how many moles of oxygen molecule is reacting with sodium according to the following reaction:
2Na+O - Na2O2
Answers
1 mole of oxygen.
Answer:
solution given:
\(2Na+O_2=Na_2O_2\\2mole\:\: 1mole \:\: 1mole\)
according to the above reaction,1 mole of oxygen is required to react with 2 moles of sodium to form sodium peroxide.
Explanation:
Related Questions
a solution of acetic acid that has a concentration of 0.10 moles per liter has a ph of 2.87. what is the likely ph of a 0.10 mole per liter solution of the conjugate base sodium acetate?
Answers
0.10 moles per liter solution of the conjugate base sodium acetate is likely to have a pH greater than 7.
Is the pH of a 0.10 mole per liter solution of the conjugate base sodium acetate likely to be acidic or basic?When acetic acid (CH3COOH) donates a proton, it forms its conjugate base, acetate ion (CH3COO-). In the given scenario, the acetic acid solution has a pH of 2.87, indicating acidity. The lower pH value suggests a higher concentration of H+ ions. As a weak acid, acetic acid partially dissociates, releasing H+ ions and acetate ions. When sodium acetate (CH3COONa) dissolves in water, it completely dissociates into sodium ions (Na+) and acetate ions. The presence of acetate ions (the conjugate base) from sodium acetate will react with the excess H+ ions in the solution, shifting the equilibrium towards the formation of acetic acid and water. This process, called the hydrolysis of salts, will consume the H+ ions, thereby increasing the pH of the solution. Consequently, the 0.10 mole per liter solution of sodium acetate is likely to have a pH greater than 7, making it basic.
Learn more about pH
brainly.com/question/2288405
#SPJ11
a current of i = is charging a capacitor that has square plates of
Answers
A current of i = is charging a capacitor that has square plates of area A and separation d. What is the capacitance of this capacitor?The capacitance of the capacitor with square plates of area A and separation d can be determined using the main answer and below.
Main answer:Capacitance C = (ε₀ * A) / dExplanation:Given:i = Charging currentA = Area of square platesd = Separation between the platesThe capacitance of a capacitor is given by:Capacitance C = Charge / VoltageThe charge on each plate is given by:Q = i * tWhere:i = Currentt = TimeThe potential difference (voltage) across the capacitor is given by:V = Ed
Where:E = Electric field strengthd = Separation between the platesThe electric field strength E is given by:E = V / dSubstituting E in Q, we have:Q = ε₀ * A * VWhere:ε₀ = Permitivity of free spaced = Separation between the platesSubstituting Q and V in the formula for capacitance, we have: Capacitance C = (ε₀ * A) / dThus, the capacitance of the capacitor with square plates of area A and separation d is given by the formula above.
To know more about capacitor visit:
https://brainly.com/question/30577408
#SPJ11
What is the number of moles of H2 produced when 23 g of sodium react with water according to the equation 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g)
Answers
Answer:
0.5 mole of H₂.
Explanation:
We'll begin by calculating the number of mole in 23 g of Na. This can be obtained as follow:
Mass of Na = 23 g
Molar mass of Na = 23 g/mol
Mole of Na =?
Mole = mass / Molar mass
Mole of Na = 23 / 23
Mole of Na = 1 mole
Next, the balanced equation for the reaction.
2Na + 2H₂O —> 2NaOH + H₂
From the balanced equation above,
2 moles of Na reacted to produce 1 mole H₂.
Finally, we shall determine the number of mole of H₂ produced by the reaction of 23 g (i.e 1 mole) of Na. This can be obtained as follow:
From the balanced equation above,
2 moles of Na reacted to produce 1 mole H₂.
Therefore, 1 mole of Na will react to produce = (1 × 1) / 2 = 0.5 mole of H₂.
Thus, 0.5 mole of H₂ is obtained from the reaction.
a beaker containing 0.400 g khp was titrated with naoh solution. the pale pink end point was reached after 16.45 ml of naoh solution was dispensed. what is the molarity (in mol/l) of the naoh solution?
Answers
The molarity (in mol/l) of the naoh solution is 0.1191M .
