Answers
It would be more accurate to say that all stars produce some elements, but not all elements are produced by all stars. While it is true that many elements are produced by stars through the process of nuclear fusion, not all elements are created by all stars.
For example, the heaviest elements such as gold and platinum are formed through a process called neutron capture, which occurs during supernova explosions or in the collision of neutron stars.
Additionally, some stars may not be massive enough or may not live long enough to produce certain elements. Therefore, it would be more accurate to say that all stars produce some elements, but not all elements are produced by all stars.
To know more about nuclear fusion, visit:
https://brainly.com/question/12701636
#SPJ1
Related Questions
A serving of a particular fruit dessert contains 15.0 g of sugar. If all the sugar is sucrose, C12H22O11 (molar mass = 342), how many molecules of sugar are present in this serving?
Answers
There are 2.64 × 1022 molecules of sucrose present in this serving of fruit dessert containing 15.0 g of sugar.
To determine the number of molecules of sugar present in the serving, we need to calculate the number of moles of sugar and then convert it to the number of molecules.
Given:
Mass of sugar (sucrose) = 15.0 g
Molar mass of sucrose (C12H22O11) = 342 g/mol
First, calculate the number of moles of sugar using the formula:
Number of moles = Mass of substance / Molar mass
Number of moles of sugar = 15.0 g / 342 g/mol ≈ 0.0439 mol
Next, we use Avogadro's number, which states that there are approximately 6.022 × 10^23 molecules in one mole of a substance. Therefore, to find the number of molecules of sugar:
Number of molecules = Number of moles × Avogadro's number
Number of molecules of sugar = 0.0439 mol × 6.022 × 10^23 molecules/mol ≈ 2.64 × 10^22 molecules
Therefore, there are approximately 2.64 × 10^22 molecules of sugar present in this serving.
for more such question on molecules
https://brainly.com/question/24191825
#SPJ8
Use the chemical equation to answer the question. 2Ag(s) + H₂S(g) → Ag₂S(s) + H₂(g) The molar mass of silver (Ag) is 108 g/mol. The molar mass of sulfur (S) is 32 g/mol. The reaction uses 0.04 mol of silver. Which steps show how to determine the mass of silver sulfide (Ag₂S) produced in the reaction?
Answers
As per the given balanced reaction, 2 moles of silver gives 1 mole of the product silver sulfide. Hence, 0.04 moles will given 0.02 moles of silver sulfide.
What is theoretical yield of a reaction?The theoretical yield of a reaction is the amount of product obtained based on the balanced chemical equation of the reaction. However, the actual yield might vary from the the theoretical value.
The given reaction of Ag and H₂S shows that, two moles of Ag produce one mole of the product silver sulfide.
Number of moles of silver sulfide produce by 0.04 moles of Ag is then half of this number that is 0.02 moles of Ag₂S.
Therefore, 0.0 moles of Ag will give 0.02 moles of silver sulfide. Using the same way, we can determine the yield of products in any reaction.
Find more on theoretical yield :
brainly.com/question/11002641
#SPJ1
How much energy is required to heat up a Styrofoam cup from 25oC to 40oC? The mass of the cup is 20 grams and the specific heat capacity of Styrofoam is 1.1 J/(g *oC).
Answers
Answer:
Q = 440 J
Explanation:
Given data:
Heat required = ?
Initial temperature = 25°C
Final temperature = 40°C
Mass in gram = 20 g
Specific heat capacity = 1.1 J/g.°C
Solution:
Formula:
Q = m.c. ΔT
Q = amount of heat absorbed or released
m = mass of given substance
c = specific heat capacity of substance
ΔT = change in temperature
ΔT = 40°C - 20°C
ΔT = 20°C
Q = 20 g×1.1 J/g.°C × 20°C
Q = 440 J
The following precipitation reaction can be used to determine the amount of copper ions dissolved in solution. A chemist added 5.00 × 10−2 L of a solution containing 0.173 mol L−1 Na3PO4(aq) to a 5.00 × 10−2 L sample containing CuCl2(aq). This resulted in a precipitate. The chemist filtered, dried, and weighed the precipitate. If 1.21 g of Cu3(PO4)2(s) were obtained, and assuming no copper ions remained in solution, calculate the following: the concentration of Cu2+(aq) ions in the sample solution. the concentrations of Na+(aq), Cl−(aq), and PO43−(aq) in the reaction solution (supernatant) after the precipitate was removed.
