11. Which of the following best explains why CO2 gas is easily compressible but solid
CO2 (dry ice) is incompressible?
The molecules of CO2 gas are much closer together than
the molecules in dry ice.
The molecules of CO2 gas are much smaller than the molecules
of solid CO2
O
The molecules of solid CO2 are much closer together than
the molecules of CO2 gas.
The molecules of CO2 gas attract one another, while the
molecules of the solid CO2 repel one another.
0
Answers
Answer:
The right answer is C. The molecules of solid CO2 are much closer together than the molecules of CO2 gas.
The molecules of CO2 gas are much closer together than the molecules in dry ice. So CO2 gas is easily compressible but solid CO2 is incompressible. The correct option is A.
What is dry ice?Carbon dioxide in its solid form is known as dry ice. Because CO2 does not have a liquid state at normal atmospheric pressure and sublimates directly from the solid to the gas state, it is commonly used for temporary refrigeration.
Dry ice can be a serious hazard in a small, poorly ventilated space. When dry ice melts, it emits carbon dioxide gas.
This gas can accumulate in a small space. A person can become unconscious and, in some cases, die if there is enough carbon dioxide gas present.
CO2 gas molecules are much closer together than dry ice molecules. As a result, CO2 gas is easily compressible, but solid CO2 is not.
Thus, the correct option is A.
For more details regarding dry ice, visit:
https://brainly.com/question/13972158
#SPJ2
Related Questions
What is the equilibrium constant of aA+bB cC +dD?
Answers
Explanation:
{c}^C . {d}^D / {a}^A . {b}^B
How many moles of oxygen
react with 37.5 moles of sulfur
dioxide in the production of sulfur
trioxide gas?

Answers
Answer:
1 L of O2 reacts with 2 L of SO2.
Explanation:
From the balanced equation, 1 mol of oxygen
reacts with 2 mol sulfur dioxide.
use molecular orbital (mo) diagrams to rank b22 , b2, and b2− in order of increasing bond order, bond energy, and bond length.
Answers
The order of increasing bond length is B22 > B2 > B2-.In summary, the order of increasing bond order is B22 < B2 < B2-, the order of increasing bond energy is B22 < B2 < B2-, and the order of increasing bond length is B22 > B2 > B2-.
Molecular orbital (MO) diagrams are used to assess the bonding in a molecule and provide information about bond order, bond energy, and bond length. In this question, we have to rank B22, B2, and B2- in order of increasing bond order, bond energy, and bond length using MO diagrams.
Bond order: Bond order refers to the number of chemical bonds between two atoms. It is determined by the number of bonding electrons minus the number of antibonding electrons divided by two. A higher bond order indicates stronger bonding between two atoms. B22 has a bond order of 1, B2 has a bond order of 1, and B2- has a bond order of 2. Therefore, the order of increasing bond order is B22 < B2 < B2-.
Bond energy: Bond energy refers to the energy required to break a chemical bond. A higher bond energy indicates a stronger bond. B22 has the weakest bond and the smallest bond energy because it is composed of two atoms in the ground state, which do not bond. B2 has a slightly stronger bond than B22, but the bond energy is still low. B2- has the strongest bond because it has the highest bond order. Therefore, the order of increasing bond energy is B22 < B2 < B2-.
Bond length: Bond length refers to the distance between the nuclei of two bonded atoms. A shorter bond length indicates a stronger bond. B22 has the largest bond length since it has no bond. B2 has a slightly shorter bond length than B22. B2- has the shortest bond length since it has the highest bond order.
Therefore, the order of increasing bond length is B22 > B2 > B2-.In summary, the order of increasing bond order is B22 < B2 < B2-, the order of increasing bond energy is B22 < B2 < B2-, and the order of increasing bond length is B22 > B2 > B2-.
To learn more about energy visit;
https://brainly.com/question/1932868
#SPJ11
How to convert acetone into methanal?
Answers
Acetone to formaldehyde, formaldehyde to acetone. Methyl magnesium bromide is used to cure formaldehyde in the presence of dry ether, producing ethanol after acid hydrolysis and isopropyl alcohol.
