Answers
Answer:
H₂²⁺(aq) + O₂²⁻(aq) + SO₃²⁻(aq) → SO²⁻₄(aq) + H₂O(l)
Explanation:
H₂²⁺(aq) + O₂²⁻(aq) + Mg²⁺(aq) + SO₃²⁻(aq) → Mg²⁺(aq) + SO²⁻₄(aq) + H₂O(l)
A careful observation of the equation above, shows that the equation is already balanced.
To obtain the net ionic equation, we simply cancel Mg²⁺ from both side of the equation as shown below:
H₂²⁺(aq) + O₂²⁻(aq) + SO₃²⁻(aq) → SO²⁻₄(aq) + H₂O(l)
Related Questions
Does iron always have a charge of 2+ when it forms an ion
Answers
Answer:
Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron(II) has a 2+ charge; iron(III) a 3+ charge.
Explanation:
3. Red light has a frequency of 401 THz. What is the wavelength?
Answers
A light wave in a medium has a wavelength of 5107 m and a frequency of 41014 Hz.
What wavelength is red light at 740 nm?Its frequencies range from 4 to 8 hertz (Hz), or cycles per second, and its wavelengths range from 380 nanometers (nm), or 1.5 nm, to 740 nm, or 2.9 to 105 inches.
Which seven electromagnetic waves are there, from smallest to largest?The parts of the electromagnetic spectrum are designated as follows, from highest to lowest energy: gamma rays, X-rays, ultraviolet radiation, visible light, infrared radiation, and radio waves. The radio wave region of the electromagnetic spectrum includes microwaves (such as those found in microwave ovens).
To know more about red light frequency visit:-
https://brainly.com/question/13104367
#SPJ4
On a long bus trip, a traveler does
not drink
any water for several hours. How
will the volume of urine she produces that day
compare to the volume on a day when she
drinks several glasses of water? Explain.
Answers
Answer:
The volume will be less.
Explanation:
The volume of urine someone produces varies based on their liquid intake. The more liquid you intake, the more you must urinate (not a direct correlation, but basically).
A template of a Venn diagram representing common and differentiating characteristics of covalent and ionic bonds is shown. Which of the following characteristics can be written only in space C?
Answers
Covalent and ionic bonds refer to atoms joined by their electrons. In covalent bonds, electrons are shared by the involved non-metal atoms. Option 2 is correct. Occurs due to the sharing of electrons between two non-metal atoms.
What are covalent and ionic bonds?
Both of them, covalent and ionic bonds, are chemical bonds that can form between atoms.
Ionic bonds occur between atoms with different electronegativity. When they bind, they transfer electrons from one atom to the other creating ions with opposite charges that attract each other.
Ionic compounds are formed by anions and cations.
• Cations are positive ions derivated from metals.
• Anions are negative ions derivated from non-metals.
The metal atoms share its electrons with the non-metal ones, creating stable configurations. Ionic bonds do not create molecules.
Covalent bonds are formed between atoms share electrons to be more stable. Atoms involved share electrons equally, creating a strong bond between them.
Covalent bonds are usually formed between non-metal atoms.
Option 2 is correct. Occurs due to the sharing of electrons between two non-metal atoms
You can learn more about covalent and ionic bonds at
https://brainly.com/question/19739192
#SPJ1
Complete question
A template of a Venn diagram representing common and differentiating characteristics of covalent and ionic bonds is shown.
Which of the following characteristics can be written only in space C?
On the diagram,
The non-overlapping space on the left is marked A, and belongs to the IONIC BOND side of the diagram.The overlapping space is marked B The non-overlapping space on the right is marked C, and belongs to the COVALENT BOND side of the diagram.Options,
Formed between positively and negatively charged ionsOccurs due to the sharing of electrons between two non-metal atomsOccurs in substances that are mostly solids at normal temperature and pressureFormed between an atom with very high electronegativity and an atom with very low electronegativityA 16.42 mL volume of 0.1327 M KMnO4 solution is needed to oxidize 25.00 mL of a FeSO4 solution in acidic medium. What is the concentration of the FeSO4 solution in molarity?
Answers
The concentration of the FeSO4 solution in molarity would be 0.4356
The concentration can be derived first by writing out the balanced equation of the reaction:
\(5Fe^2^++MnO^4^-+8H^+--->5Fe^3^++Mn^2^++4H_2O\)From the balanced equation: for every 1 mole of KMnO4, 5 moles of FeSO4 is needed for complete oxidation.
