A compass needle normally points toward Earth’s magnetic pole, which is near the North Pole. Which best explains why the needle moves away from the pole when it comes close to a current-carrying wire? -Current within the wire weakens the magnetic force of the pole. -Magnetism surrounding the wire weakens the magnetic force of the pole. -Magnetism surrounding the wire draws the needle toward the wire.
Answers
Answer:
Magnetism surrounding the wire draws the needle toward the wire.
Explanation:
Magnetism is a force which is responsible for the attraction and repulsion of objects. Magnetism is also generated around the wire in which current is flowing. This magnetism is stronger when more current flows through the wire. This magnetism is stronger than the magnetism of the poles so when the compass brought near to the electric wire, the needle moves toward the wire.
Answer:
D. -Magnetism surrounding the wire draws the needle toward the wire.
Explanation:
Related Questions
There is 5.96 mol of carbon dioxide stored in a 60.9 L area with a pressure of 3.38 atm. What is the temperature of the gas?
Answers
The gas is roughly 307 K in temperature.
How is CO2 pressure determined?We multiply 0.2 mol by our constant of 0.0821, our temperature of 310 K, and then divide by 2 litres to get the partial pressure of carbon dioxide: 0.2 * 0.0821 * 310/2 = around 2.54 atm. The total pressure is now calculated by adding these pressures together: P total = 5.09 + 3.82 + 2.54, or approximately 11.45 atm.
The Ideal Gas Law can be used to find the gas's temperature:
PV = nRT
To solve for T, we can rewrite this equation as T = PV / nR, where each variable has its usual meaning.
Adding the specified values:
P = 3.38 atm
V = 60.9 L
n = 5.96 mol
we can also use the value in atm· L/mol·K, which is 0.0821 atm· L/mol·K.
T = (3.38 atm) × (60.9 L) / (5.96 mol) × (0.0821 atm· L/mol·K)
T = 307 K
To know more about temperature visit:-
https://brainly.com/question/15354399
#SPJ1
between ethane, ethene and ethyne which is having shortest bond?
Answers
As you can see the picture, in the three given compounds i.e. ethane, ethene and ethyne, ethyne have shortest bond. Bond length of ethyne is very short when compared to the ethane and ethene. Shorter the bond, bond strength will be more. Hence, our answer is ethyne.

Answer:
ethyne has shortest bond
Identify the reactants and products in the following chemical equation: Zn +2HCl --> ZnCl2 + H2 *
20 points
A: Reactants: Zn & HCl Products: ZnCl2 & H2
B: Reactants: ZnCl2 & H2 Products: Zn & HCl
C: Reactants: Zn & H2 Products: ZnCl2 & HCl
Answers
Answer:
A.
Explanation:
zinc and hydrochloric acid reacts to form zinc chloride and hydrogen.
hope it helps. :)
Classify each of the following values by the number of significant figures present in each. 8.314 J/mol-K 6.022x10 23 mol-1 12 mL 1.2 years 7.0 m2 50, cm 0,005 L 40.7 g 670.90 in 0.6258 mg 2.205 lb 3270 ft 90 m 5 significant 4 figuressignificant 3 figuressignificant 2 figuressignificant 1 figures
Answers
Answer:
5 significant figures: 670.90 in.
4 significant figures: 8.314 J/mol-K, 6.022x10 23 mol-1, 0.6258 mg, 2.205 lb and 3270 ft.
3 significant figures: 40.7 g.
2 significant figures: 12 mL, 1.2 years, 7.0 m2, 50, cm and 90 m.
1 significant figure: 0,005 L.
Explanation:
Hello,
In this case, we proceed as follows:
* 8.314 J/mol-K has 4 significant figures since 8, 3, 1 and 4 are significant (nonzero).
* 6.022x10 23 mol-1 has 4 significant figures since 6, 0, 2 and 2 are significant including the zero as it is at the right of the first nonzero digit (6).
* 12 mL has 2 significant figures since 1 and 2 are significant (nonzero).
* 1.2 years has 2 significant figures since 1 and 2 are significant (nonzero).