Thus, Acetaldehyde is produced when ethanol is heated with copper at 373 K and is oxidized. Isopropyl alcohol is produced by treating acetaldehyde with methyl magnesium bromide while dry ether is present.
Acet is produced when isopropyl alcohol is heated with copper at 373 kelvin.
In 2010, around 6.7 million tonnes were manufactured globally, primarily for use as a solvent and for the synthesis of bisphenol A and methyl methacrylate, which are precursors to common isopropyl alcohol.
Thus, Acetone to formaldehyde, formaldehyde to acetone. Methyl magnesium bromide is used to cure formaldehyde in the presence of dry ether, producing ethanol after acid hydrolysis and isopropyl alcohol.
Learn more about Isopropyl alcohol, refer to the link:
https://brainly.com/question/14896958
#SPJ1
What did Thomson contribute to atomic theory?
A. The discovery that positive charge is concentrated in a core, or nucleus
B. The discovery that negative charge is concentrated in a core, or nucleus
C. The discovery of negatively charged particles, now called electrons
D. The idea that electrons have quantized amounts of energy
Answers
When the [CO2] and [H2CO3] are both horizontal lines, the rate of the forward reaction is
the rate of the reverse reaction
faster than
slower than
the same as
Answers
When \(CO_{2}\) and \(H_{2} CO_{3}\) are both horizontal lines, the rate of the forward reaction is the same as the rate of the reverse reaction. The reaction is occurring at equilibrium, with no net change in the concentrations of reactants and products over time.
When the concentration of carbon dioxide \(CO_{2}\) and the concentration of carbonic acid \(H_{2} CO_{3}\) are both horizontal lines, it indicates that their concentrations remain constant over time. In such a scenario, the rate of the forward reaction is the same as the rate of the reverse reaction. A horizontal line on a concentration-time graph suggests that the concentrations of the reactants and products are not changing, implying that the reaction has reached equilibrium. At equilibrium, the rate of the forward reaction equals the rate of the reverse reaction. This is a fundamental principle of chemical equilibrium, described by the principle of microscopic reversibility.
For more question on reaction
https://brainly.com/question/25769000
#SPJ8
explain how the direction of electron flow in a voltaic cell is consistent with what you would predict from the activity series
Answers
In voltaic cells, electrons flow from the negative electrode (anode) to the positive electrode (cathode).
Electrons are moved from one species to another in redox reactions. When a reaction occurs spontaneously, energy is released that can be put to good use. The process must be divided into the oxidation reaction and the reduction reaction in order to capture this energy. A wire is used to move the electrons from one side of the reactions to the other after they have been placed into two separate containers. A voltaic/galvanic cell is produced as a result.
To know more about voltaic/galvanic cell, visit;
brainly.com/question/28051381
#SPJ4
Which explains the law of conservation of mass?
a. Every reaction creates an equal amount of mass related to the amount of
energy required for the reaction.
b. The total mass before a chemical reaction is the same as the total mass after
the reaction.
c. Matter can not be conserved.
d. The total amount of mass is equal to the volume of both chemicals in the
reaction.
Answers
Answer:
A is your answer.
Explanation:
This law states that, despite chemical reactions or physical transformations, mass is conserved — that is, it cannot be created or destroyed — within an isolated system. In other words, in a chemical reaction, the mass of the products will always be equal to the mass of the reactants.
given the following steady-state kinetic data for an enzyme catalyzed reaction in the presence (molecule a) and absence of an inhibitor, what type of inhibitor is molecule a?
Answers
If the presence of molecule a decreases the reaction rate and increasing substrate concentration does not overcome the inhibition, it is likely a noncompetitive or uncompetitive inhibitor.
To determine the type of inhibitor molecule a is, we need to first analyze the steady-state kinetic data. If the presence of molecule a decreases the rate of the enzyme-catalyzed reaction, it is likely an inhibitor.
Next, we need to look at the effect of increasing concentrations of substrate on the reaction rate in the presence and absence of molecule a. If molecule a is a competitive inhibitor, increasing substrate concentration can overcome the inhibition because the inhibitor and substrate are competing for the same active site on the enzyme. Therefore, the reaction rate will increase with increasing substrate concentration in the presence of molecule a.