The reaction between KHP and NaOH is 1:1molar
KHP(aq)+ NaOH(aq) -----> NaKHP(aq) + H2O(l)
mole= mass/molar mass
moles of KHP = 0.400g /204.22g/mol = 0.001959mol
0.001959moles of KHP react with 0.001959moles of NaOH
moles of NaOH present in the 16.45ml of NaOH solution = 0.001959mol
molarity = number of moles of solute per liter of solution
molarity of NaOH = (0.001959mol/16.45ml) ×1000ml = 0.1191M
To learn more about molarity visit:https://brainly.com/question/8732513
#SPJ4
Suggest a procedure for separating iron shavings from sawdust. Explain why this procedure would work.
Dont send a file i will report you if you do
Answers
Answer:
Magnetic separation
Sawdust is a non-magnetic material whereas iron shavings are magnetic. When a magnet is brought near a mixture of iron shavings and sawdust, the iron shavings will be attracted to the magnet while the sawdust will not. Thus, the iron shavings will be separated from the sawdust.
Explanation:
Separation techniques makes use of the differences in the properties of components of mixtures to separate theses components, one from another.
In a moxturemof iron shavings and sawdust, a suitable separation technique would be the use of magnetic separation.
The use of this separation technique is abasednonnthe principle that when a magnetnis brought near to a mixture containing magnetic and non-magnetic materials, the magnetic materials will be separated from the non-magnetic materials due to their attraction to the magnet. Sawdust is a non-magnetic material whereas iron shavings are magnetic. When a magnet is brought near a mixture of iron shavings and sawdust, the iron shavings will be attracted to the magnet while the sawdust will not. Thus, the iron shavings will be separated from the sawdust.
give two examples of energy transfer by conduction between two
objects
Answers
Bringing water to the boil in an electric kettle
Which statement is true with respect to the investigation? The student investigated only the physical properties of lead. The student investigated only the chemical properties of lead. The student investigated more physical properties than chemical properties of lead. The student investigated more chemical properties than physical properties of lead.
Answers
The question is incomplete, the complete question is;
A student investigated some properties of lead and recorded the findings in the table below. Property Observation Color Dull gray Density 11.3 g/cm3 Volume of sample 3 cm3 Melting point 327°C Reaction with air Lead oxide forms on the surface Electrical conductivity Conducts some electricity Which statement is true with respect to the investigation?
A. The student investigated only the physical properties of lead.
B. The student investigated only the chemical properties of lead.
C. The student investigated more physical properties than chemical properties of lead.
D. The student investigated more chemical properties than physical properties of lead.
Answer:
The student investigated more physical properties than chemical properties of lead.
Explanation:
If we look at the things mentioned in the question as regards the approach of the student towards the study of lead, we will discover that he/studied;
Colour, density, volume, melting point and electrical conductivity which are all physical properties. Physical properties of a substance does not involve chemical reactions of the substance under study.
The only chemical property studied by the student is its formation of an oxide- lead oxide with air as follows;
2Pb(s) + O₂(g) → 2PbO(s)
Thus we can safely conclude that; the student investigated more physical properties than chemical properties of lead.
Answer:
c
Explanation:
edge 2021 :)
How many liters does a 70. 9 gram sample of Cl2 (g) occupy at STP?
A. 5. 60 L
B. 11. 2 L
C. 22. 4 L
D. 44. 8 L
Answers
70.9-gram sample of \(Cl_{2}\) gas will occupy Opton C. 22.4 liters at STP.
To determine the volume occupied by the sample of \(Cl_{2}\) (g) at STP, we can use the ideal gas law equation, PV = nRT
where P = pressure
V = volume
n = number of moles
R = ideal gas constant
T = temperature.
At STP (Standard Temperature and Pressure), the pressure is 1 atmosphere (atm) and the temperature is 273.15 Kelvin (K).
First, calculate the number of moles of \(Cl_{2}\) (g) using its molar mass. The molar mass \(Cl_{2}\) is 70.9 grams/mol.