Answers
From the stoichiometry of the reaction, 0.094 g of \(Cu^{2+}\) is contained in the reaction.
What is the amount of the copper ion?The reaction equation is given by; \(2Na_{3}PO_{4} (aq) + 3CuCl_{2} (aq) ----- > 6NaCl (aq) + Cu_{3}(PO_{4}) _{2} (s)\). We must note that the balanced reaction equation is the entry way into obtaining the correct stoichiometry of the reaction in order to solve the problem.
Number of moles of \(Na_{3}PO_{4}\) = 5.00 × 10−2 L * 0.173 mol L−1 = 0.00865 moles
If 2 moles of \(Na_{3}PO_{4}\) produces 1 mole of\(Cu_{3}(PO_{4}) _{2}\)
0.00865 moles of \(Na_{3}PO_{4}\) produces 0.00865 moles * 1 mole/2 moles
= 0.0043 moles of \(Cu3(PO4)2\)
Number of moles of \(Cu^{2+}\) = 0.0043 moles/3 = 0.00144 moles
Mass of \(Cu^{2+}\) = 0.00144 moles * 63.5 g/mol = 0.094 g
Learn more about stoichiometry:https://brainly.com/question/9743981
#SPJ1
What is the nickname for mitochondria
Answers
Answer:
the powerhouse of the cell
a. Identify the structures shown in the diagram. b. Identify the information that is contained within these structures. c. Describe how the structures from this cell would compare to the structures in the nucleus of another body cell from the same person. d. Explain why the structures are in pairs.
Answers
The answer responses to the structures shown in the diagram are:
A. chromosomes
C. They would be the same.
B. They are in pairs because each one comes from a different parent.
What is the structure about?The chromosomes are in pairs because humans have a diploid number of chromosomes, meaning they have two sets of chromosomes, one inherited from each parent.
The nucleus is important in eukaryotic cells and has many important parts that help the cell work properly. There are some parts inside cells called the nuclear membrane, nucleoplasm, nucleolus, and chromatin. Chromatin is made up of DNA and other proteins.
Every part of a person's body has the same genes, but the way they are organized can be different in different types of cells. The chromosomes in our skin cells might not be the same as the chromosomes in our muscle cells, even if they come from the same person.
Learn more about nucleus from
https://brainly.com/question/9376695
#SPJ1
Identify the structures shown.
A. chromosomes
B. mitochondria
C. nuclei
D. vacuoles
C
Describe how the structures from this cell would compare to the structures in the nucleus of another body cell from the same person.
A. There would be longer.
B. They would be shorter.
C. They would be the same.
D. They would be different.
Describe how the structures from this cell would compare to the structures in the nucleus of another body cell from the same person.
A. There would be longer.
B. They would be shorter.
C. They would be the same.
D. They would be different.
Explain why the structures are in pairs.
A. They aren't in pairs.
B. They are in pairs because each one comes from a different parent.
C. This cell is making a copy of itself.
D. The cell always has 2 copies in case 1 is damaged.
How many molecules are in
5.657g H2SO4?
Answers
There are approximately 3.47 x 10²² molecules in 5.657g H₂SO₄.
To calculate the number of molecules in 5.657g H₂SO₄, we need to use the Avogadro's number and the molar mass of H₂SO₄.
The molar mass of H₂SO₄ is 98.079 g/mol.
We need to calculate the number of moles of H₂SO₄:
Number of moles = mass/molar mass
= 5.657g / 98.079 g/mol
= 0.05767 mol.