Recall that: mole = molarity x volume
Mole of KMnO4 needed = 0.1327 x 0.01642
= 0.002179 mole
1 mole KMnO4 = 5 mole FeSO4
0.002178 mole = 5 x 0.002178/1
= 0.01089 moles of FeSO4
Molarity of FeSO4 = mole/volume
= 0.01089/0.025
= 0.4356 M
More on the calculation of molarity of solutions can be found here: https://brainly.com/question/16587536
What would the molarity of a NaOH solution that was made by taking 45.5 mL of 0.875 M NaOH and then diluting with water to a total volume of 500 mL?
Answers
0.0796M
Explanation;Using the dilution formula:
\(C_1V_1=C_2V_2\)where:
C1 and C2 are the initial and final concentration
V1 and V2 are the initial and final volume
Given the following parameters
C1 = 0.875M
V1 = 45.5mL
V2 = 500mL
Required
Final concentration C2
Substituting the given parameters into the formula
\(\begin{gathered} C_2=\frac{C_1V_1}{V_2} \\ C_2=\frac{0.875\times45.5mL}{500mL} \\ C_2=\frac{39.8125}{500} \\ C_2=0.0796M \end{gathered}\)Hence the final concentration is 0.0796M
A solution of dispersant is made by taking 15.0 mL of a 50.0 mg/mL solution of Randyne and mixing it with 50.0 mL of water. Calculate the final concentration of the Randyne in this solution, in units of grams per milliliter.
Answers
Answer:
The final concentration of the Randyne in grams per milliliter = 0.011 g/mL
Explanation:
As we know
C1V1 = C2V2
C1 and C2 = concentration of solution 1 and 2 respectively
V1 and V2 = Volume of solution 1 and 2 respectively
Substituting the given values, we get -
\(50 * 15 = X * (15+50)\\X = 11.54\) mg/mL
The final concentration of the Randyne in grams per milliliter = 0.011 g/mL
The final concentration of the Randyne in this solution is 0.01 g /mL.
How to calculate dilutions?It is very important to know the dilution methods in a chemistry lab. The dilution from the stock solution can be prepared by using the formula,
\(C_1V_1 = C_2V_2\)
Where,
\(C_1\)- concentration of the stock solution
\(V_1\) - the volume of the stock solution
\(C_2\) - concentration of the diluted solution
\(V_2\) - the volume of diluted solution
Put the values in the formula,
\(50 \times 15 = C_2 \times 75 \\\\C_2 = \dfrac {750}{75 }\\\\C_2 = 10{\rm \ mg/mL \ \ \ or} \\\\ C_2 = 0.01 \rm \ g/mL\)
Therefore, the final concentration of the Randyne in this solution is 0.01 g /mL.
Learn more about dilution methods:
https://brainly.com/question/25307719
expaln the conductivity of magnesium
Answers
Magnesium is not a good conductor. Hence, the conductivity of magnesium is not good.
Magnesium is placed in group 2 of the periodic table. It has 2 valence electrons which are in the fully filled s-subshell. When the subshell is fully filled, the electrons are not available to move to conduct. For example, group 1 has half filled subshell which allows the electrons to move.
But, how does magnesium conduct? As its subshell is fully filled and therefore no electrons are available to move.
In group 2 metals, there is a property called s-p hybridization. The s- and p- subshells overlap so that metals can now move to the unfilled states of the p-subshell.
Hence, magnesium is not a good conductor.
To learn more about magnesium, visit: https://brainly.com/question/488824
#SPJ9
871g of sodium chloride is how many moles
Answers
Answer:
14.9 mol
Explanation:
To find the number of moles in a given mass of a sample of sodium chloride (NaCl), we can multiply the number of grams in the sample by the molar mass of sodium chloride, which is 58.44 g/mol.
871 g × (1 mol / 58.44 g)
= 871/58.44 mol
≈ 14.9 mol
Note that we rounded to 3 significant figures in the final answer because that is how many significant figures were given in the mass measurement of the sodium chloride sample.