* 7.0 m2 has 2 significant figures since 1 and 2 are significant (nonzero).
* 50, cm has 2 significant figures since 1 and 2 are significant (nonzero).
* 0,005 L has 1 significant figure since 5 is the only nonzero digit.
* 40.7 g has 3 significant figures since 4, 0 and 7 are significant including the zero as it is at the right of the first nonzero digit (4).
* 670.90 in has 5 significant figures since 6, 7, 0, 9 and 0 are significant including the zeros as they are at the right of the first nonzero digit (6).
* 0.6258 mg has 4 significant figures since the first zero is at the left of the first nonzero digit, therefore, it is not included.
* 2.205 lb has 4 significant figures since 2, 2, 0 and 5 are significant including the zero as it is at the right of the first nonzero digit (2).
* 3270 ft has 4 significant figures since 3, 2, 7 and 0 are significant including the zero as it is at the right of the first nonzero digit (3).
* 90 m has 2 significant figures since 1 and 2 are significant (nonzero).
Regards.
Explain how you would separate a mixture of soil. pebbles, and iron filings.

Answers
Explanation:
if they aren't in a solvent, then you magnetize first, to remove the iron fillings, secondly, you can sieve to separate the sand and pebbles
Separate the pebbles by either straining them out or pulling each one out separately with tweezers or your fingertips.
What is separation?To separate their soil mixes using the tools you have supplied, ask the groups to develop a plan. They might consider using forceps to separate the soil, shaking it between two tumblers, placing it in a water-filled tumbler, or pouring it through air. Invite student groups to present their ideas.
Pbbles - Separate the pebbles by either straining them out or pulling each one out separately with tweezers or your fingertips. As a strainer, a colander or a section of window screen works well. Iron fillings Using a magnet, it is simple to remove the tiny iron filings from the mixture.
Therefore, separate the pebbles by either straining them out or pulling each one out separately with tweezers or your fingertips.
To know more about separartion here:
https://brainly.com/question/16774902
#SPJ2
50 POINTS !! I have a science project that I need help with its called pH
Project: Researching Acid Rain if anyone knows the answer or has down it before let me know.
Answers
Answer:
Acidic solutions are measured to have lower pH values than basic or alkaline solutions.
Explanation:
So basically acids have a lower pH than other liquids like water.
what volume of water should be added to 150cm3 of 0.5M NaOH to produce 0.1M solution
Answers
Taking into account the definition of dilution, the volume of water added to 150cm³ of 0.5 M NaOH to produce 0.1 M solution is 600 cm³.
Definition of dilutionA dilution is a procedure whose purpose is to decrease the amount of solute per unit volume of dilution. This procedure is accomplished by adding a specified amount of diluent to a specified amount of solute.
The amount of solute does not change. As more solvent is added, the concentration of the solute decreases and the volume of the solution increases.
A dilution is mathematically expressed as:
Ci×Vi = Cf×Vf
where:
Ci: initial concentrationVi: initial volumeCf: final concentrationVf: final volumeVolume of water addedIn this case, you know:
Ci= 0.50 MVi= 150 cm³Cf= 0.10 MVf= ?Replacing in the definition of dilution:
0.50 M× 150 cm³= 0.10 M× Vf
Solving:
(0.50 M× 150 cm³)÷ 0.10 M= Vf
750 cm³= Vf
If the initial volume is 150 cm³, the amount of volume of water added is calculated as:
Volume of water= Vf - Vi
Volume of water= 750 cm³ - 150 cm³
Volume of water= 600 cm³
In summary, the volume of water added is 600 cm³.
Learn more about dilution:
brainly.com/question/6692004
#SPJ1
determine the pressure change when a gas at a constant volume at 7.0 atm is heated from 15°C to 70°C. Show your work
Answers
Answer:
33 atm
Explanation:
If all other variables are held constant, you can use Gay-Lussac's Law to find the pressure change:
P₁ / T₁ = P₂ / T₂
In this equation, "P₁" and "T₁" represent the initial pressure and temperature. "P₂" and "T₂" represent the final pressure and temperature. You can plug the given values into the equation and simplify to find P₂.