On the other hand, if molecule a is a noncompetitive or uncompetitive inhibitor, increasing substrate concentration will not overcome the inhibition because the inhibitor binds to a different site on the enzyme than the substrate. Therefore, the reaction rate will not increase with increasing substrate concentration in the presence of molecule a.
Overall, if the presence of molecule a decreases the reaction rate and increasing substrate concentration does not overcome the inhibition, it is likely a noncompetitive or uncompetitive inhibitor. However, if increasing substrate concentration does overcome the inhibition, it is likely a competitive inhibitor.
For more such questions on reaction rate , Visit:
https://brainly.com/question/24795637
#SPJ11
Which atom has the highest ionization energy?
O K
O zn
O Se
O Ca
Answers
The least amount of energy necessary to liberate the most loosely bound electron from an isolated gaseous atom, positive ion, or molecule is known as the ionization energy.
Which atom has the highest ionization energy?
KZnSeCaSe (Ionization energy 97542) has the highest out of Potassium, Zinc and Calcium.
A chemical element with the atomic number 34 and the symbol Se is selenium. It is a nonmetal (occasionally referred to as a metalloid) that shares characteristics with arsenic and has features halfway between sulfur and tellurium in the periodic table. In the crust of the Earth, it hardly ever exists in either its elemental state or as pure ore complexes.
The energy required to remove one mole of electrons from an atom and produce a positively charged selenium ion is known as the ionization of selenium energy.
Se -> Se+ + e-
Although this procedure can be performed numerous times, the energy cost rises sharply each time. The selenium's general equation is:
SeN+ -> Se(N+1)+ + e-
To know further information about ionization energy, click on the link below:
https://brainly.com/question/20275162
#SPJ9
1. A substance has a mass of 5.55 g and a volume of 3.00 cm”. Determine the density of this
substance and then use Table Sto identify the substance.
I need help please
Answers
Answer:
1.85 g per cm^3
The substance is Beryllium(if it is a single element, and not a compound)
Explanation:
How many Phosphorous (P) are in the product?
Answers
Answer:
5
Explanation:
1. A sodium ion has 11 protons and 10 electrons. What is its charge?
Answers
+1
If sodium loses an electron, it now has 11 protons, 11 neutrons, and only 10 electrons, leaving it with an overall charge of +1. It is now referred to as a sodium ion.
he scientist had a 35% saline solution that he mixed with 10 milliliters of a 75% saline solution to get a 40% saline solution. How many milliliters of the 35% solution were used? a) 30 milliliters b) 40 milliliters c) 50 milliliters d) 60 milliliters e) 70 milliliters
Answers
The answer is (e) 70 milliliters.
Let's assume that x milliliters of the 35% saline solution were used.
The total amount of saline in the solution can be calculated as follows:
Saline in 35% solution = 0.35 * x
Saline in 75% solution = 0.75 * 10 (since 10 milliliters of the 75% solution were used)
The total amount of saline in the resulting 40% solution can be calculated as:
Saline in 40% solution = 0.40 * (x + 10)
Since the saline is being mixed, the total saline in the resulting solution is equal to the sum of the saline in the individual solutions:
0.35 * x + 0.75 * 10 = 0.40 * (x + 10)
Simplifying the equation:
0.35x + 7.5 = 0.40x + 4
Subtracting 0.35x and 4 from both sides:
7.5 - 4 = 0.40x - 0.35x
3.5 = 0.05x
Dividing both sides by 0.05:
x = 3.5 / 0.05
x = 70
Therefore, 70 milliliters of the 35% saline solution were used. The answer is (e) 70 milliliters.
For more details regarding the saline solution, visit:
https://brainly.com/question/24498665
#SPJ4
Which compound has the strongest hydrogen bonding at STP?
1. H2O 3. H2Se
2. H2S 4. H2Te
Answers
Answer: H2O
Explanation:
The strongest hydrogen bond type is the one involving a bond between oxygen, nitrogen and fluorine, with a hydrogen atom. Since in the choices, only water shows a O-H bond, therefore this is the strongest hydrogen bonding. The answer is water.