Then, we can use Avogadro's number, which is 6.022 x 10²³ molecules/mol, to find the number of molecules:
Number of molecules = number of moles x Avogadro's number
= 0.05767 mol x 6.022 x 10²³ molecules/mol
= 3.47 x 10²² molecules
To calculate the number of molecules in a given sample of a substance, you need to use the Avogadro's number, which is 6.022 x 10²³ molecules/mol. This means that one mole of a substance contains 6.022 x 10²³ molecules.
We are given the mass of H₂SO₄, which is 5.657 g. To calculate the number of molecules, we first need to determine the number of moles of H₂SO₄ in the given sample. The molar mass of H₂SO₄ is 98.08 g/mol. So, the number of moles of H₂SO₄ can be calculated as follows:
moles = mass / molar mass
moles = 5.657 g / 98.08 g/mol
moles = 0.0576 mol
Now, we can use the Avogadro's number to determine the number of molecules of H₂SO₄ in 0.0576 moles:
number of molecules = moles x Avogadro's number
number of molecules = 0.0576 mol x 6.022 x 10²³ molecules/mol
number of molecules = 3.47 x 10²² molecules
As a result, in 5.657 g of the material, there are roughly 3.47 x 1022 molecules of H₂SO₄.
To know more about the Molecules, here
https://brainly.com/question/11488454
#SPJ1
Convert .00612 kg to decigrams.
Answers
0.00612(10000) = 61.2 decigrams
Answer should be 61.2 decigrams.
Hope this helps! :)
what is erosion? what are the four agents of erosion
Answers
Answer:
Erosion is the transportation of sediment at the Earth's surface. 4 agents move sediment: Water, Wind, Glaciers, and Mass Wasting (gravity).
Explanation:
Answer:
Erosion is a process where materials are worn away and transported by natural forces such as wind or water
Explanation:
The type of reactions that alkanes undergo is A) electrophilic substitution reactions. B) electrophilic addition reactions. C) free radical substitution reactions. D) free radical addition reactions. E) nucleophilic substitution reactions.
Answers
Answer:
FREE RADICAL SUBSTITUTION REACTIONS
Explanation:
Alkanes undergo substitution reactions with halogens in the presence of ultra-violent light. In substitution reaction, an atom is exchanged with another atom. For exmaple, methane, an alkane reacts with bromine or chlorine( halogens) to form methylbromine or methylchlorine and hydrogen bromide or hydrogen chloride.
This type of substitution reaction is a free radical substitution reaction. In free radical substitution reaction, free radicals are the reactive intermediates and involves two or more steps as the case may be.
The initiation step where the free radicals are produced by homolysis either by heat or ultra-violent ray of light. The free radicals produced are exchanged at the termination step as the produced free radicals recombines with another radical species of the reacting substances.
Methane + Bromine --------> Methylbromine + Hydrogen Bromide
CH4 + Br2 → CH3Br + HBr
You can see there is an exchange of a bromine atom with one hydrogen of methane that is one hydrogen atom in methane is replaced by a bromine atom.
what trends become apparent from the arrangement of electrons in the periodic table
Answers
Answer:
Elements in the same period have the same number of electron shells moving across a period elements gain electrons and protons and become less metallic. This arrangement reflects the periodic recurrence of similar properties as the atomic number increases.
Explanation:
ASAP PLEASE!!!B. Complete the drawing for the sample reaction below to show the law of conservation of
mass, when XY is produced.