Number of moles (n) = mass (m) / molar mass (M)
n = 70.9 g / 70.9 g/mol
n = 1 mol
Now, we can calculate the volume using the ideal gas law:
V = (nRT) / P
V = (1 mol * 0.0821 L·atm/mol·K * 273.15 K) / 1 atm
V ≈ 22.4 L
Therefore, the correct answer is C. 22.4 L.
Know more about STP here:
https://brainly.com/question/13656229
#SPJ8
How could you change a molecule into an element
Answers
We can change a molecule into a series of one or more elements by breaking down the chemical bonds that connect atoms in the molecule.
What are the chemical bonds of a molecule?The chemical bonds of a molecule can be defined as the different types of attractive forces that link atoms in a molecule, which include, for example, covalent bonds, ionic bonds, hydrogen bonding, metallic bonding, etc.
Therefore, with this data, we can see that we have to break the chemical bonds of a molecule in order to separate molecules into their simplest constituents, i.e., atoms.
Learn more about chemical bonds here:
https://brainly.com/question/819068
#SPJ1
When filtered through a funnel into a flask, a mixture of substances X, Y and Z gets separated as below: - X stays in the funnel and Y and Z filter through and collect in the flask. X and Y are solids and Z is water. Which of the following can you conclude about the properties of X and Y?
Answers
Z is the solvent, Y is soluble in water while X is insoluble in water.
FiltrationFiltration is a method of separation of substances based on particle size. Only a particular particle size can pass through the filter. The substance that remains in the filter is the residue while the substances that passes through the filter is called the filtrate.
From the observation in the question Z is the solvent, Y is soluble in water while X is insoluble in water.
Learn more about separation of mixtures: https://brainly.com/question/863988
According to the
graph, what happens
to the concentration
of A over time?
Concentration (M)
Reaction: 2A A₂
Time (sec)
A. It decreases and then levels out.
B. It decreases consistently.
C. It increases and then levels out.
D. It increases consistently.
Answers
The concentration of A decreases and then levels out. Option A
How does concentration of the reactant change?
In many chemical reactions, a reactant is consumed as the reaction progresses, leading to a decrease in its concentration over time. The reactant molecules are transformed into products, and as the reaction proceeds, the concentration of the reactant gradually diminishes.
At equilibrium, the concentrations of both reactants and products remain relatively constant over time, although they can coexist.
Learn more abaout reactant:https://brainly.com/question/30129541
#SPJ1
List and describe two differences between pure substances and mixtures. Check all that apply. Check all that apply. The composition of a pure substance depends on the source, but the composition of a mixture doesn't depend on the source. The composition of a pure substance is always the same, regardless of the source, but the composition of a mixture can vary. The state of a pure substance is always the same, but the state of a mixture can vary. Mixtures can be separated into their components only by chemical changes; some pure substances can be separated into components by physical change. Mixtures can be separated into their components by physical changes; some pure substances can be separated into components by chemical change.
Answers
Answer:
The composition of a pure substance is always the same, regardless of the source, but the composition of a mixture can vary.
Mixtures can be separated into their components by physical changes; some pure substances can be separated into components by chemical change.
Explanation:
A chemically pure substance has a well defined and constant composition. The composition of a chemically pure substance remains the same irrespective of its source. Also, the components of a chemically pure substance may be separated by chemical changes.
Mixtures have a variable composition depending on their respective sources. The composition of a mixture varies with the source of the mixture and mixtures are separated by physical processes.
11. In the boxes below make Bohr models for each of the elements.
a. Determine how many electrons, protons, and neutrons there are in each atom.
b. Draw a Bohr model of each element using the number of electrons, protons, and neutrons
Electrons.
Protons
Neutrons
HELIUM
Electrons
Protons
Neutrons
OXYGEN
Answers
The atomic number represents the number of protons, whereas the mass number represents the quantity of protons and neutrons. The number of protons equals the amount of electrons.
In a Bohr diagram, where do you determine the number of neutrons?The atomic number is the number of protons. The number of neutrons may be calculated by subtracting the number of protons from the atomic mass, which is then rounded to the closest whole number.