When a substance is changing state, temperature stays the same at the melting and boiling point is this true or false
Answers
Answer:
True
Explanation:
hope this helps :)
Math the Definitions! Please & Thank You :) Screenshot Attached.
SOMEONE HELP!
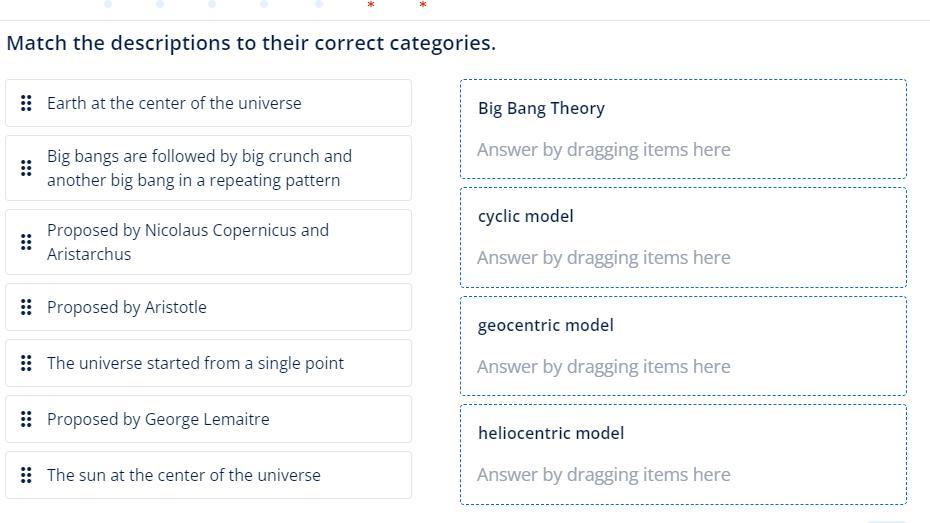
Answers
After analysing the given options and data we conclude that the matching of the given options as
1) Proposed by Nicolaus Copernicus and Aristarchus - heliocentric model
2) Proposed by Aristotle - 3) geocentric model
3) geocentric model - A) Earth at the center of the universe
4) The universe started from a single point - C) Big Bang Theory
5) Proposed by George Lemaitre - C) Big Bang Theory
6) The sun at the center of the universe - 3) geocentric model
7) heliocentric model - 6) The sun at the center of the universe
The Big Bang Theory is a scientific explanation regarding the original creation of the universe. The theory is based on several key assumptions, one of which is the isotropy of the universe. This assumption projects that the universe, more or less, appears the same in all directions of time and space.
Another key theory is that of cosmic inflation, which projects why the universe is so symmetrical on such a significant scale.
To learn more about Big bang theory
https://brainly.com/question/6841128
#SPJ1
Which type of plate boundary does not involve plates moving towards or away from each other, but sliding past each other?
A. Conservative
B. Convergent destructive (subduction)
C. Constructive
D. Convergent collision
Answers
Answer:
B - convergent destructive (subduction)
Explanation:
option A is not a plate boundary. A constructive plate boundary moves away from eachother and a convergent collision boundary would suggest no slipping whereas a convergent destructive boundary has subduction take place which is when a denser plate slips under a less dense plate.
Hypothesis II: Write the equation with Iron (III) Chloride and balance it: Iron + Copper (II) chloride --> Iron (III) chloride + Copper
Answers
Answer:
Fe + CuCl2 = FeCl2 + Cu
Explanation:
This is already balanced.
What happened during the fusion reaction shown? 2H Зн 4He neutron O A. Two H nuclei fused into one He nucleus. O B. One He nucleus split into two H nuclei. O C. The nuclei that fused lost all their neutrons. D. The nuclei that fused lost some of their protons.

Answers
Answer: C.
Explanation:
two H nuclei fused into one He nucleus
Answer: A!
Explanation: just did it

What are some convincing reasons for researcher's to choose your biome ( P.S. this is for a project) pls reply as soon as you can
Answers
Choosing a biome for a research project can provide a wealth of opportunities to better understand the natural world and contribute to important conservation and management efforts.
Why is biome important in research?There are several convincing reasons for researchers to choose a particular biome for their project, some of which include:
Biodiversity: Biomes are characterized by their unique plant and animal species, which can provide a diverse range of research opportunities, including studying their adaptations, behaviors, and interactions.
