Dr. Laila Malik has been tasked to help her graduate student with making a ground-breaking molecule, but she has to start with helping the student understand how different elements bond to each other and the characteristics they have. Help Dr. Malik identify whether the compound is polar or non-polar: AgBr
Answers
We must identify if AgBr is polar or non-polar.
In order to determine this we must use that
Now, we can see that the compound has 2 atoms: Ag and Br and they are different. So, the compound is polar.
ANSWER:
The compound is polar.
Related Questions
An early arrangement of the then known elements was proposed by a British scientist John Newlands, which he called the Law of Octaves. Like other scientists at the time, Newlands arranged the elements in order of increasing atomic mass and noted that every eighth element had similar physical/chemical properties. In the modern Periodic Table, which of the following represents the last pair of elements for which Newlands' Law of Octaves would hold true?
Answers
Give the name for SnO
A) tin(I) oxide
B) tin(II) oxide
C) tin(III) oxide
D) tin(IV) oxide
Answers
The name for the compound SnO is tin(II) oxide. Therefore, option B is correct.
What is compound ?A chemical compound is a material that contains atoms from many chemical elements bonded together by chemical bonds. It is made up of numerous similar molecules. Hence, a molecule made up of only one type of atom is not a compound.
The difference in electronegativity between the electronegative O and the somewhat electropositive Sn is 1.48, which suggests that the bond between SnO is polar covalent.
The name of the compound SnO is tin monoxide.
Thus, the compound SnO is tin(II) oxide, option B is correct.
To learn more about the compound, follow the link;
https://brainly.com/question/13516179
#SPJ1
Electron transfer between redox centers in proteins is controlled by quantum tunneling. We can model the region between two redox centers as an energy barrier which the electron must cross. If the distance between the redox centers is 0.681 nm and the energy of the electron is 0.5540.554eV lower than the height of the barrier, what is the probability that the electron will successfully cross to the next redox center