For neutrally charged species, the number of electrons in an atom equals the atomic number of an element. This signifies that the number of electrons and protons in an element are equal. As a result, the number of electrons in oxygen is eight.
The number of valence electrons in neutral atoms equals the atom's main group number. A periodic table column can be used to determine an element's main group number. Carbon, for example, belongs to group 4 and contains four valence electrons.
learn more about valence electrons refer
https://brainly.com/question/13552988
#SPJ9
The interval shown here represents a _____.
strong base
weak base
weak acid
strong acid

Answers
Answer:
the answer is strong acid
The correct answer is strong acid
What do transition metals have in common
Answers
Answer: The transition metals have the following physical properties in common: they are good conductors of heat and electricity. they can be hammered or bent into shape easily. they have high melting points (but mercury is a liquid at room temperature)
Explanation:
Explain the mechanism of a Horner-Wadsworth-Emmons reaction between diethyl benzylphosphonate and 3,4-methylenedioxybenzaldehyde in the presence of aqueous NaOH forming 3,4-methylenedioxystilbene as the product.
Answers
Horner-Wadsworth-Emmons (HWE) reaction is an important synthetic reaction in organic chemistry. It is widely used for synthesizing various compounds. The reaction is between an aldehyde or ketone and a phosphonate or phosphonate ester in the presence of a strong base.
The Horner-Wadsworth-Emmons reaction is one of the most convenient and well-known methods of constructing carbon-carbon double bonds. The reaction proceeds via the formation of an ylide intermediate. The HWE reaction is particularly useful for the synthesis of compounds with a Z-configuration.
The mechanism for the reaction of diethyl benzylphosphonate and 3,4-methylenedioxybenzaldehyde in the presence of aqueous NaOH, forming 3,4-methylenedioxystilbene as the product, can be explained in the following steps:
Step 1: Formation of the ylide intermediate
The reaction starts with the formation of an ylide intermediate. This is achieved by the reaction of diethyl benzylphosphonate and 3,4-methylenedioxybenzaldehyde in the presence of a strong base like NaOH or KOH. In this reaction, a deprotonated species called an ylide intermediate is generated.
Step 2: Addition of the ylide intermediate to the aldehyde
The ylide intermediate then attacks the aldehyde, leading to the formation of a betaine intermediate.
Step 3: Formation of the phosphonate ester
The betaine intermediate undergoes elimination to form the final product, 3,4-methylenedioxystilbene, and the by-product phosphonate ester.
The mechanism of the Horner-Wadsworth-Emmons (HWE) reaction between diethyl benzylphosphonate and 3,4-methylenedioxybenzaldehyde in the presence of aqueous NaOH, forming 3,4-methylenedioxystilbene as the product, is complete. This reaction is significant in organic chemistry and finds applications in the pharmaceutical industry.
Learn more about reaction
https://brainly.com/question/30464598
#SPJ11
Write a Lewis structure that obeys the octet rule for the following species. Assign the formal charge for the central atom of. ClO3-If multiple resonance structures exist, use one that does not involve an expanded valence
Answers
The Lewis structure for ClO3- is as follows:
O
|
Cl--O
|
O-
To determine the formal charge of the central atom Cl, we need to calculate the valence electrons and nonbonding electrons present in ClO3-. Chlorine has 7 valence electrons, and each oxygen atom contributes 6 electrons for a total of 24 valence electrons. In this structure, there are 3 lone pairs on each oxygen atom and one Cl-O double bond.
The formal charge of Cl can be calculated as follows:
Formal charge = Valence electrons - Nonbonding electrons - 1/2 (bonding electrons)Formal charge of Cl = 7 - 6 - 4 = -3The formal charge on the central atom, Cl, is -3. This indicates that Cl has an extra electron compared to its neutral state. The other oxygen atoms have a formal charge of -1 each, indicating that they have an extra electron as well. This arrangement of formal charges indicates that the ClO3- ion is a negatively charged species. The Lewis structure shows that ClO3- obeys the octet rule as each atom has a full outer shell of electrons.