Climate Change: Biomes are also influenced by climate change, making them an important area of research to better understand how these ecosystems are being affected by changes in temperature and precipitation patterns.
Human Impacts: Human activities, such as deforestation, agriculture, and urbanization, can have a significant impact on biomes. Studying these impacts can help researchers better understand how to mitigate and manage these effects.
Biogeochemical Cycles: Biomes are also important for studying biogeochemical cycles, such as the carbon and nitrogen cycles, which are essential for sustaining life on Earth.
Conservation: Finally, studying biomes can also contribute to conservation efforts, helping to preserve these unique ecosystems and the species that inhabit them.
Find out more on biome here: https://brainly.com/question/2096327
#SPJ1
Once alchol is in the bloodstrram it will reach the brain I'm a few
Answers
Once alcohol is in the bloodstream it will reach the brain in a few seconds to minutes, depending on various factors such as the amount and concentration of alcohol consumed, body weight, metabolism, and other individual factors.
Alcohol's Effects on BrainAlcohol can swiftly cross the blood-brain barrier after it is ingested, having an impact on the brain and neurological system. Depending on the quantity and frequency of drinking, alcohol's effects on the brain can range from minor disturbances in judgment and coordination to more serious consequences including loss of consciousness and, in the worst circumstances, death.
Long-term changes in brain structure and function, such as cognitive impairment and a higher chance of developing specific neurological and mental illnesses, can also result from chronic alcohol consumption.
Once alcohol is in the bloodstream it will reach the brain in a few seconds to minutes, depending on various factors such as the amount and concentration of alcohol consumed, body weight, metabolism, and other individual factors.
Learn more on effects of alcohol on the body here https://brainly.com/question/6511905
#SPJ1
Suppose 3.005 g of a nonvolatile solute is added to 20.02 g of water (the solvent), and the boiling point increases from 100.000 OC to 101.286 OC. Determine the TB, molality, moles, and molecular weight for the solute if kb for water is 0.512 OC/m. Report each value using the correct number of significant digits. Refer to Example 1.2 and pages 3-4 in the chapter 1 notes for general chemistry 1 to understand significant figures.
Answers
Answer:
* \(\Delta T_B=1.286\°C\)
* \(m=2.5m\)
* \(n=0.05mol\)
* \(M=59.76g/mol\)
Explanation:
Hello,
In this case, considering the boiling point rise problem, we consider its appropriate equation:
\(\Delta T_B=imK_b\)
Whereas i is the van't Hoff factor that for this nonvolatile solute is 1, m is the molality, Kb the boiling point constant of water as it is the solvent and ΔT the temperature difference. In such a way, with the given information we obtain:
- ΔT:
\(\Delta T_B=101.286\°C-100.000\°C\\\\\Delta T_B=1.286\°C\)
- Molality (mol/kg):
\(m=\frac{\Delta T_B}{i*K_b}=\frac{1.286\°C}{1*0.512\°C/m}\\ \\m=2.5m\)
- Moles for 20.02 g (0.02002 kg) of water:
\(n=2.5mol/kg*0.02002kg\\\\n=0.05mol\)
- Molar mass:
\(M=\frac{mass}{moles}=\frac{3.005g}{0.050mol} \\\\M=59.76g/mol\)
Best regards.
Use the drop-down menus to complete each sentence.
A
is an organism that eats other organisms to gain energy; it is also called
.
A
is an organism that makes its own food; it is also called
.
A
is an organism that breaks down waste and dead organisms.
Answers
Answer:
A consumer is an organism that eats other organisms to gain energy; it is also called a heterotroph.
A producer is an organism that makes its own food; it is also called an autotroph.
A decomposer is an organism that breaks down waste and dead organisms.
Explanation:
Green plants are examples of autotrophs that make their own food by photosynthesis - they don't rely on other organisms to provide them with energy.
In contrast, many organisms rely on other organisms as an energy source. These organisms are called consumers or heterotrophs. They consume producers and other consumers.
Decomposers such as some fungi, bacteria, and certain insects break down dead or decaying biological material and recycle the nutrients back into the soil.
Answer:consumer a heterotroph producer an autotroph decomposer
Explanation:
Four bonding pairs around a central atom results in a ________.
Answers
Do you think there’s an advantage of process 1 over process 2 for this species?