+
->
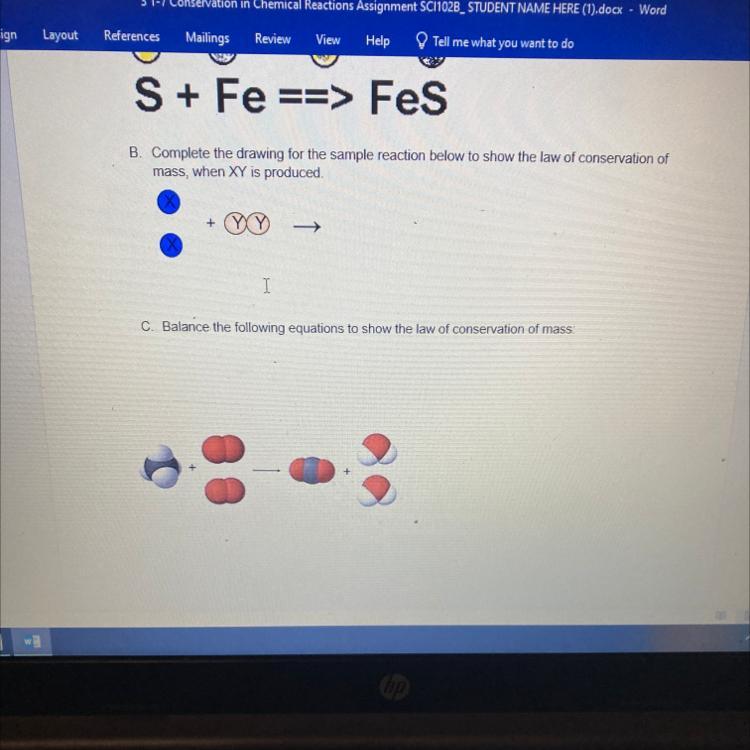
Answers
The complete reaction, according to the law of conservation of mass is:
XX + YY → 2XY
The Law of Conservation is a fundamental principle in chemistry and physics. It states that in a closed system, mass cannot be created or destroyed during a chemical reaction or a physical change. The total mass of the substances involved before the reaction or change must equal the total mass of the substances after the reaction or change.
This principle is based on the understanding that atoms are not created or destroyed, but they can combine or separate to form different substances.
Learn more about the law of mass conservation, here:
https://brainly.com/question/28711001
#SPJ1
03
A force of 20 N acts upon a 5 kg block. Calculate the acceleration of the object.
Answers
(Force = mass x acceleration)
We are going to put each number in the equation:
F=ma
20N = 5kg x (?)
So, to find a (acceleration), we do F/m
[acceleration = force/mass]
a = 20/5 = 4
The answer is 4m/s^2
if you reply with a link, I will report you

Answers
Answer:
1 2 2
Explanation:
HELP HELP HELP HELPPP PLSSSSSS

Answers
what are atoms and molecules and elements In a essay
Answers
Atoms are the smallest indivisible part of an element while molecules are combinations of two or more atoms. See details below.
What are atoms and molecules?Atoms are the smallest possible amount of matter which still retains its identity as a chemical element, now known to consist of a nucleus surrounded by electrons.
Molecules, on the other hand, are the smallest particle of a specific element or compound that retains the chemical properties of that element or compound. They are two or more atoms held together by chemical bonds.
An element is the simplest chemical substance that cannot be decomposed in a chemical reaction or by any chemical means and made up of atoms all having the same number of protons.
Learn more about atoms and molecules at: https://brainly.com/question/7013021
#SPJ1
Select the correct answer.
What are factors of production?
OA.
О в.
O c.
OD.
all the human efforts involved in the production process
all the resources used to produce any goods and services
all the physical tools and equipment used in the production process
all the naturally occurring resources found in land, air, and water
Reset
N

Answers
Answer: B. all the resources used to produce any goods and services
Explanation:
The factors of production are all the resources used to produce any goods and services. This means our answer is option B.
This includes, but is not limited to, land, labor, capital, and entrepreneurship.
Which portion of a molecule of F2O has partial positive charge?
Question 3 options:
A)
The F atoms
B)
The central O atom
C)
The partial charge on each atom is zero
D)
The partial charge on each atom is negative
Answers
The partial charges on each fluorine atom are negative. Option B) The central O atom is the correct answer. Option B
The partial charges in a molecule are determined by the electronegativity values of the atoms involved. Electronegativity is the ability of an atom to attract electrons towards itself in a chemical bond. In the case of \(F_2O\), fluorine (F) is more electronegative than oxygen.