Answers
Answer:
5.61 e⁻³
Explanation:
Given that:
The barrier length = 0.681 nm = 6.81 × 10⁻¹⁰
The difference between the height of the barrier & the energy of the electron is;
\(V - E = 0.554 \ eV (1.6 \times 10^{-19} )\)
= 8.864 × 10⁻²⁰ V
where;
m = 9.1 × 10⁻³¹ kg
The probability\(P = e^{-2 \ kd}\)
\(k = 2 \pi \sqrt{\dfrac{2m (V-E)}{h^2} }\)
\(k = 2 \pi \times \dfrac{\sqrt{2 \times 9.1 \times 10^{-31} \times 8.864 \times 10^{-20}}}{6.63 \times 10^{-34}}\)
k = 3.806 × 10⁹
\(P = e^{-2 \ kd}\)
\(P = e^{2 \times 3.806 \times 10^9 \times 6.81 \times 10^{-10}}\)
P = 0.005606
P = 5.61 × 10⁻³
P = 5.61 e⁻³
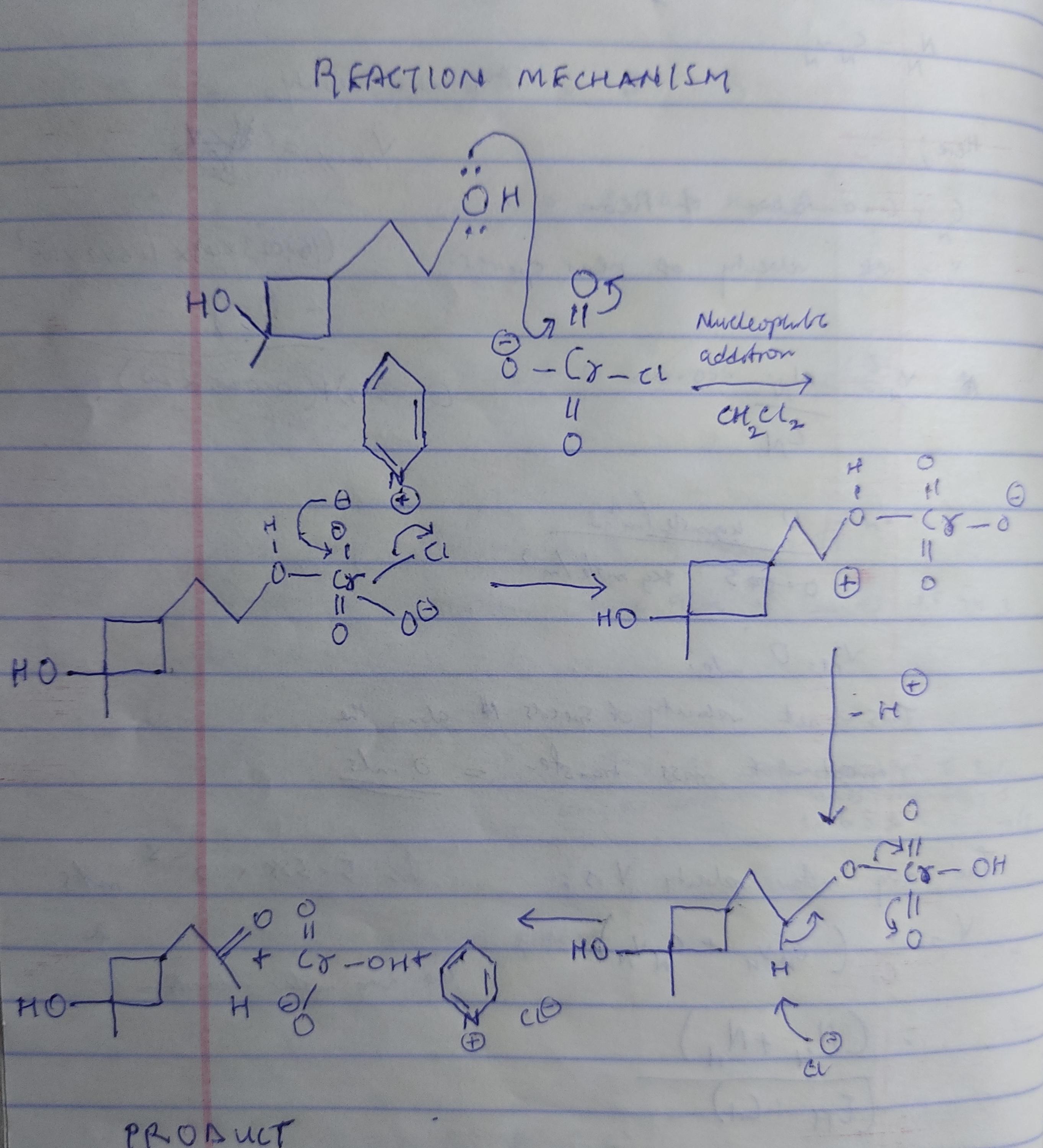
Give the product expected when the following alcohol reacts with pyridinium chlorochromate (PCC). (Assume that PCC is present in excess.) The starting material is H O C H 2 C H 2 C H 2 O H. THis reacts with P C C and C H 2 C L 2 to give the product.
Answers
Answer:
Explanation:
From the information given, we are to show the product of the reaction between the alcohol (stick formula) with pyridinium chlorochromate. The reaction starts with the nucleophilic addition of Cr with oxygen molecule on the alcohol. Then, deprotonation of oxygen occurs, this process is followed by the removal of hydrogen ion to yield the final product. The reaction mechanism can be seen from the attached image below.
The diagram shows changes of state between solid, liquid, and gas. The atoms of a substance lose energy during a change of state. After the change, the atoms are close together but are able to slide past one another. A diagram has a triangle at center with gas on top, liquid on bottom right, solid on bottom left. An arrow from gas to liquid is labeled O, and an arrow from liquid to gas is labeled N. An arrow from solid to liquid is labeled P, and an arrow from liquid to solid is labeled Q. An arrow from solid to gas is labeled L, and an arrow from gas to solid is labeled M. Which arrow represents the change of state described above? L N O P
Answers
Answer:
IM NOT TO SURE ABOUT THIS DO YOU HAVE THE DIAGRAM
Explanation:
PLEASE HELP
Step 3: Apply the scaling factor to the
empirical formula.