Answers
Answer:
witch one witch species witch bone of 1 and 2
Answer:
Yes, process 1 has an advantage over process 2 for the planarian worm. Process 1 requires only one parent. If the planarian can’t find a mate, the life cycle will continue because it can still reproduce.
Explanation:
i did it
why do different types of matter have different requirements to phase change?
Answers
Different types of matter have different requirements because the pressure and temperature of a substance's environment dictate its phase.
Matter is anything that occupies space and has mass. The states or phases of matter include solids, liquids, gases, and plasma. Any material in a form that has a distinct shape and volume is referred to as a solid. Phase change refers to a shift in the condition of matter. Phase shifts can be carried out by people and occur frequently in nature.
A set of molecules will experience various movements, such as vibrations, rotations, and translations, depending on the average temperature in their vicinity.
To know more about the States of matter, refer to this:
brainly.com/question/20348735
How does removing thermal energy from liquid water affect the molecules in the water?
Answers
Answer:
For deposition to happen, thermal energy must be removed from the gas. ... As water vapor loses thermal energy, it changes into solid frost. States of Water. Water is the only substance that exists naturally as a solid, a liquid, and a gas within Earth's temperature range.
Explanation:
HELP! An atom of vanadium
(V)
has 23 protons. It has a net charge of 0.
How many electrons does it have?
1. 23
2. 24
3. 25
4. 26
Answers
An atom of vanadium (V) has 23 electrons.
Given :
A vanadium (V) atom with 23 protons and has a net charge of 0.
To find:
The number of electrons in a vanadium atom
Solution:
Number of protons in vanadium atom = 23
The 0 net charge on the atom indicates that the atom has an equal number of protons and electrons which makes it a neutral atom.
Number of electrons in vanadium atom = Number of protons in vanadium atom = 23
An atom of vanadium (V) has 23 electrons.
Learn more about subatomic particles here:
brainly.com/question/7221719?referrer=searchResults
brainly.com/question/489853?referrer=searchResults
a. How many moles of ammonium sulfate will be produced with 25.56 grams of ammonia gas
react completely?
Answers
What variable must be held constant to use the combined gas law?
a. mass of gas sample
b. number of moles of gas
c. number of gas molecules
d. all of the above
e. none of the above
Answers
Answer:
The answer is (e) none of the above. The combined gas law relates the pressure, volume, and temperature of a gas, so all three variables (pressure, volume, and temperature) must be considered and at least one of them should be held constant to use the combined gas law. The mass of the gas sample, number of moles of gas, and number of gas molecules are not directly related to the combined gas law.
An atomic cation with a charge of +1 has the following electron configuration:
1s 2s 2p 3s 3p 3d ¹4s¹
What is the chemical symbol for the ion?
How many electrons does the ion have?
How many 3p electrons are in the ion?
Answers
A) The chemical symbol for the ion is Fe+
B) It has 20 electrons in total, and there are 6 3p electrons in the ion.
C) There are 6 electrons present in the 3p orbital.
The atomic cation with the given electron configuration is represented by the chemical symbol Fe+.
To determine the number of electrons in the ion, we need to count the electrons present in the electron configuration. In the given configuration, we can see that the 1s orbital has 2 electrons, the 2s orbital has 2 electrons, the 2p orbital has 6 electrons, the 3s orbital has 2 electrons, the 3p orbital has 6 electrons, the 3d orbital has 1 electron, and the 4s orbital has 1 electron. Adding up these numbers, we have:
2 + 2 + 6 + 2 + 6 + 1 + 1 = 20
Therefore, the ion has 20 electrons.
To determine the number of 3p electrons in the ion, we look at the 3p orbital. In this case, there are 6 electrons present in the 3p orbital.
In summary, the chemical symbol for the ion is Fe+, it has 20 electrons in total, and there are 6 3p electrons in the ion.
For more question on electrons
https://brainly.com/question/26084288
#SPJ8
A mixture of 65 percent N2 and 35 percent CO2 gases (on a mass basis) enters the nozzle of a turbojet engine at 60 psia and 1400 R with a low velocity, and it expands to a pressure of 12 psia. If the isentropic efficiency of the nozzle is 88 percent, determine:
(a) the exit temperature
(b) the exit velocity of the mixture.
Assume constant specific heats at room temperature.