Fluorine is the most electronegative element on the periodic table, meaning it has a high ability to attract electrons. Oxygen is also relatively electronegative but less so than fluorine. When fluorine atoms bond with oxygen, the shared electrons will be pulled more towards the fluorine atoms, creating a polar covalent bond.
In \(F_2O\), each fluorine atom will pull the shared electrons towards itself, resulting in a higher electron density around the fluorine atoms. This creates a region of partial negative charge around the fluorine atoms.
Conversely, the oxygen atom will have a region of lower electron density and, therefore, a partial positive charge. This is because the shared electrons spend more time around the fluorine atoms due to their higher electronegativity.
Option B
For more such question on partial charges visit:
https://brainly.com/question/29974793
#SPJ8
How many moles of Sr2+ are present in 250 mL of a 0.45 M solution of strontium phosphide?
Answers
Moles of Sr²⁺ = 0.3375
Further explanationMolarity is a way to express the concentration of the solution
Molarity shows the number of moles of solute in every 1 liter of solute or mmol in each ml of solution
\(\large{\boxed {\bold {M ~ = ~ \frac {n} {V}}}\)
Where
M = Molarity
n = Number of moles of solute
V = Volume of solution
So to find the number of moles can be expressed as
\(\boxed{\bold{n=V\times M}}\)
Strontium phosphide (Sr₃P₂)
Sr₃P₂ ⇒ 3Sr ²⁺ + 2P³⁻
mol of Sr₃P₂\(\tt 0.45~M \times 0.25~L=0.1125\)
mol of Sr ²⁺mol ratio Sr₃P₂ : Sr ²⁺ = 1 : 3
\(\tt \dfrac{3}{1}\times 0.1125=0.3375\)
A platinum solid weighs 175 g. Look up its density and calculate the volume in dm3 this platinum solid occupies.
Answers
The volume of the platinum solid is determined as 8.16 cm³.
Volume of platinum solid
The volume of the platinum solid is calculated from its density and mass as shown below.
V = m/ρ
where;
V is the volume of the platinum solidm is the mass of the platinum solidρ is the density of the platinum solid = 21.45 g/cm³Volume of the platinum solid is calculated as follows;
V = 175 g / 21.45 g/cm³
V = 8.16 cm³
Thus, the volume of the platinum solid is determined as 8.16 cm³.
Learn more about platinum solid here: https://brainly.com/question/13261338
#SPJ1
calculate the number of moles for the quanity 8.06 x 1021 atoms of Pt
Answers
The number of moles for the quanity 8.06 x\(10_{21\) atoms of Pt is approximately 2.61 grams.
To calculate the number of moles for a given quantity of atoms, we can use Avogadro's number and the molar mass of the element. Avogadro's number is 6.022 x 10²³ atoms/mol.
In this case, you have 8.06 x 10²¹ atoms of Pt. To find the number of moles, divide this quantity by Avogadro's number:
8.06 x 10²¹ atoms Pt / 6.022 x 10²³ atoms/mol = 0.0134 mol Pt
So, there are approximately 0.0134 moles of Pt in 8.06 x 10²¹ atoms of Pt.
The molar mass of Pt (platinum) is 195.08 g/mol. To convert the number of moles to grams, multiply the number of moles by the molar mass:
0.0134 mol Pt x 195.08 g/mol = 2.61 g Pt
Therefore, there are approximately 2.61 grams of Pt in 8.06 x10²¹ atoms of Pt.
In summary, the number of moles for the quantity 8.06 x 10²¹ atoms of Pt is approximately 0.0134 moles. This is equivalent to approximately 2.61 grams of Pt. Remember to use Avogadro's number and the molar mass to perform these calculations accurately.
Know more about moles here:
https://brainly.com/question/29367909
#SPJ8
A beaker containg ice and water is placed on a warm hotplate. Will the ice in the beaker undergo a physical or chemical change?