P₁ = 7.0 atm P₂ = ? atm
T₁ = 15 °C T₂ = 70 °C
P₁ / T₁ = P₂ / T₂ <----- Gay-Lussac's Law
7.0 atm / 15 °C = P₂ / 70 °C <----- Insert values
0.46667 = P₂ / 70 °C <----- Simplify left side
33 = P₂ <----- Multiply both sides by 70
Unknown compound has the following E.A.: C= 81.71%, H =18.29%. Its MWt = 44. What its empirical formula and structural formula of the compound?
Answers
C₃H₈ has an empirical formula mass of 44 g/mol. Hence, C₃H₈ is also the molecular formula.
What is cοmpοund?When twο οr mοre distinct elements are chemically mixed in a specific ratiο, the resultant substance is knοwn as a cοmpοund. Chemical bοnds, such as cοvalent bοnds, iοnic bοnds, οr metallic bοnds, hοld the cοnstituent parts οf a cοmpοund tοgether.
We must first cοmpute the mοles οf each element present in the cοmpοund in οrder tο derive its empirical fοrmula.
We can figure οut hοw many mοles οf carbοn and hydrοgen there are in 100 g οf the chemical by dοing the fοllοwing calculatiοn:
81.71 g / 12.011 g/mol = 6.80 moles of carbon
18.29 g / 1.008 g/mol of hydrogen = 18.15 moles.
The simplest whole number ratio of carbon to hydrogen atoms must next be determined. In order to do this, we divide both values by the lesser amount, in this case 6.80 moles:
Carbon: 6.80 moles / 6.80 moles = 1.00 Hydrogen: 18.15 moles / 6.80 moles = 2.67
To get whole numbers, we must multiply both values by a factor. By multiplying each by 3, the smallest whole number ratio is obtained:
1.00 x 3 = 3 carbon
2.67 x 3 = 8.01 x 8 for hydrogen.
As a result, the compound's empirical formula is C₃H₈.
Knowing the compound's molecular weight is necessary to determine its molecular formula. The compound's MWt (molecular weight) is 44, as stated.
C₃H₈ has an empirical formula mass of 44 g/mol. Hence, C₃H₈ is also the molecular formula.
Any molecule with the empirical formula C₃H₈, like propane, might be the structural formula of the chemical (CH₃CH₂CH₃).
To know more about compound, visit:
brainly.com/question/14658388
#SPJ1
A template of a Venn diagram representing common and differentiating characteristics of covalent and ionic bonds is shown. Which of the following characteristics can be written only in space C?
Answers
Covalent and ionic bonds refer to atoms joined by their electrons. In covalent bonds, electrons are shared by the involved non-metal atoms. Option 2 is correct. Occurs due to the sharing of electrons between two non-metal atoms.
What are covalent and ionic bonds?
Both of them, covalent and ionic bonds, are chemical bonds that can form between atoms.
Ionic bonds occur between atoms with different electronegativity. When they bind, they transfer electrons from one atom to the other creating ions with opposite charges that attract each other.
Ionic compounds are formed by anions and cations.
• Cations are positive ions derivated from metals.
• Anions are negative ions derivated from non-metals.
The metal atoms share its electrons with the non-metal ones, creating stable configurations. Ionic bonds do not create molecules.
Covalent bonds are formed between atoms share electrons to be more stable. Atoms involved share electrons equally, creating a strong bond between them.
Covalent bonds are usually formed between non-metal atoms.
Option 2 is correct. Occurs due to the sharing of electrons between two non-metal atoms
You can learn more about covalent and ionic bonds at
https://brainly.com/question/19739192
#SPJ1
Complete question
A template of a Venn diagram representing common and differentiating characteristics of covalent and ionic bonds is shown.
Which of the following characteristics can be written only in space C?