The scale factor is 5 and the empirical
formula is CH2O. What is the molecular
formula?
Answers
The molecular formula is (CH2O)5 when the scale factor is 5 and the empirical formula is CH2O.
Considering the question given above, the following data were obtained:
Empirical formula is CH₂O
Molar mass of compound = 150 g/mol
Scaling factor (n) =?
Empirical formula × n = molar mass
[CH₂O]n = 150
[12 + (2×1) + 16]n = 150
[12 + 2 + 16]n = 150
30n = 150
Divide both sides by 30
n = 5
Therefore, the scaling factor is 5
A scale factor is a ratio between corresponding measurements of an object and a representation of that object.In chemistry, the empirical formula of a chemical compound is the simplest whole-number ratio of atoms present in a compound. .Learn more about empirical formula at:
https://brainly.com/question/14044066
#SPJ1
Answer:
The scale factor is 5. The formula is CH2O.
Multiply each by 5.
So:
C (there is only one)= 1×5
H (there are 2)= 2×5
O(there is only one)= 1×5
Equalling= C5H10O5
Question 1 (1 point)
Stars come in different
a:colors
b:sizes
c:temperatures
d:all of the above
PLESE HELP ANYONE HURRY!
Answers
Answer:
All of above.
Explanation:
Stars. They come in different sizes, temperatures, and colors. Not all are yellow, and not all are small or big. Some can be hot, and others can be cold. Stars can exist for a while, years on end. It just varies on how long it's been there.
I hope this helps you!
Which of the following would cause an increase in the magnetic force
between two magnets?
A. Decreasing the separation between the two magnets
B. Increasing the separation between the two magnets
C. Decreasing the amount of excess charge on the first magnet
D. Increasing the amount of excess charge on the first magnet
Answers
Decreasing the separation between the two magnets will result in increase in the magnetic force between two magnets.
What is a Magnet?This is referred to a substance which produces magnetic field and result in the attraction and repulsion of certain types of things.
Increase in the separation will reduce the magnetic force and vice versa which is why option A was chosen.
Read more about Magnet here https://brainly.com/question/14997726
#SPJ1
Which of the following elements can form double covalent bonds?
Answers
Answer:
double bond
Explanation:
Sometimes two covalent bonds are formed between two atoms by each atom sharing two electrons, for a total of four shared electrons. For example, in the formation of the oxygen molecule, each atom of oxygen forms two bonds to the other oxygen atom, producing the molecule O2. Similarly, in carbon dioxide (CO2), two double bonds are formed between the carbon and each of the two oxygen atoms
A student applies a force to a box with a mass of 30 kg. If the student applies the same force to a box with a mass of 15 kg, which best describes the effect on the acceleration of the 15-kg box?
Answers
The effect on the acceleration of the 15-kg box would be that it would experience a greater acceleration than the 30-kg box.
Newton's second lawAccording to Newton's second law of motion, the acceleration of an object is directly proportional to the net force acting on it and inversely proportional to its mass. Therefore, for a given force, an object with a smaller mass will experience a greater acceleration than an object with a larger mass.
In this scenario, if the student applies the same force to both the 30-kg box and the 15-kg box, the 15-kg box will experience a greater acceleration than the 30-kg box. This is because the smaller mass of the 15-kg box means that it has less inertia and requires less force to accelerate.
Therefore, the effect on the acceleration of the 15-kg box would be that it would experience a greater acceleration than the 30-kg box.
Learn more on Newton's laws of motion here https://brainly.com/question/7578203
#SPJ1
Sexual reproduction occurs when two specialized sex cells also known as _____________, fuse together.
Answers
Answer:
Explanation:
During sexual reproduction the two gametes join together in a fusion process known as fertilization, to create a zygote, which is the precursor to an embryo offspring, taking half of its DNA from each of its parents. In humans, a zygote contains 46 chromosomes: 23 from its mother and 23 from its father
Describe how lead as a toxic metal can be determine in borehole water?
Answers
We can be able to determine the amount of toxic lead in the water by thee use of atomic absorption spectrophotometry.
What is a toxic metal?