Number 1.17?? Please :)

Answers
Answer:
In 7.3288, digit is on first place after decimal, so its place value will be tenths. Digit is on second place after decimal, so its place value will be hundredths. Bold digit is on third place after decimal, so its place value will be thousandths.
PLEASE HELP ASAP
*Please complete all calculations on a piece of loose leaf. Use the factor label method. BOX ANSWERS
Ammonia reacts with oxygen to form nitrogen monoxide gas and water via the following balanced reaction:
4HN, + 50, → 4NO + 6H2O
If 6.95 g of ammonia (HN,) reacts with 2.70 g of oxygen (O2), answer the following questions:
a) Identify the limiting reagent/reactant and identify what mass of water (H,) can be produced from the
reaction? (4 pts)
b) What mass of excess reagent/reactant is required for the reaction to take place? (2 pts)
c) What mass of excess reagent/reactant is left over after the reaction has gone to completion? (2 pts)
Answers
Answer:
you can see the answer at the pic
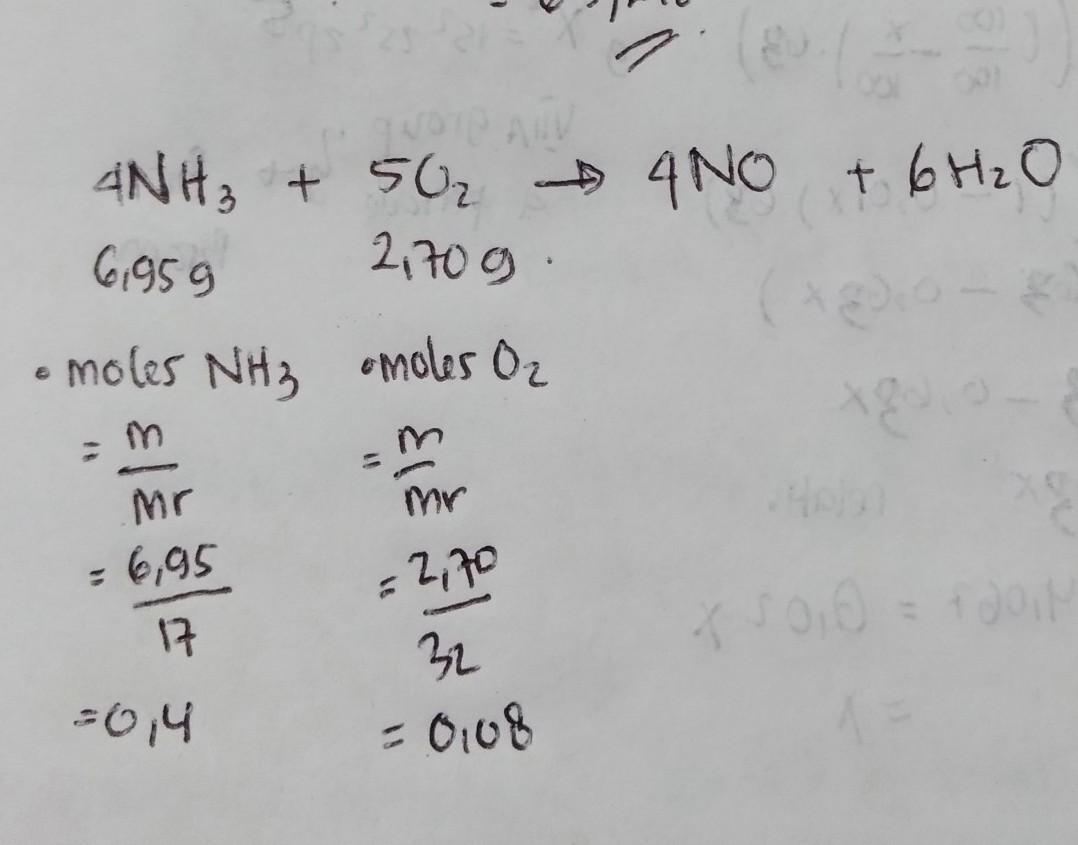

Please Help Me Balance The equation!! will mark brainliest

Answers
Answer: XCl2(aq) + 2 AgNO3(aq) ---> X(NO3)2(aq) + 2 AgCl(s)
Explanation:
Hehe I remembered how...