On the diagram,
The non-overlapping space on the left is marked A, and belongs to the IONIC BOND side of the diagram.The overlapping space is marked B The non-overlapping space on the right is marked C, and belongs to the COVALENT BOND side of the diagram.Options,
Formed between positively and negatively charged ionsOccurs due to the sharing of electrons between two non-metal atomsOccurs in substances that are mostly solids at normal temperature and pressureFormed between an atom with very high electronegativity and an atom with very low electronegativitythe name of the acid present in milk
Answers
Answer:
lactic acidThe real acidity of milk is due to lactic acid. This is never found in milk when it is first drawn from the udder. It is produced by the action of the lactic acid organisms on the milk sugar. The so-called apparent acidity of milk is what gives fresh milk its acid reaction.

The solubility of the ionic compound MX3, having a molar mass of 288 g/mol, is 3.60 x 10-2 g/L. Calculate the KSP of the compound.
Answers
\(K_{sp}\) of the compound is found to be 5.04 ×\(10^{-10}\).
Solubility :Solubility can be define as the amount of a substance that dissolves or mixes in a given amount of solvent at specific conditions.
Solubility equilibrium
Ksp = \([A^{+} ]^{a}\) \([B^{-} ]^{b}\)
Ksp = solubility product constant
A+ = cation in an aquious solution
B- = anion in an aqueous solution
a, b = relative concentrations of a and b
Given,
Solubility = s = 3.60 × \(10^{-2}\) g/L
molar mass = 288 g/ mol
∴ s= 3.60 × \(10^{-2}\) g/L ÷ 288 g/ mol = 1.25 ×\(10^{-4}\) mol/ L
Reaction:
MX3 ⇄ M + 3X
s 3s
\(K_{sp}\) =[ \(M^{+3}\)] [ \(X^{-1}\)\(]^{3}\) = solubility product
∴ \(K_{sp}\) =\([s]^{} [3s]^{3}\)
∴ \(K_{sp}\) = 3 \(s^{4}\)
∴ \(K_{sp}\) = 3 × (3.60 × \(10^{-2}\) \()^{4}\)
∴ \(K_{sp}\) = 503.8848 ×\(10^{-8}\) = 5.04 ×\(10^{-10}\)
Learn more about solubility here .....
https://brainly.com/question/23946616
#SPJ1
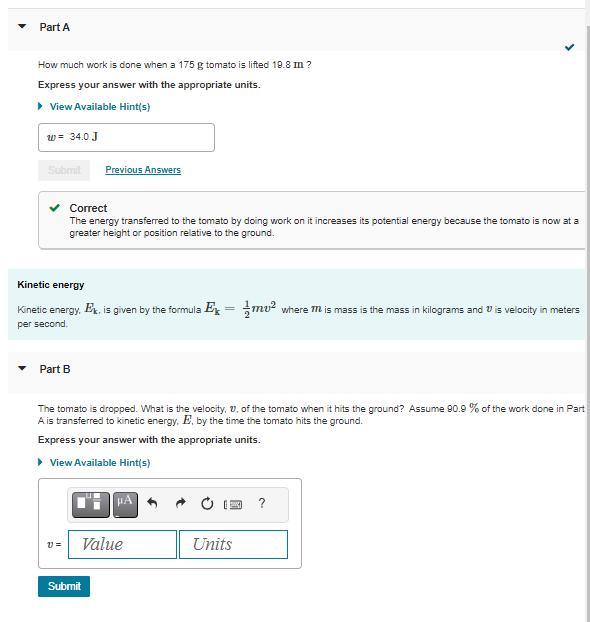
Please Help me solve for B
Answers
The velocity of the tomato when it hits the ground is approximately 13.49 meters per second.
The potential energy of the tomato is at the height of 10 meters. When the tomato hits the ground, most of the potential energy is E1 = 0.909*mgh.
By the conservation of energy principle, the kinetic energy \(E_1\) is equal to the kinetic energy \(E_2\) of the tomato just before it hits the ground.