Answers
Answer: physical
Explanation: water doesn't undergo chemical change when it melts and freezes, because either way frozen or not the molecules are the same. hope i helped ;] BRAINLYEST?????
Who were the terrorists that attacked the united states on September 11, 2001
Answers
As an FDA physiologist, you need 0.625 L of phosphoric acid acid / dihydrogen phosphate (H3PO4 (aq) / H2PO4 - (aq) ) buffer with a pH of 2.75. How do you prepare this solution from stock solutions of 1.0 M H3PO4 (formic acid) and 1.5 M NaOH
Answers
Answer:
0.4058L of 1.0M H3PO4
0.2192L of 1.5M NaOH
Explanation:
The pKa of the H3PO4 / H2PO4- buffer is 2.12
To solve this question we must use H-H equation for this system:
pH = pKa + log [H2PO4-] / [H3PO4]
2.75 = 2.12 + log [H2PO4-] / [H3PO4]
0.63 = log [H2PO4-] / [H3PO4]
4.2658 = [H2PO4-] / [H3PO4] (1)
Where [] could be taken as the moles of each reactant
As you have H3PO4 solution, the reaction with NaOH is:
H3PO4 + NaOH → H2PO4- + Na+ + H2O
As you can see, both H3PO4 and H2PO4- comes from the same 1.0M H3PO4 solution
The moles of H3PO4 are:
[H3PO4] = Moles H3PO4 - Moles NaOH
And for H2PO4-:
[H2PO4-] = Moles NaOH added
Replacing in (1):
4.2658 = [Moles NaOH] / [Moles H3PO4 - Moles NaOH]
4.2658 Moles H3PO4 - 4.2658 moles NaOH = Moles NaOH
4.2658 Moles H3PO4 = 5.2658 moles NaOH (1)
In volume:
0.625L = Moles H3PO4 / 1.0M + Moles NaOH / 1.5M
0.625 = Mol H3PO4 + 0.6667 Moles NaOH (2)
Replacing (2) in (1):
4.2658 Moles H3PO4 = 5.2658 (0.625 - Mol H3PO4 / 0.6667)
4.2658 Moles H3PO4 = 5.2658 (0.625 - Mol H3PO4) / 0.6667
4.2658 Moles H3PO4 = 5.2658*(0.9375 - 1.5 mol H3PO4)
4.2658 Moles H3PO4 = 4.9367 -7.8983 mol H3PO4
12.1641 mol H3PO4 = 4.9367
Mol H3PO4 = 0.4058moles * (1L / 1.0moles) =
0.4058L of 1.0M H3PO4
And:
0.625L - 0.4058L =
0.2192L of 1.5M NaOH
4. A compound has both ionic and covalent bonds if it contains what?chemistry
Answers
Hope this helps! I hope you have a great rest of your day! Also, if you would, please make me brainliest! <3
2.0 moles of H3PO4 equals how many atoms of O
Answers
Considering the definition of compound, chemical formula and Avogadro's Number, there are 4.8184×10²⁴ atoms of O in 2 moles of H₃PO₄.
What is compound and chemical formulaIn a compound there are atoms of different elements joined by forces that we call chemical bonds.
The chemical formula indicates the number and type of different atoms present in the molecule.
Avogadro's NumberAvogadro's Number or Avogadro's Constant is called the number of particles that make up a substance (usually atoms or molecules) and that can be found in the amount of one mole of said substance. Its value is 6.023×10²³ particles per mole. Avogadro's number applies to any substance.
Atoms of OIn this case, in 1 mole of the compound H₃PO₄ you have:
3 moles of H.1 mol of P.4 moles of O.Then, in 2 moles of H₃PO₄ you have 8 moles of O.
Now, you can apply the following rule of three: if by definition of Avogadro's number 1 mole of oxygen contains 6.023×10²³ atoms, 8 moles of oxygen contains how many oxygen atoms?