To learn more about Lewis structure, here
https://brainly.com/question/20300458
#SPJ4
i just need #2, its due any minute. giving extra points, and will mark brainliest!!!!!!!!
which unknown has the weakest attractive forces?
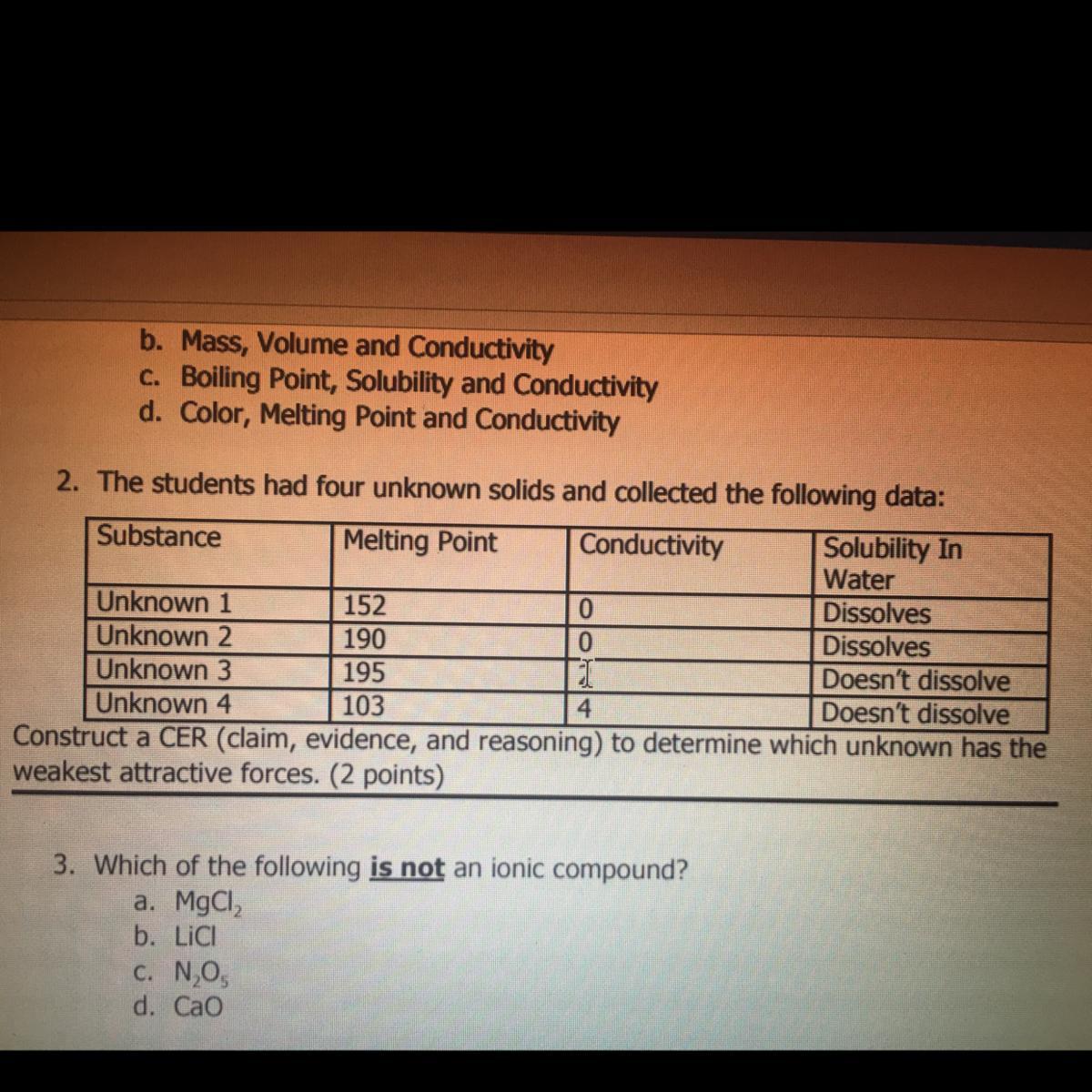
Answers
Answer:
Unknown 4
Explanation:
HELP PLEASE !!!