Answers
Answer:
a. 969.1 R
b. 2237 ft/s
Explanation:
First the apparent specific heats are determined from the mass fractions of the gases:
\(c_{p} &=\left(\mathrm{mf} c_{p}\right)_{\mathrm{N}_{2}}+\left(\mathrm{mf} c_{p}\right) \mathrm{CO}_{2} \\ &=(0.65 \cdot 0.248+0.35 \cdot 0.203) \frac{\mathrm{Btu}}{\mathrm{lbm} \mathrm{R}} \\ &=0.232 \frac{\mathrm{Btu}}{\mathrm{lbmR}} \\ c_{v} &=\left(\mathrm{mf} c_{v}\right)_{\mathrm{N}_{2}}+\left(\mathrm{mf} c_{v}\right)_{\mathrm{CO}_{2}} \\ &=(0.65 \cdot 0.177+0.35 \cdot 0.158) \frac{\mathrm{Btu}}{\mathrm{lbmR}} \\ &=0.170 \frac{\mathrm{Btu}}{\mathrm{lbmR}}\)
The isentropic coefficient then is:
\(k &=\frac{c_{p}}{c_{v}} \\ &=\frac{0.232}{0.17} \\ &=1.365\)
The final temperature is determined from the isentropic nozzle efficiency relation:
\(T_{2} &=T_{1}-\eta_{N}\left(T_{1}-T_{2 s}\right) \\ &=T_{1}\left(1-\eta_{N}\left(1-\left(\frac{P_{2}}{P_{1}}\right)^{(k-1) / k}\right)\right) \\ &=1400\left(1-0.88\left(1-\left(\frac{800}{100}\right)^{(1.365-1) / 1.365}\right)\right) \mathrm{R} \\ &=969.1 \mathrm{R}\)
b. The outlet velocity is determined from the energy balance:
\(h_{1} &+\frac{v_{1}^{2}}{2}=h_{2}+\frac{v_{2}^{2}}{2} \\ v_{2} &=\sqrt{2 c_{p}\left(T_{1}-T_{2}\right)} \\ &=\sqrt{2 \cdot 0.232(1400-969.2) \cdot 25037} \frac{\mathrm{ft}}{\mathrm{s}} \\ &=2237 \frac{\mathrm{ft}}{\mathrm{s}}\)
Determine the mass fraction of hydrochloric acid obtained by merging 100 cm3 of a solution of hydrochloric acid with a mass fraction of HCl 36% (density 1.180 g/cm3) and 50 cm3 of a solution of hydrochloric acid with a mass fraction of HCl 20% (density 1.100 g/cm3)
Answers
Answer:
Solution:
1. Calculate the total volume of the two solutions:
Volume 1 = 100 cm3
Volume 2 = 50 cm3
Total Volume = 150 cm3
2. Calculate the total mass of each solution:
Mass of Solution 1 = (Volume x Density) x Mass Fraction HCl
= (100 cm3 x 1.180 g/cm3) x 0.36
= 43.2 g
Mass of Solution 2 = (Volume x Density) x Mass Fraction HCl
= (50 cm3 x 1.100 g/cm3) x 0.20
= 11 g
3. Calculate the total mass of the two solutions combined:
Total Mass = 43.2 g + 11 g
= 54.2 g
4. Calculate the mass fraction of hydrochloric acid in the new solution:
Mass Fraction HCl = Total Mass of HCl / Total Mass of Solutions
= 54.2 g / (43.2 g + 11 g)
= 0.55
Therefore, the mass fraction of hydrochloric acid obtained by merging 100 cm3 of a solution of hydrochloric acid with a mass fraction of HCl 36%, and 50 cm3 of a solution of hydrochloric acid with a mass fraction of HCl 20%, is 0.55.
What processes are related to metamorphism
Answers
Answer:
Metamorphism is the change of minerals or geologic texture (distinct arrangement of minerals) in pre-existing rocks (protoliths), without the protolith melting into liquid magma (a solid-state change). The change occurs primarily due to heat, pressure, and the introduction of chemically active fluids.
Explanation:
i hope this helps :)
How does matter and energy cycle through an ecosystem?
a. Cellular respiration alone
b. The atmosphere
c. Photosynthesis and cellular respiration
d. Photosynthesis alone
Please help D;
Answers
Your welcome