A toxic metal is known as any metal that is able to affect the health of people. We know that toxic metals are mostly the metals that are in the group of the heavy metals.
Now we know lead as a metal that is able to cause brain damage especially in children. This is why it is very important that there should be a thorough examination in order to know the amount of lead that is present in water.
There are several methods that could be applied in the determination of lead and one of the most common methods is by the use of atomic absorption spectrophotometry which is able to detect even the minutest amount of the led in solution.
Learn more about toxic metals:https://brainly.com/question/28331004
#SPJ1
Determine the number of moles in 5.00 × 10²³ formula units of BaBr₂.
Answers
The number of moles in 5.00 × 10²³ formula units of BaBr₂ is approximately 0.83 moles, calculated by dividing the given number of formula units by Avogadro's number (6.022 × 10²³).
Explanation:To determine the number of moles in 5.00 × 10²³ formula units of BaBr₂, you have to know the Avogadro's number, which is 6.022 × 10²³ formula units per mole.
Therefore, using Avogadro's number, you divide the given number of formula units by this value. So, the calculation is as follows: 5.00 × 10²³ formula units / 6.022 × 10²³ formula units per mole = 0.83 moles of BaBr₂.
So, there are approximately 0.83 moles in 5.00 × 10²³ formula units of BaBr₂.
Learn more about Moles here:https://brainly.com/question/34302357
#SPJ2
it was believed that earth was a stationary object at the center of the universe all other objects in space without to go around earth which of these is a criticism of this idea
Answers
Answer: is that a question or
Explanation:
Answer:
its c
Explanation:
(b) How many moles of hydrogen are needed to make 30 moles of Fe?
PLEASE HELP ASAP
Answers
Answer:
1 mole of Fe is 55.85 g per mole
Explanation:
hope it helps
The moles of hydrogen that are needed to make 30 moles of Fe is 45 moles.
What are moles?Moles is a measurement unit. It is the quantity amount of substance.
The balanced equation is
Fe2O3 + 3H2 ---> 2Fe + 3H2O
Given the moles of iron is 30
To find the moles of hydrogen is?
According to the given balanced equation, the ratio of moles of hydrogen and iron is 3 : 2
To produce 30 moles of Fe
30 x 3 / 2 = 45 moles.
Thus, the number of moles of hydrogen is 45 moles.
To learn more about moles, refer to the link:
https://brainly.com/question/20486415
#SPJ5
Your question is incomplete. Your most probably complete question is given below:
One way to change iron are, Fe2O3, into metallic iron is to heat it together with hydrogen:
Fe2O3 + 3H2 ---> 2Fe + 3H2O
The enthalpy of fusion of butane, CH1o, is 24.3 kJ/mol at the melting point of -0.5°C. What is the change in entropy for the crystallization of 1.25 mole of butane?
Answers
The change in entropy for the crystallization of 1.25 moles of butane can be calculated using the formula ΔS = ΔH_fusion / T, where ΔS is the change in entropy, ΔH_fusion is the enthalpy of fusion, and T is the temperature in Kelvin.
In the given problem, the enthalpy of fusion of butane is 24.3 kJ/mol. To convert this to J/mol, we multiply it by 1000, resulting in 24,300 J/mol. The melting point of butane is -0.5°C, which is equivalent to 272.65 K.
Using the formula, we can calculate the change in entropy as follows:
ΔS = (24,300 J/mol) / 272.65 K
≈ 89.25 J/(mol·K)
Therefore, the change in entropy for the crystallization of 1.25 moles of butane is approximately 89.25 J/(mol·K).
The change in entropy (ΔS) for the crystallization of a substance can be determined using the equation ΔS = ΔH_fusion / T, where ΔH_fusion is the enthalpy of fusion and T is the temperature in Kelvin. In this case, the enthalpy of fusion of butane is given as 24.3 kJ/mol, which is converted to 24,300 J/mol. The melting point of butane is -0.5°C, which is equivalent to 272.65 K.
By substituting the values into the equation, we find that the change in entropy is approximately 89.25 J/(mol·K). This means that for the crystallization of 1.25 moles of butane, the entropy decreases by 89.25 J/(mol·K).