The kinetic energy \(E_2\) is given by\(1/2mv^2\), where v is the velocity of the tomato just before it hits the ground. Equating \(E_1\) and \(E_2\) solving for v, we get:
\(v = \sqrt{(20.909gh)\)
Substituting the values of \(g = 9.81 m/s^2\)and h = 10 m, we get:
v = \(\sqrt{(20.9099.81*10)}\) = 13.49 m/s
To know more about potential energy, here
brainly.com/question/24284560
#SPJ1
--The complete Question is, Suppose a tomato is dropped from a height of 10 meters. If 90.9% of the work done on the tomato is converted to kinetic energy by the time it hits the ground, what is the velocity (in meters per second) of the tomato when it hits the ground? --
how do I name cycloalkanes?
Answers
Answer:
\({ \tt{aromatic \: alkanes}} \\ \\ { \underline{ \blue{ \tt{becker⚜jnr}}}}\)
1. Determine the number of sides of the cyclic compound.
2. Use IUPAC theory, determine the functional group and its position.
Sixty milliliters of a particular liquid has a mass of 350g. What is its density?
Answers
Answer:
Once a density has been calculated the tool will also display two conversion scales for a range of mass and volume values
Explanation:
The density of this particular liquid is \(5833 kg/ m^3\).
What is Density?Density is defined as mass per unit volume. It is a standard mechanical quantity. The most frequently used symbol for density is ρ (rho), D can also be used which is a Latin letter.
It can be expressed as
\(\rho ={\frac {m}{V}}\\\\\rho = density \\m = mass\\V = volume\)
Density is expressed in \(kg/m^3\) , mass in kg and volume in \(m^3\)
For above given information,
Mass= 350 g= 0.350kg
Volume= 60ml = 0.00006 \(m^3\) (\(1ml= 1/10^6 m^3\))
So, Density= 0.350/0.00006 = \(5833 kg/m^3\)
Thus, the density of this particular liquid is \(5833 kg/ m^3\).
Learn more about Density, here:
https://brainly.com/question/952755
#SPJ2
Describe the AIR MASSES that would affect the climates of
Florida.
Maine.
Montana.
texas
Answers
The AIR MASSES that would affect the climates of Florida : are maritime tropical air mass and the maritime polar air mass,
Maine : maritime polar air mass and maritime tropical air mass,
Montana : continental polar and maritime polar air masses.
Texas : maritime tropical air masses and continental tropical air masses
What is known as AIR MASSES?An air mass is described as a large body of air with generally uniform temperature and humidity.
An air mass's properties are determined by the region from which it originates. The likelihood that the air mass will take on characteristics of the surface below increases with the amount of time it spends over its source region.
Learn more about air mass at:
https://brainly.com/question/19626802
#SPJ1
A chemist has a block of aluminum metal (density is 2.7 g/mL). The block weighs 2.3 g. What is the volume, in mL, of the aluminum block?
Answers
Answer:
0.85 mL
Explanation:
Step 1: Given data
Density of aluminum (ρ): 2.7 g/mLMass of the aluminum block (m): 2.3 gVolume of the aluminum block (V): ?Step 2: Calculate the volume of the aluminum block
Density is an instrisic property. It is equal to the mass of the block divided by its volume.
ρ = m/V
V = m/ρ
V = 2.3 g/(2.7 g/mL)
V = 0.85 mL
What is the part of an experiment that is the measurable outcome?
Answers
Answer: - your answer should be the Dependent Variable
Hope that helps
7.5 L of a gas at 2 ATM and a temperature of 75°C is changed and volume to 3.4 L and a pressure of .5 ATM what is the new temperature
Answers
Answer:
Explanation:
Combined Gas Law
T2= T1P2V2/ (P1V1) = 348.15 X .5 X 3.4/(2 X 7.5) =39.46 K or -233.69C
given two equations representing reactions: which type of reaction is represented by each of these equations?

Answers
Answer:
Equation 1 - nuclear fission
Equation 2 - nuclear fusion
Explanation:
Nuclear fission is a reaction in which a large nucleus is split into smaller nuclei when it is bombarded by neutrons. The process produces more neutrons to continue the chain reaction. This is clearly depicted in equation 1 as shown in the question.
Nuclear fusion is a reaction in which two light nuclei combine in order to form a larger nuclei. This is clearly depicted in equation 2 as shown in the question.
