an electron micrograph
Answers
If there had been five exons and four introns, the result would have been. The four introns will be forced to loop in order to bring out the adjacent exons together.
An exon is any part of a gene that will be included in the final mature RNA produced by that gene after introns are removed via RNA splicing. Exons refer to both the DNA sequence within a gene and the corresponding RNA transcript sequence. In RNA splicing, introns are removed and exons are covalently joined to form mature RNA. The genome is made up of the entire set of genes for a species, and the exome is made up of the entire set of exons."The concept of the cistron must be replaced by that of a transcription unit," wrote American biochemist Walter Gilbert in 1978.
Learn more about exon here:
https://brainly.com/question/26464408
#SPJ4

the pressure on 20 milliliters of a gas at constant temperature is changed from 4 atmospheres to 2 atmospheres. what is the new volume of the gas?
Answers
The new volume of the gas whose pressure was changed would be = 40 milliliters.
How to calculate the new volume of the given gas?The initial volume(V1)of the gas= 20ml
The initial pressure(P1) = 4 atm
The final pressure(P2) = 2 atm
The final volume(V2) = ?
Using the general gas formula;
P1V1 = P2V2
V2 = P1V1/P2
= 4×20/2
= 40ml
Learn more about volume here:
https://brainly.com/question/27710307
#SPJ1
Which shows an isomer of the molecule below?

Answers
Answer:
The answer is B, in this case.
Explanation:
An isomer is a molecule with the same number of atoms as another compound, but they differ in arrangement of the atoms.
The table below shows the dimensions of two colored cubes.
Dimensions of Cubes
Cube Side (cm) Mass (g)
Red 4 12
Green 3 10
Which cube is denser?
Red, because it has more volume and more amount of matter.
Green, because it has less volume and less amount of matter.
Red, because it has less volume and more amount of matter.
Green, because it has less volume and more amount of matter.
Answers
Answer:
Red, because it has less volume and more amount of matter.
Explanation:
the answer is "Green, because it has less volume and less amount of matter."
A cake recipe calls for 170.0 mL of buttermilk. How many cups is this?
(1.0567 quart = 1
L and 4 cups =
1
quart

Answers
A cup is 240 ml and the cake recipe calls for 170.0 ml of milk. It's 2/4 of a cup.
How many mls are in a cup of tea?
the cups have a standard measure of 240ml, that is, if you buy one of these good quality utensils, even if in different places, the measure of 1 cup must always be equivalent to 240ml.
With this information, we can conclude that Ml is the abbreviation milliliter, the smallest unit of measurement for liquids. We start with the liter as the base unit, we must go down to one tenth of a liter, which is deciliter, then 100 tenths of a liter than the centiliter and we go up to one thousand tenths of a liter, which is milliliter. One liter contains one thousand milliliters.
Learn more about milliliter in brainly.com/question/8513068
#SPJ1
please help me i need this asp

Answers
22 Agas that is collected by upward delivery is likely to be A heavier than air B insoluble in water C lighter than air D Soluble in water
Answers
Answer:
joib
Explanation:
chemistry
Definition in your own words. I will check if you got it from online.
Word: Melting Point
Answers
Answer:
The specification of the query in the discussion is characterized below.
Explanation:
The given term would be the temperature through which the solid, as well as the liquid sequence, could sometimes exist side by side throughout equilibration as well as the atmospheric pressure beyond which the matter keeps changing from a solid-state to the liquid one. The concept shall apply to the liquid phase as well as alternatives. The melting point changes depending on either the pressure, therefore it needs to be clearly stated.Please help answering 46)

Answers
Answer:
CO2 is a trigonal planar.
9. When 1.10 g of magnesium reacted with unlimited amount of HCl, the products were
hydrogen gas and magnesium chloride. What volume of hydrogen gas would be collected
if the reaction had been run at STP?
Answers
As per the balanced equation of the reaction one mole of Mg give s one mole of H2. 1.10 g of magnesium is 0.045 moles. Thus it gives 0.045 moles of H2 which is 1.02 litres.
What is magnesium chloride?Magnesium chloride is an ionic compound formed by the reaction of magnesium metal with two moles of hydrochloric acid. The reaction also produce one mole hydrogen gas.
The atomic mass of magnesium is 24 g/mol. Thus number of moles in 1.10 g is 1.10/24 = 0.045 moles.
As per the reaction equation one mole of magnesium gives one mole of hydrogen gas. Thus 0.045 moles of magnesium gives 0.045 moles of hydrogen gas.
At STP, the volume of one mole of any gas will be equal to 22.4 L. Thus volume of 0.045 moles of hydrogen at STP is:
volume = 0.045 × 22.4
=1.02 L.
Hence, the volume of hydrogen produce here is 1.02 L.
To find more on magnesium, refer here:
https://brainly.com/question/1533548
#SPJ1
NO LINKS/ please round two decimal places !!