Entropy is a measure of the disorder or randomness of a system. In this case, the crystallization process involves the transition from a disordered liquid state to an ordered solid state, resulting in a decrease in entropy. The magnitude of the entropy change depends on the enthalpy of fusion and the temperature. A higher enthalpy of fusion or a lower temperature leads to a larger change in entropy.
for such more questions on entropy
https://brainly.com/question/419265
#SPJ8
A sprinter is running 200.0 m in 21.9s. What is the average speed of the sprinter in km per hour?
Answers
Answer:
Average speed = 333.3 km/hrExplanation:
The average speed of a body can be found by using the formula
\(Average \: \: speed = \frac{distance}{time} \)
To find the average speed in km/h first convert the distance from m to km and the seconds from s to hr.
For the distance
2000 m
1000 m = 1 km
2000 m = 2000/1000 = 2 km
For the seconds
21.9 s
3600 s = 1 hr
21.9 s = 21.9/3600 = 0.006 hrs
Substitute the values into the above formula and solve for the Average speed
That's
\(Average \: \: speed = \frac{2}{0.006} \)
We have the final answer as
Average speed = 333.3 km/hr
Hope this helps you
Which of these is an example of an agricultural use for radiation?
A. making heavy isotopes to find new elements.
B. irradiating wheat to kill fungus.
C. diagnostic procedures that image inside the body, such as a PET scan.
D. locating leaks in a water line in a building.
Answers
Option B. irradiating wheat to kill fungus is an example of agricultural use for radiation.
What is the relative significance of agricultural use for radiation?The relative significance of agricultural use for radiation is based on the fact that radiation is a physic mutagenic agent and therefore it can be sued to produce mutations in undesired organisms in order to kill them.
Therefore, with this data, we can see that the relative significance of agricultural use for radiation is based on the generation of triggered mutations in undesired organisms such as plagues.
Learn more about the use of radiation here:
https://brainly.com/question/1333944
#SPJ1
Calculate the volume of a box, in cubic centimeters, which is 423 cm long, 12 cm wide, and 25 cm high. Report your answer with
correct significant figures in cubic centimeters.
Answers
Answer:
\(V=1.3x10^5cm^3\)
Explanation:
Hello,
In this case, since the volume is obtained by multiplying the length, width and height of the box:
\(V=W*L*H\)
Given each dimension we find:
\(V=423cm*12cm*25cm\\\\V=126900cm^3\)
Nevertheless, since the shortest number is 12, we need to show the volume with two significant figures and in scientific notation as follows:
\(V=1.3x10^5cm^3\)
Best regards.
Perform the following mathematical operation, and report the answer to the correct number of significant figures.
0.34 x 0.568 = [?]
Answers
Answer:
0.34 x 0.568 = [0.19312]
Explanation:
just multiply it
Answer: 0.19
Explanation: 0.34 x 0.568 = 0.19312. this answer does not have the correct amount of significant figures though, so you have to round. 0.568 has 3 significant figures, and 0.34 has 2 significant figures. you always want to round to the smaller amount of significant figures, which is two in this case. 0.19312 rounds down to 0.19.
A cube of an unknown metal was dropped in a graduated cylinder containing 45.0mL of water. After the metal cube sank to the bottom of the graduated cylinder, the water level read 45.5mL. If the metal cube weighs 10.7grams what is the identity?
A.) platinum: D= 21.4 g/cm^3
B.) Gold: D=19.3g/cm^3
C.) silver: D=10.5 g/cm^3
D.) Copper: D=8.92g/cm^3
Answers
Answer:
Explanation:
A
3. A Wilkinson’s catalyst is widely used in the hydrogenation of alkenes. Show a catalytic cycle, including: i. chemical structure of the catalyst, with complete stereochemistry ii. molecular geometry of catalyst iii. type of reactions involved iv. the appropriate starting material, reagent and solvent v. major and minor end-products vi. all intermediates, for each reaction stated in (iii)
Answers
We can see here that the catalytic cycle for the hydrogenation of alkenes using Wilkinson's catalyst involves several steps.