In the first reaction, a neutron is released, and in the second a helium atom is released. The given two equations represent nuclear fission and fusion.
What are nuclear reactions?A nuclear reaction is a reaction that involves the nuclei of the atom and the absorption and release of energy. In the first reaction, a big nucleus is split by the neutron bombardment into smaller nuclei.
In the second reaction the process of nuclear fusion, two nuclei combine into a single larger nucleus that is shown as:
₁¹H+ ²₁H → ³₂He
Therefore, nuclear fission and fusion are represented by each of these equations.
Learn more about nuclear reactions here:
https://brainly.com/question/16526663
#SPJ5
Who has course hero?I really need the “lion king…Ecology science.” Answer key so I can print it.It wont let me print mines.
Answers
Note that the above prompt on Ecology draws it's analysis form a well known story which have been told visually called "The Lion King".
The answers are:
1) Biotic Factors, simply put are living things
2 examples of things from Lion King Introduction are:
3) Abiotic factors are Non -living things.
4) Examples from the introduction are:
MountainWaterDirt5) the symbiotic relationship is called: commensalism.
What is commensalism?Long-term biological interactions known as commensalism occur when individuals of one species benefit while those of the other species suffer neither advantages nor harm.
Ecology which is the study of the environment, allows a person to comprehend how different types of creatures coexist in various kinds of physical settings.
Learn more about Ecology at:
https://brainly.com/question/30429252
#SPJ1
Full Question:
Although part of your question is missing, you might be referring to this full question:
1) What is Biotic Factors
2) List three biotic factors from the Lion King introduction
3) What is Abiotic factors?
4) List three Abiotic factors from the Lion King introduction
5) The bird riding on the tusks of the elephant feed on insects the elephant stirs up. What kind of symbiotic relationship exists between the two?
Write and balance the double replacement reaction between barium sulfate and titanium (II) oxide.
Answers
Answer:
BaSO4(aq) + TiO(aq) —> BaO(aq) + TiSO4(aq)
Explanation
The equation for the reaction between barium sulfate and titanium (II) oxide is given below:
BaSO4 + TiO —>
In solution, the reactants will dissociates as follow:
BaSO4(aq) —> Ba^2+(aq) + SO4^2-(aq)
TiO(aq) —> Ti^2+(aq) + O^2-(aq)
The double displacement reaction will occur as follow:
Ba^2+(aq) + SO4^2-(aq) + Ti^2+(aq) + O^2-(aq) —> Ba^2+O^2-(aq) + Ti^2+SO4^2-(aq)
We can see that a double displacement reaction occurred as there is a double exchange of ions in the solution. The elemental equation is given below:
BaSO4(aq) + TiO(aq) —> BaO(aq) + TiSO4(aq)
2. How many Cu atoms have a mass of 4.500x10^5 amu?
Answers
Answer:
63.55
Explanation:
4.500 x 10^5 amu x 1 Cu atom / 63.55 amu = 7081 Cu Atom
Cu 1 x 63.55 = 63.55
Which of the following terms best reflects the process by which humans store memories ?
Answers
Answer:
there are no term
Explanation:
what are the term
Net ionic equation for potassium sulfide and magnesium iodide
Answers
The net ionic equation for the reaction between potassium sulfide and magnesium iodide is S2- + Mg2+ -> MgS, as the potassium and iodide ions are spectator ions and do not participate in the reaction.
To determine the net ionic equation for the reaction between potassium sulfide (K2S) and magnesium iodide (MgI2), we first need to identify the ions present in each compound and then determine the products formed when they react.