Sand dunes are large piles of of sand that can be found in the dessert. What causes these sand dunes to form
A. Ice
B. Wind
C. Earthquakes
D. Volcanoes
Answers
Explanation: fun fact: wind also moves rocks
calculate the energy processed by a single proton of the each following types electromagnetic radiation:
a.6.32 x 10^20 s-1
b. 9.50 x 10^13 hz
c.1.05 x10^16 s^-1
#G10 Chemistry
Answers
a. 4.2 x 10⁻¹³ J
b. 6.3 x 10⁻²⁰ J
c. 7 x 10⁻¹⁸ J
Further explanationRadiation energy is absorbed by photons
The energy in one photon can be formulated as
\(\large{\boxed{\bold{E\:=\:h\:.\:f}}}\)
Where
h = Planck's constant (6,626.10⁻³⁴ Js)
f = Frequency of electromagnetic waves
a.
\(\tt E=6.626.10^{-34}\times 6.32.10^{20}=4.1876.10^{-13}~J\)
b.
\(\tt E=6.626.10^{-34}\times 9.50.10^{13}=6.2947.10^{-20}~J\)
c.
\(\tt E=6.626.10^{-34}\times 1.05.10^{16}=6.9573.10^{-18}~J\)
Molar mass of 500g of Cl2
Answers
Explanation:
What's Your Question ??..
How might knowing the indicators of a chemical reaction help you explain what happened when the sulfuric acid was mixed with powdered sugar? How does this relate to the rearrangement of atoms that occurs in a chemical reaction? Record your evidence.
Answers
Answer:
I don't know I'm sorry I will tell you another
WHAT IS THE ROLE OF THE CHARTERER, BROKER AND THE VESSEL OWNER
IN THE CHARTERING PROCESS? PLEASE PROVIDE THREE EXAMPLES.
Answers
It is the charterer's duty to hire a ship for a certain period of time to transport cargo or passengers.
The charterer's job is to hire a ship for a set amount of time to transport goods or people. They are in charge of negotiating the charter conditions with the vessel owner or broker, setting up the required licences and paperwork, and ensuring the timely and safe delivery of cargo or passengers. Broker: The broker's job is to make it easier for the charterer and the vessel owner to come to an agreement. They serve as middlemen, using their connections and market expertise to identify suitable vessels and agree upon conditions of charter that are acceptable to both sides. A appropriate vessel must be provided by the vessel owner in order to satisfy the charterer's needs. They keep the vessel seaworthy, adhere to pertinent laws, and guarantee effective operations throughout the charter.
To know more about charter, here:
https://brainly.com/question/27998149
#SPJ4
What are ancient plants and animal formed into rocks called it is a 7 letter word that starts with f
Answers
Answer:
Fossils
Explanation:
Ancient plants and animal formed into rocks called fossils.
A fossil can be defined as the mineral impression or remains of living organisms such as plants and animals that are prehistoric in nature.
A geologic column can be defined as a collection of rock layers that are typically grouped based on their relative ages in a descending order i.e from oldest to the youngest.
Generally, the layers of the rock that are found in various parts of the world are known to be different because they all comprises of distinct Earth's geological history (fossils).
8. Study the given table and answer the following questions. i) Name metals and non-metals. Elements ii) What is the valency of A and why? iii) Write the name and symbol of all the elements. iv) Write down the molecular formula of the compound formed by the combination of A and B; and C and B. A B C D Electronic configuration 2,8,1 2,8,7 2,8,8,2 2,8 v) Which element is more active between A and D? vi) Name the elements which can lose the valence electron to become stable.