What are the steps involved?Here's an overview of the catalytic cycle, including the necessary details:
i. Chemical structure of the catalyst:
Wilkinson's catalyst, also known as chloridotris(triphenylphosphine)rhodium(I), has the following chemical structure: [RhCl(PPh3)3]
ii. Molecular geometry of the catalyst:
The Wilkinson's catalyst has a trigonal bipyramidal geometry around the rhodium center. The three triphenylphosphine (PPh3) ligands occupy equatorial positions, while the chloride (Cl) ligand occupies an axial position.
iii. Type of reactions involved:
The catalytic cycle involves several reactions, including:
Oxidative addition: The rhodium center undergoes oxidative addition, reacting with molecular hydrogen (H2) to form a dihydride intermediate.Alkene coordination: The alkene substrate coordinates to the rhodium center, forming a π-complex.Hydrogenation: The coordinated alkene undergoes hydrogenation, resulting in the addition of hydrogen atoms to the double bond and formation of a metal-alkyl intermediate.Reoxidation: The metal-alkyl intermediate reacts with a hydrogen molecule to regenerate the rhodium dihydride species.iv. Starting material, reagent, and solvent:
The starting material is an alkene, and the reagent is Wilkinson's catalyst ([RhCl(PPh3)3]). The reaction is typically carried out in a suitable solvent, such as dichloromethane (CH2Cl2) or tetrahydrofuran (THF).
v. Major and minor end-products:
The major end-product of the hydrogenation reaction is the fully saturated alkane, resulting from the addition of hydrogen across the double bond. The minor end-product may include cis- or trans-configured alkanes if the original alkene substrate possesses geometric isomers.
vi. Intermediates:
The intermediates in the catalytic cycle include:
Rhodium dihydride complex: [RhH2(PPh3)3]Alkene-Rhodium π-complex: [Rh(η2-alkene)(PPh3)3]Metal-alkyl intermediate: [Rh(alkyl)(PPh3)3]These intermediates play a crucial role in facilitating the hydrogenation reaction and enabling the catalytic cycle to proceed.
Learn more about Wilkinson’s catalyst on https://brainly.com/question/31972308
#SPJ1
Substances that are physical combinations of two or more different types of matter are known as
Answers
Answer:
A mixture!
Explanation:
Mixtures are a substance made of combining two or more different kinds of matter :)
Hope this helped!
Please calculate your BMI by taking your mass in kilograms divided by your
height in meters squared. Please help me out
Answers
Answer:
you find your bmi by dividing your weight in kg by your height in Meters then square it
this is the formula kg/m2
Explanation:
The irreversible isomerization A
B was carried out in a batch reactor and the following concentration time data were obtained:
Time vs Concentration data in a Batch reactor
t 0 3 5 8 10 12 15 7.5
mol/h 4 2.89 2.25 1.45 1.0 0.65 0.25 0.07
Determine the reaction order,
, and the specific reaction a rate constant, k, using any method of your choice.

Answers
The reaction order and specific reaction rate constant can be determined by performing the kinetics experiment on irreversible polymerization A. Kinetic experiments can be used to investigate the rate and mechanism of chemical reactions. Chemical kinetics is the study of chemical reactions' speed and pathway.
The term "kinetics" refers to the study of reaction rates, which are determined by measuring the concentration of reactants and products as a function of time.Kinetics experiments can be used to determine the reaction rate and order of reaction. A chemical reaction's rate is defined as the change in the concentration of a reactant or product per unit time. The order of a reaction refers to the number of molecules that must react to produce a product. The order of reaction can be determined by measuring the initial rate of the reaction as a function of concentration.Methods for determining the reaction rate order include the initial rate method, the half-life method, and the integrated rate method. The initial rate method determines the reaction order by measuring the initial rate of the reaction at different reactant concentrations. The half-life method determines the reaction order by measuring the time it takes for the reactant concentration to decrease by half.The integrated rate method determines the reaction order by measuring the concentration of the reactant or product at different times.The specific rate constant can be determined by using the Arrhenius equation, which relates the rate constant to the activation energy, temperature, and frequency factor. The frequency factor can be determined by measuring the rate constant at different temperatures.For such more question on polymerization
https://brainly.com/question/1602388
#SPJ8
Which number is the same as 2.5
10-3?