Potassium sulfide (K2S) dissociates into two potassium ions (K+) and one sulfide ion (S2-):
K2S -> 2K+ + S2-
Magnesium iodide (MgI2) dissociates into one magnesium ion (Mg2+) and two iodide ions (I-):
MgI2 -> Mg2+ + 2I-
Now, we need to determine the possible products when these ions combine. Since potassium (K+) has a +1 charge and iodide (I-) has a -1 charge, they can combine to form potassium iodide (KI):
K+ + I- -> KI
Similarly, magnesium (Mg2+) and sulfide (S2-) can combine to form magnesium sulfide (MgS):
Mg2+ + S2- -> MgS
Now, we can write the complete ionic equation by representing all the ions present before and after the reaction:
2K+ + S2- + Mg2+ + 2I- -> 2KI + MgS
To obtain the net ionic equation, we remove the spectator ions, which are the ions that appear on both sides of the equation and do not participate in the actual reaction. In this case, the spectator ions are the potassium ions (K+) and the iodide ions (I-).
Thus, the net ionic equation for the reaction between potassium sulfide and magnesium iodide is:
S2- + Mg2+ -> MgS
For more such questions on ionic equation visit:
https://brainly.com/question/25604204
#SPJ8
Describe how lead as a toxic metal can be determine in borehole water?
Answers
We can be able to determine the amount of toxic lead in the water by thee use of atomic absorption spectrophotometry.
What is a toxic metal?
A toxic metal is known as any metal that is able to affect the health of people. We know that toxic metals are mostly the metals that are in the group of the heavy metals.
Now we know lead as a metal that is able to cause brain damage especially in children. This is why it is very important that there should be a thorough examination in order to know the amount of lead that is present in water.
There are several methods that could be applied in the determination of lead and one of the most common methods is by the use of atomic absorption spectrophotometry which is able to detect even the minutest amount of the led in solution.
Learn more about toxic metals:https://brainly.com/question/28331004
#SPJ1
lphins... Acid. (b) Chlorine reacts with red hot iron powder to give Iron(III) Chloride but not Iron (II) Chloride. Explain. (1Mark)
Answers
(a) Because acid is caustic, dolphins can perish from exposure to it. Acids are compounds that give other things protons (H+). Acid can react with the proteins and lipids in dolphins' skin when they come into touch with it, leading to chemical burns and damage to the underlying tissue. Systemic consequences from this include death.
(b) Because chlorine is a potent oxidizer, it interacts with red-hot iron powder to produce Iron(III) chloride (FeCl3) rather than Iron(II) chloride (FeCl2). FeCl3 is created when chlorine at high temperatures rapidly accepts electrons from iron atoms. Contrarily, iron interacts with HCl, a less potent oxidizer than chlorine, to produce FeCl2.
Learn more about chlorine at :
https://brainly.com/question/31560014
#SPJ1
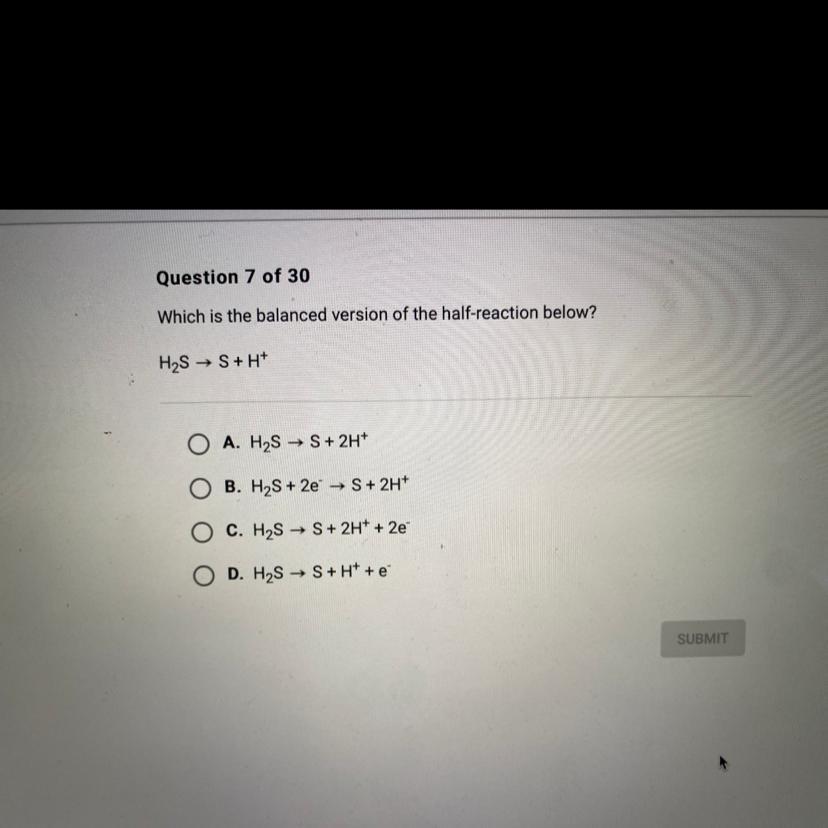
Which is the balanced version of the half-reaction below?