\(number of atoms=\frac{8 molesx6.023x10^{23}atoms }{1 mole}\)
number of atoms= 4.8184×10²⁴ atoms
Finally, there are 4.8184×10²⁴ atoms of O in 2 moles of H₃PO₄.
Learn more about Avogadro's Number:
brainly.com/question/11907018?referrer=searchResults
brainly.com/question/1445383?referrer=searchResults
brainly.com/question/1528951?referrer=searchResults
#SPJ1
How many grams of H2 would be formed if 34 grams of carbon reacted with an unlimited amount of H2O?
Answers
Answer:
The reaction between carbon (C) and water (H2O) forms carbon monoxide (CO) and hydrogen gas (H2). The balanced chemical equation for this reaction is:
C(s) + H2O(g) -> CO(g) + H2(g)
According to this balanced equation, one mole of carbon reacts with one mole of water to produce one mole of carbon monoxide and one mole of hydrogen gas.
First, calculate the number of moles of carbon in 34 grams. The molar mass of carbon is approximately 12.01 grams/mole.
Moles of carbon = 34 grams / 12.01 grams/mole = 2.831 moles
As the stoichiometry of the reaction shows a 1:1 ratio between carbon and hydrogen, the moles of hydrogen produced would also be 2.831 moles.
The molar mass of hydrogen (H2) is approximately 2 grams/mole.
So, the mass of hydrogen produced = 2.831 moles * 2 grams/mole = 5.662 grams
Therefore, if 34 grams of carbon reacts with an unlimited amount of water, approximately 5.66 grams of hydrogen gas would be formed.
Explanation:
Approximations followed for answer.
Calculate the ionization constant for the following acids or bases from the ionization constant of its conjugate base or conjugate acid: Keep your answer to 2 significant figures (CH3)3NH+
Answers
Answer:
7.41 × 10⁻⁵
Explanation:
Let's consider the basic dissociation reaction of trimethylamine (CH₃)N).
(CH₃)N + H₂O = (CH₃)NH⁺ + OH⁻
According to Brönsted-Lowry, in this reaction (CH₃)N is a base and (CH₃)NH⁺ is its conjugate acid. The pKb for (CH₃)N is 9.87. We can calculate the pKa of (CH₃)NH⁺ using the following expression.
pKa + pKb = 14
pKa = 14 - pKb = 14 - 9.87 = 4.13
Then, we can calculate the acid dissociation constant for (CH₃)NH⁺ using the following expression.
pKa = -log Ka
Ka = antilog - pKa = antilog -4.13 = 7.41 × 10⁻⁵
How did he show that these particles had a charge on them?
Answers
J.J. Thomson discovered electrons and their negative charge through the cathode ray experiment, leading to the development of the plum pudding model of the atom.
J.J. Thomson, a British physicist, was the first to discover electrons in 1897.
He conducted the cathode ray experiment to identify the negatively charged particles.
The cathode ray tube is a vacuum-sealed glass tube with two electrodes at each end: a cathode and an anode.
When a high voltage electrical current is applied to the electrodes, the tube glows, indicating that the cathode rays are being emitted from the cathode and traveling through the tube towards the anode.
The cathode rays were found to have a negative charge, according to Thomson.
These rays were identified as particles by the presence of a magnet, which caused the particles to bend in the direction opposite to the magnet's polarity.
This discovery indicated that the particles had a charge on them because they were deflected by the magnetic field, which is only possible if the particles have an electric charge.
Thomson further concluded that these particles were about 1,000 times smaller than hydrogen atoms because of the degree of deflection they experienced in the magnetic field.
Furthermore, Thomson created the plum pudding model of an atom, in which electrons are dispersed throughout a positively charged matrix, based on his findings.
For more such questions on electrons
https://brainly.com/question/26084288
#SPJ8
Convert 1000mg to centrigrams
Answers
Answer:
100cg
Explanation:
1mg is equal to 0.1 cg