Name the Salt formed by Phosphoric acid *
Sulfate Salt
Nitrate Salt
Phosphate Salt
Chloride Salt
Answers
Answer:
sodium dihydrogen phosphate (NaH2PO4),
Phosphate Salt
- BRAINLIEST answerer
Q-3 Determine the fugacity in atm for pure ethane at 310 K and 20.4 atm and change in the chemical potential between this state and a second state od ethane where temperature is constant but pressure is 24 atm.
Answers
The fugacity in atm for pure ethane at 310 K and 20.4 atm is given by the equation: f = 20.4 exp (-Δg1/RT). The change in chemical potential between this state and a second state of ethane where the temperature is constant but the pressure is 24 atm is -0.0911RT.
Fugacity is a measure of the escaping tendency of a component in a mixture, which is defined as the pressure that the component would have if it obeyed ideal gas laws. It is used as a correction factor in the calculation of equilibrium constants and thermodynamic properties such as chemical potential. Here we need to determine the fugacity in atm for pure ethane at 310 K and 20.4 atm and the change in the chemical potential between this state and a second state of ethane where the temperature is constant but the pressure is 24 atm. So, using the formula of fugacity: f = P.exp(Δu/RT) Where P is the pressure of the system, R is the gas constant, T is the temperature of the system, Δu is the change in chemical potential of the system. Δu = RT ln (f / P)The chemical potential at the initial state can be calculated using the ideal gas equation as: PV = nRT
=> P
= nRT/V
=> 20.4 atm
= nRT/V
=> n/V
= 20.4/RT The chemical potential of the system at the initial state is:
Δu1 = RT ln (f/P)
= RT ln (f/20.4) Also, we know that for a pure substance,
Δu = Δg. So,
Δg1 = Δu1 The change in pressure is 24 atm – 20.4 atm
= 3.6 atm At the second state, the pressure is 24 atm.
Using the ideal gas equation, n/V = 24/RT The chemical potential of the system at the second state is: Δu2 = RT ln (f/24) = RT ln (f/24) The change in chemical potential is Δu2 – Δu1 The change in chemical potential is
Δu2 – Δu1 = RT ln (f/24) – RT ln (f/20.4)
= RT ln [(f/24)/(f/20.4)]
= RT ln (20.4/24)
= - 0.0911 RT Therefore, the fugacity in atm for pure ethane at 310 K and 20.4 atm is:
f = P.exp(Δu/RT)
=> f
= 20.4 exp (-Δu1/RT)
=> f
= 20.4 exp (-Δg1/RT) And, the change in the chemical potential between this state and a second state of ethane where the temperature is constant but pressure is 24 atm is -0.0911RT. Therefore, the fugacity in atm for pure ethane at 310 K and 20.4 atm is given by the equation: f = 20.4 exp (-Δg1/RT). The change in chemical potential between this state and a second state of ethane where the temperature is constant but the pressure is 24 atm is -0.0911RT.
To know more about chemical potential visit:-
https://brainly.com/question/31100203
#SPJ11
If a 200 g piece of aluminum has a density of 5.0 g/cm^3. what is its volume?
Answers
Answer:
Volume=mass in g /density
Answer: 40cm^3 or 40ml
Explanation:
\(Density=\frac{mass}{volume}\)
so 5=200/V
V=200/5
V=40cm^3
Using the chemical formulae alone, how can you differentiate between Type I Ionic, Type II Ionic, Type III Ionic, and Covalent compounds?
Answers
Explanation:
Type I
These are compounds in which the cation (metal ion) has a fixed charge.
Examples are;
MgO. NaCl
Type II
These are ionic compounds in which the cation (metal ion) has a variable charge.
Examples are;
FeCl3, PbS2
Type III
These are covalent molecules with no cation.
Examples are;
SO3, N2O5
3.A 50.00 mL sample of vinegar was titrated with 0.35 M NaOH. The endpoint was reached when 32.48 mL of the NaOH was added. What was the concentration of Acetic Acid in the vinegar solution
Answers
The concentration of Acetic Acid in the vinegar solution is 0.22736 mol/L.