Answers
i) Metals: A and D
Non-metals: B and C
ii) The valency of A is 1. This is because A has one valence electron, and elements in Group 1 (such as A) typically have a valency of 1 as they tend to lose that single valence electron to achieve a stable electron configuration.
iii)
A: Sodium (Na) - Symbol: Na
B: Chlorine (Cl) - Symbol: Cl
C: Oxygen (O) - Symbol: O
D: Calcium (Ca) - Symbol: Ca
iv) The compound formed by the combination of A and B: Sodium Chloride (NaCl) The compound formed by the combination of C and B: Oxygen Chloride (OCl2)
v) Element D (Calcium) is more active than element A (Sodium). This is because elements in Group 2 (such as D) tend to lose two valence electrons, which requires less energy compared to losing only one valence electron (as in the case of A).
vi) The elements that can lose the valence electron to become stable are A (Sodium) and D (Calcium).
Metals: A, B, C, D; Non-metals: None. Elementsii) The valency of A is 1 because it has only one valence electron.iii) The name and symbol of all the elements are:A - Sodium (Na)B - Chlorine (Cl)C - Argon (Ar)D - Calcium (Ca)iv) The molecular formula of the compound formed by the combination of A and B is NaCl. The molecular formula of the compound formed by the combination of C and B is BCl2.
v) A is more active than D because A is a metal and metals are more active than non-metals. A tends to lose electrons more easily than vi) The elements which can lose the valence electron to become stable are metals.
i) Metals: A and D Non-metals: B and C ii) The valency of A is 1. This is because A has one valence electron, and elements in Group 1 (such as A) typically have a valency of 1 as they tend to lose that single valence electron to achieve a stable electron configuration. iii) A: Sodium (Na) - Symbol: Na B: Chlorine (Cl) - Symbol: Cl C: Oxygen (O) - Symbol: O D: Calcium (Ca) - Symbol: Ca
iv) The compound formed by the combination of A and B: Sodium Chloride (NaCl) The compound formed by the combination of C and B: Oxygen Chloride (OCl2) v) Element D (Calcium) is more active than element A (Sodium). This is because elements in Group 2 (such as D) tend to lose two valence electrons, which requires less energy compared to losing only one valence electron (as in the case of A). vi) The elements that can lose the valence electron to become stable are A (Sodium) and D (Calcium).
for such more questions on electrons
https://brainly.com/question/26084288
#SPJ8
The characteristic which distinguishes a true solid from other phases of matter at STP is that in a true solid the particles are 1 vibrating and changing their relative positions by vibrating without changing their relative positions 2 3 motionless but changing their relative positions 4 motionless without changing their relative positions.
Answers
Answer:
Background
Solids – relatively rigid, definite volume and shape. In a solid, the atoms and molecules are attached to each other. ...
Liquids – definite volume but able to change shape by flowing. In a liquid, the atoms and molecules are loosely bonded. ...
Gases – no definite volume or shape
All objects made of matter create what pull?
Answers
Answer:
energy
that seems like the most relevant answer
Cellular respiration uses sugar and _________ to produce carbon dioxide and water
Answers
Why are cations positive?
Answers
Answer:
A cation has more protons than electrons, consequently giving it a net positive charge.
Answer:
A cation has more protons than electrons, making it a net positive charge as a result. One or two electrons must be lost in order for a cation to form, normally dragged away by atoms with a greater affinity for them The battery pumps electrons away from the anode and into the cathode . The positive anode draws anions to it while the adverse cathode draws cations to it.
Explanation:
Hope this helps!
Anatom of an element has no electron,will that atom have any mass or not? Can antom exist
without electron? if so give an example.
Answers
Answer:
Generally, all atoms of elements contain three sub-atomic particles- Protons(Positively-charged), Neutrons(Neutral) and Electrons(Negatively-charged).
The Magnitude of the Positive Charge possessed by the Protons and the Magnitude of the Negative Charge possessed by the Electrons cancel each other, which make the atom electrically neutral.
Hence,
Without any electrons, the magnitude of the positive and negative charges wouldn't balance, and the atom would be unstable and disintegrate almost immediately. Hence, all stable atoms should have electrons revolving around them, where the number of protons and electrons are the same, under normal conditions.
To Conclude,
Every atom of an element will have electrons arranged in different shells, or matter couldn't exist without electrons.