Answers
3. Which is larger, 5 kg or 500 g?* Easy way to remember the order of prefixes: Kids Have Dreams But Dreams Cost Money 5 kg O 5009 They are equal
what is the answers
Answers
Answer:
your mom
Explanation:
How do you know if the girl your dating is into the size or onto personality? 100 POINTS
Answers
Answer:
here
Explanation:
My guess is if she's into you for your size she's gonna want to have s## alot, but if she's into your personality she wants to hang out with you and compliments you this is my guess so sorry if this isnt good
How many atoms or molecules are in 10 grams of table salt?
Answers
Answer:
1.03 x 10²³ atoms NaCl
Explanation:
To find the amount of table salt (NaCl) in atoms, you need to (1) convert grams to moles (using the molar mass) and then (2) convert moles to atoms (using Avogadro's Number). It is important to arrange the ratios/conversions in a way that allows for the cancellation of units.
(Step 1)
Molar Mass (NaCl): 22.99 g/mol + 35.45 g/mol
Molar Mass (NaCl): 58.44 g/mol
10 grams NaCl 1 mole
------------------------ x ----------------------- = 0.17 moles NaCl
58.44 grams
(Step 2)
Avogadro's Number:
6.022 x 10²³ atoms = 1 mole
0.17 moles NaCl 6.022 x 10²³ atoms
-------------------------- x -------------------------------- = 1.03 x 10²³ atoms NaCl
1 mole
The substance that is dissolved in another substance to create
a solution is called the
Answers
Answer:
The substance that is dissolved in another substance to create a solution is a Solute. A Solute is a minor component in a solution which is a substance that is being dissolved, while the solvent is the dissolving medium. Solutions can be formed with many different types and forms of solutes and solvents.
Using the information in the table to the right, calculate the average atomic mass of strontium. Report to two decimal places.
A 3-column table with 4 rows titled Strontium. Column 1 is labeled Isotope with entries upper S 4 84, upper S r 86, upper S r 87, upper S r 88. Column 2 is labeled Mass in atomic mass units with entries 83.913428, 85.909273, 86.908902, 87.905625. Column 3 is labeled abundance with entries 0.56 percent, 9.86 percent, 7.00 percent, 82.58 percent.
Answers
The column 1 has the value of Isotope, column 2 has the value of mass in atomic mass units, and column 3 has the value of abundance and the average atomic mass of strontium is 87.47 amu.
To calculate the average atomic mass of strontium using the given information, we need to multiply the mass of each isotope by its abundance and then sum up these values. Here's the calculation:
Isotope | Mass (amu) | Abundance
^84Sr | 83.913428 | 0.56%
^86Sr | 85.909273 | 9.86%
^87Sr | 86.908902 | 7.00%
^88Sr | 87.905625 | 82.58%
To find the average atomic mass, we multiply each isotope's mass by its abundance (in decimal form) and sum up the values:
Average atomic mass = (\(Mass of ^{84Sr}\) × \(Abundance of^{84Sr}\)) + (\(Mass of ^{86Sr}\)× \(Abundance of^{86Sr}\)) + (\(Mass of ^{87Sr}\) × \(Abundance of^{87Sr}\)) + (\(Mass of ^{88Sr}\) × \(Abundance of^{88Sr}\))
Average atomic mass = (83.913428 amu × 0.0056) + (85.909273 amu × 0.0986) + (86.908902 amu × 0.0700) + (87.905625 amu × 0.8258)
Calculating this expression yields:
Average atomic mass = 0.469901638 + 8.468098826 + 6.08462314 + 72.44409075
= 87.466714354 amu
Rounding the result to two decimal places, the average atomic mass of strontium is approximately 87.47 amu.
Know more about atomic mass here:
https://brainly.com/question/30390726
#SPJ8
Which scientist determined that electrons had predicted zones orbiting the nucleus?
Rutherford
Bohr
Dalton
Schrödinger
Marthie
Answers
Answer:
Bohr
Explanation:
Neils Bohr was the scientist that suggested that electrons follow predictable orbits round the nucleus.
In 1913, he put forward his own model of the atom based on the quantum mechanics originally developed by Planck. He assumed the Rutherford's model and suggested that the extranuclear part consists of electrons in specific spherical orbits around the nucleus. The orbits/energy levels are designated as K, L, M, N.....Answer:
schrodinger
Explanation:
jelly benee
which of the following measurements is equivalent to 5.461x10^-7m?
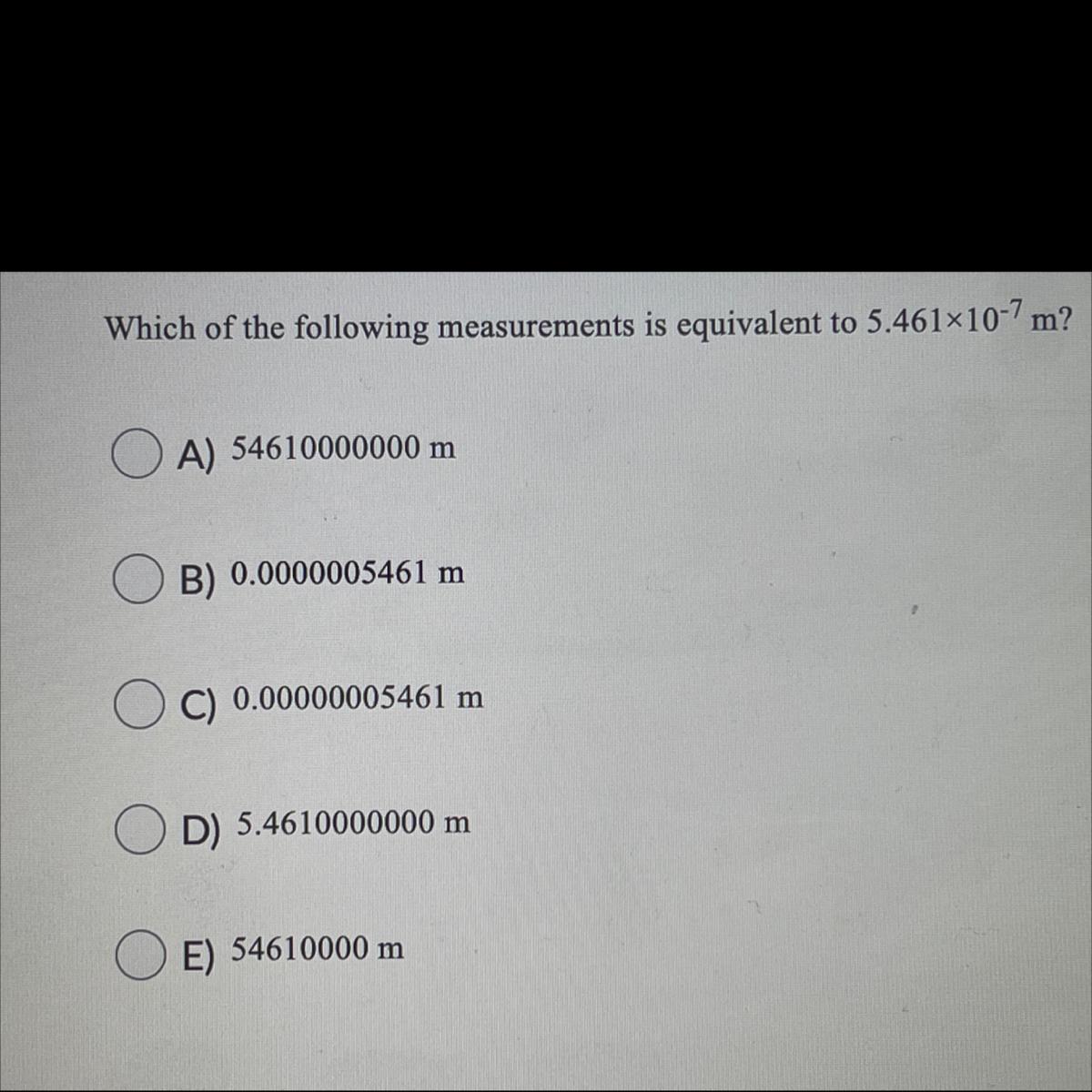
Answers
Answer:
B. 0.0000005461m
I used the method of moving the decimal.
2. 20.0 mL of a 0.75 M solution
of potassium permanganate,
KMnO solution is used to
make a 250.00 mL solution.
What is the concentration of
the new solution?
Answers
The concentration of the new potassium permanganate, KMnO solution is 0.06 M.
To find the concentration of the new solution, we can use the formula,
C₁V₁ = C₂V₂,
C₁ = 0.75,
V₁ = 20.0 mL,
V₂ = 250.0mL
C₂ is what we have to find. Plugging in the values we know, we get,
0.75 M x 20.0 mL = C₂ x 250.00 mL
Solving for C₂, we get the final concentration of the potassium permanganate.
C₂ = (0.75 M x 20.0 mL) / 250.00 mL
C₂ = 0.06 M
Therefore, the concentration of the solution has changed to 0.06 M from 0.75M.
To know more about concentration, visit,
https://brainly.com/question/17206790
#SPJ1
Iron is obtained by reaction of hematite (Fe2O3) with carbon monoxide. From the balanced reaction below, how many grams of iron is produced when 24.6 mol of hematite is used up in the reaction?
Fe2O3 + 3CO ® 2Fe + 3CO2
Answers
Answer: 2755.2 g of \(Fe\) will be produced from 24.6 mol of haematite.
Explanation:
The balanced chemical equation is:
\(Fe_2O_3+3CO\rightarrow 2Fe+3CO_2\)
According to stoichiometry :
1 mole of \(Fe_2O_3\) produce = 2 moles of \(Fe\)
Thus 24.6 moles of \(Fe_2O_3\) will produce=\(\frac{2}{1}\times 24.6=49.2moles\) of \(Fe\)
Mass of \(Fe=moles\times {\text {Molar mass}}=49.2moles\times 56g/mol=2755.2g\)
Thus 2755.2 g of \(Fe\) will be produced from 24.6 mol of haematite.