To find the concentration of acetic acid in the vinegar solution we can use the following equation :
CH3COOH(aq) + NaOH(aq) → NaCH3COO(aq) + H2O(l)
Let's determine the number of moles of NaOH in the solution :
moles NaOH = Molarity × Volume = 0.35 mol/L × 0.03248 L = 0.011368 mol
The balanced chemical equation is used to find the number of moles of acetic acid present in the vinegar solution.
1 mole of NaOH reacts with 1 mole of CH3COOH.
The number of moles of acetic acid in the vinegar solution can be calculated as
moles CH3COOH = moles NaOH = 0.011368 mol
The molar concentration of the acetic acid in the vinegar solution is given by the expression :
concentration of CH3COOH = moles CH3COOH / Volume of vinegar solution in Liters
= 0.011368 mol / 0.0500 L = 0.22736 mol/L
Therefore, the concentration of Acetic Acid = 0.22736 mol/L.
To lean more about concentration :
https://brainly.com/question/17206790
#SPJ11
what does Le châteliers principle state?
Answers
Hope this helps!
A solution made by dissolving 25. 0 mg of insulin in 5. 00 mL of water has an osmotic pressure of 15. 5 mmHg at 25°C. Calculate the molar mass of insulin. (Assume that there is no change in volume when the insulin is added to the water and that insulin is a non-dissociating solute. )
Answers
The molar mass of insulin is approximately 0.798 g/mol, calculated using the equation for osmotic pressure and the given values of mass and volume.
To calculate the molar mass of insulin, we can use the equation for osmotic pressure:
π = (n/V)RT
where π is the osmotic pressure, n is the number of moles of solute, V is the volume of the solution in liters, R is the ideal gas constant, and T is the temperature in Kelvin.
First, convert the given values to appropriate units:
25.0 mg = 0.025 g
5.00 mL = 0.005 L
Next, rearrange the equation to solve for n (number of moles):
n = (πV) / (RT)
Substituting the given values:
n = (15.5 mmHg * 0.005 L) / ((0.0821 L·atm/(mol·K)) * 298 K)
Calculate n:
n ≈ 0.0313 mol
Finally, divide the mass of insulin (0.025 g) by the number of moles (0.0313 mol) to find the molar mass:
Molar mass = 0.025 g / 0.0313 mol
Molar mass ≈ 0.798 g/mol
So, the molar mass of insulin is approximately 0.798 g/mol.
Learn more about osmotic pressure here:
https://brainly.com/question/29823250
#SPJ11
Which statement describes the relative energy of the electrons in the shells of a calcium atom?
Answers
Answer:
An electron in the first shell has more energy than an electron in the second shell.
Explanation:
What is the average speed of a bicyclist who rides 75 km in 2.5 hours? please show how you worked it out if you can bc my teacher is very specific
Answers
Answer:
30
Explanation:
I’m pretty sure it is

I'd love some help please thank you

Answers
Answer:
the answer would be B
Explanation:
The move vigorously it is the higher the reactivity so if you put it in order the answer would be B
Because that answer makes the most sense.
if i have 50 liter container that holds 1.41 ,oles of o2 at a tempature of 473k what is the presure contain inside of the conteiner
Answers
The pressure inside the container is 3.25 × 10⁴ Pa.
Calculation-The ideal gas law, which connects a gas's pressure, volume, temperature, and molecular weight:
PV = nRT
We are given the volume of the container (50 L = 0.050 m³), the number of moles of oxygen gas (1.41 mol), and the temperature (473 K). We can rearrange the ideal gas law to solve for the pressure:
P = nRT/V
Plugging in the values we get:
P = (1.41 mol) × (8.31 J/(mol·K)) × (473 K) / (0.050 m³)
P = 3.25 × 10⁴ Pa
to know more about temperature here:
brainly.com/question/17425457
#SPJ1
the mass of the atom comes from?
A) the electrons
B) the protons
C) the neutrons
D) the nucleus
Answers
Explanation-
The nucleus contains the majority of an atom's mass because protons and neutrons are much heavier than electrons, whereas electrons occupy almost all of an atom's volume.