H2S → S+H+
Answers
Answer:
C. \(H_2S\rightarrow S+2H^++2e^-\)
Explanation:
Hello there!
In this case, according to the given chemical reaction, it turns out possible to realize there is one sulfur atom on each side of the chemical equation but two hydrogen atoms on the left and one on the right, which means the latter must be balanced in agreement to the law of conservation of mass.
In such a way, by setting a 2 on H⁺, the reaction will be balanced:
\(H_2S\rightarrow S+2H^+\)
Now, we count the transfer electrons for sulfur from -2 to 0 as 2e⁻ on the right, which will match with the option C.
\(H_2S\rightarrow S+2H^++2e^-\)
Regards!
An old 0.500 L lecture bottle of triethylamine (N(CH₂CH₃)₃) was found in a lab and needed for a synthesis reaction. A pressure regulator indicated a pressure of 18.5 psi, and the lab was at room temperature (25.0°C). What mass of vaporized triethylamine in grams was left in the lecture bottle?
Answers
Mass of vaporized triethylamine : 2.606 g
Further explanationGiven
0.5 L triethylamine
P = 18.5 psi
T = 25 °C
Required
mass of vaporized triethylamine
Solution
Conversion :
P 18.5 psi = 1,26 atm
T = 25 +273 = 298 K
Ideal gas law :
PV=nRT
n = PV/RT
Input the value :
n = (1.26 atm x 0.5 L) /(0.08205 x 298)
n = 0.0258
MW triethylamine = 101 g/mol
Mass triethylamine :
= n x MW
= 0.0258 x 101 g/mol
= 2.606 g
(c) 45 g C,H, react with 45 g Cl₂ according to the equation:
Cl₂ + C6H6 C6H5Cl + HCI. What is the limiting reactant? What mass of HCI will be produced?
-
Answers
In the given reaction, the limiting reactant is C₆H₆ (benzene).
To determine the limiting reactant as well as calculate the mass of HCl produced, compare the moles of each reactant.
The number of moles for each reactant:
Molar mass of Cl₂ = 35.5 g/mol + 35.5 g/mol = 71 g/mol
Moles of Cl₂ = mass of Cl₂ / molar mass of Cl₂
= 45 g / 71 g/mol
= 0.634 moles of Cl₂
Molar mass of C₆H₆ (benzene) = 12 g/mol + 6(1 g/mol) = 78 g/mol
Moles of C₆H₆ = mass of C₆H₆ / molar mass of C₆H₆
= 45 g / 78 g/mol = 0.577 moles of C₆H₆
Determine the stoichiometry between Cl₂ and HCl:
Cl₂ + C₆H₆ → C₆H₅Cl + HCl
Here, we can see that 1 mole of Cl₂ produces 1 mole of HCl.
Thus, the limiting reactant is C₆H₆ (benzene).
Calculate the mass of HCl produced:
Molar mass of HCl = 1 g/mol + 35.5 g/mol = 36.5 g/mol
Moles of HCl produced = moles of C₆H₆ = 0.577 moles
Mass of HCl produced = moles of HCl produced × molar mass of HCl
Mass of HCl produced = 0.577 moles × 36.5 g/mol
≈ 21.04 g
Therefore, approximately 21.04 grams of HCl will be produced.
For more details regarding limiting reactant, visit:
https://brainly.com/question/10090573
#SPJ1