Answers
The number that is the same as the exponentiation given as follows: 2.5 × 10-³ is 0.0025.
What is exponentiation?Exponentiation is the process of calculating a power by multiplying together a number of equal factors, where the exponent specifies the number of factors to multiply.
For example, if 10 is multiplied three times, then it can be written as "10 raised to 3" which means 10³. In this case, 10 is the base, and 3 is the exponent.
Therefore, a number 0.0025 can be written in exponentiation as 2.5 × 10-³ by counting the number of zeros forward.
Learn more about exponentiation at: https://brainly.com/question/28596571
#SPJ1
Using the two cell reduction potentials shown for their corresponding reaction, calculate the cell potential for a voltaic cell made from these two systems.
A)
–1.68 V
B)
1.68 V
C)
0.78 V
D)
–0.78 V

Answers
Answer:
C: 0.78 V
Explanation:
The Cr2O7 cell has a higher reduction potential, so it will be reduced (as it is a better oxidizing agent). This means that Fe will be oxidized. Its oxidation potential is simply the negative of its reduction potential, 0.45: \(E_{ox}=-0.45\). We know that cell potential is the sum of the reduction and oxidation potentials of each reaction (it is not an extensive property like enthalpy or entropy, so we don't need to worry about multiplying anything). Therefore, \(E_{cell}=(-0.45+1.23)V=0.78V\).
Would having different atmospheric pressures have an effect on the accuracy of gas laws? If so, which planets would be the most reliable and which would be the least?
Answers
Answer:
With the changes in atmospheric pressure, the gas laws also change accordingly, that is, the Charle's law, the Boyle's law, the Ideal Gas Law, and the Gay-Lussac's law changes with the change in atmospheric pressure. Therefore, the planets, which exhibits the constant value of atmospheric pressure will be more reliable, and those possessing different atmosphere or no atmosphere will be the least reliable.
A 25 L sample of oxygen gas (O2) has a mass of 48 grams and a pressure of 3.0 atm. What would be the temperature of the sample? Reminder: Use the equation PV=nRT, with the constant R = 0.0821 L atm/mol K.
A.
609 K
B.
305 K
C.
19.0 K
D.
1.60 x 10-2 K
Answers
The temperature of the oxygen gas sample is 609 K, which is approximately 336°C or 637°F. The answer is A.
We can use the ideal gas law equation, PV = nRT, to solve for the temperature of the oxygen gas sample.
First, we need to calculate the number of moles of oxygen gas present in the sample using its mass and molar mass:
n = m/M
where:
n = number of moles
m = mass (in grams)
M = molar mass (in g/mol)
The molar mass of oxygen gas (O2) is 32.00 g/mol.
n = 48 g / 32.00 g/mol = 1.50 mol
Next, we can rearrange the ideal gas law equation to solve for temperature (T):
T = (PV) / (nR)
where:
T = temperature (in Kelvin)
P = pressure (in atm)
V = volume (in liters)
n = number of moles
R = gas constant (0.0821 L atm/mol K)
Plugging in the given values, we get:
T = (3.0 atm x 25 L) / (1.50 mol x 0.0821 L atm/mol K)
T = 609 K
For more question on temperature click on
https://brainly.com/question/4735135
#SPJ11
if a sample of gas at 25.2 c has a volume of 536mL at 637 torr, what will its volume be if the pressure is increased to 712 torr?
Answers
PV= RTn
T= 25.2°C = 298.2 K
P1= 637 torr = 0.8382 atm
V1= 536 mL = 0.536 L
:. R=0.082 atm.L/K.mol
:. n= (P1V1)/(RT) = ((0.8382 atm) x (0.536 L))/
((0.082 atmL/Kmol) x (298.2K))
:. n= O.0184 mol
Then,
P2= 712 torr = 0.936842 atm
V2 = RTn/P2 = [(0.082atmL/
Kmol) x (298.2K) x (0.0184mol) ]/(0.936842atm)
:.V2 = 0.4796 L
OR
V2 = 479.6 ml